Advanced Biocatalytic Synthesis of Chiral Intermediates for Scalable Pharmaceutical Production
The pharmaceutical industry continuously seeks robust, cost-effective, and environmentally sustainable pathways for the synthesis of chiral building blocks, particularly those serving as critical precursors for high-value active pharmaceutical ingredients (APIs). Patent CN1228448C introduces a groundbreaking biocatalytic methodology for the preparation of (R)-2-halo-1-(substituted phenyl) ethanol, a pivotal chiral intermediate utilized in the manufacture of Formoterol, a potent long-acting beta-2 receptor agonist used in asthma management. This patent discloses a novel approach that leverages the stereoselective power of yeast enzymes to reduce substituted phenyl ethanones, achieving high optical purity without the reliance on hazardous chemical reducing agents. By shifting from traditional chemical synthesis to biological catalysis, this technology addresses key pain points in modern drug manufacturing, including reagent toxicity, waste generation, and the prohibitive costs associated with chiral resolution. For R&D directors and procurement specialists alike, understanding the nuances of this yeast-mediated reduction is essential for optimizing supply chains and ensuring the consistent quality of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral intermediates like (R)-2-bromo-1-(4-benzyloxy-3-nitrophenyl) ethanol has relied heavily on chemical reduction strategies or classical resolution techniques, both of which present significant industrial drawbacks. As detailed in prior art such as WO 00/21487, the use of amino alcohol catalysts in conjunction with borane (BH3) as a reducing agent creates a hazardous operational environment due to the volatility and high reactivity of borane. Furthermore, the catalyst systems required for these chemical reductions are often synthetically complex and expensive to produce, driving up the overall cost of goods. Alternatively, resolution methods disclosed in documents like CN 1063422C involve reacting racemic mixtures with resolving agents like phthalic anhydride, a process that inherently limits the maximum theoretical yield to 50% unless dynamic kinetic resolution is employed. These traditional routes also necessitate the use of multiple solvents and expensive alkaloids for separation, generating substantial chemical waste and complicating the purification process, which is untenable for green chemistry initiatives.
The Novel Approach
In stark contrast, the methodology outlined in CN1228448C utilizes a simple, eco-friendly yeast catalytic system that operates under mild conditions, typically between 1°C and 50°C. This biological approach eliminates the need for dangerous borane reagents and complex synthetic catalysts, replacing them with readily available, inexpensive yeast and a carbon source such as glucose. The process demonstrates remarkable versatility, accommodating various halogen substituents (Cl, Br, I) and functional groups (NO2, OMe, OH) on the phenyl ring while maintaining high stereoselectivity. By employing whole-cell biocatalysis, the reaction achieves enantiomeric excess (ee) values ranging significantly from 80% to 98%, depending on the specific substrate and conditions, thereby minimizing the formation of unwanted isomers. This shift not only simplifies the operational workflow to basic mixing and filtration but also drastically reduces the environmental footprint by utilizing biodegradable catalysts and potentially aqueous solvent systems.
Mechanistic Insights into Yeast-Catalyzed Asymmetric Reduction
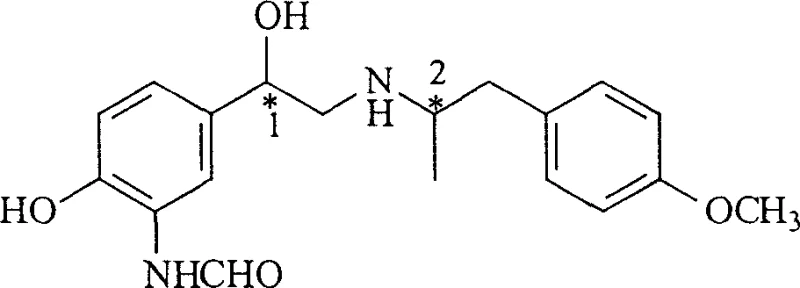
The core mechanism driving this synthesis is the enzymatic asymmetric reduction of the ketone carbonyl group by oxidoreductases present within the yeast cells. These enzymes, primarily alcohol dehydrogenases, utilize cofactors like NADPH to transfer hydride ions to the prochiral ketone substrate with high spatial precision. The yeast cell acts as a natural chiral pool, providing the necessary stereochemical environment to favor the formation of the (R)-enantiomer over the (S)-enantiomer. This biological specificity is crucial because, as seen in the final drug Formoterol, the pharmacological activity is heavily dependent on the absolute configuration of the chiral centers. The patent data indicates that the (R,R) isomer possesses significantly higher bronchodilator activity compared to other diastereomers, making the control of the first chiral center during the intermediate stage vital for downstream efficacy.
Furthermore, the impurity profile in this biocatalytic process is inherently cleaner than in chemical reductions. Because the enzyme active sites are highly specific, side reactions such as dehalogenation or over-reduction are minimized, leading to a crude product that requires less intensive purification. The patent examples demonstrate that even with diverse substrates, such as 2-iodo-1-(4-methoxy-3-bromophenyl) ethanone or 2-chloro-1-(4-hydroxyphenyl) ethanone, the yeast system maintains consistent performance. The reaction conditions, involving a mixture of yeast, glucose, and solvents like water, ethanol, or toluene, create a biphasic or homogeneous environment that facilitates substrate uptake while protecting the sensitive enzymatic machinery. This robustness allows for the processing of substrates with varying solubilities and electronic properties, ensuring that the stereochemical integrity is preserved throughout the transformation from ketone to chiral alcohol.
How to Synthesize (R)-2-halo-1-(substituted phenyl) ethanol Efficiently
The operational protocol for this synthesis is designed for simplicity and scalability, requiring standard laboratory or plant equipment without the need for specialized high-pressure or cryogenic setups. The process begins with the preparation of a reaction medium where fresh or dry yeast is suspended with glucose and a chosen solvent, creating a biologically active slurry. The substrate, a 2-halo-1-(substituted phenyl) ethanone, is then introduced at a controlled loading rate to prevent substrate inhibition, allowing the yeast enzymes to effectively reduce the ketone functionality over a period of 1 to 10 days. Following the reaction completion, the biomass is removed via diatomite filtration, and the product is extracted into an organic phase, dried, and purified through recrystallization to achieve the desired pharmaceutical grade purity. For a detailed, step-by-step breakdown of the specific ratios and conditions optimized for different substrates, please refer to the standardized synthesis guide below.
- Prepare the reaction mixture by combining yeast, glucose, and a suitable solvent (such as water, ethanol, or buffered solutions) in a weight ratio of approximately 1: 0-5:10-50.
- Add the substrate, 2-halo-1-(substituted phenyl) ethanone, to the mixture at a loading of 0.8 to 2 mmol per 100 grams of the mixture, ensuring thorough mixing.
- Maintain the reaction temperature between 1°C and 50°C for a duration of 1 to 10 days, followed by filtration, extraction, and recrystallization to isolate the pure chiral product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this yeast-catalyzed route offers transformative benefits for procurement managers and supply chain heads focused on cost efficiency and risk mitigation. The primary driver of value is the drastic reduction in raw material costs; yeast is an abundant, commodity-grade biological material that costs a fraction of the price compared to specialized chiral ligands or borane reagents. Additionally, the elimination of hazardous chemicals simplifies regulatory compliance and lowers the costs associated with safety infrastructure and waste disposal. The process's reliance on mild temperatures and ambient pressure further reduces energy consumption, contributing to a lower overall carbon footprint and aligning with corporate sustainability goals. These factors combine to create a supply chain that is not only more economical but also more resilient to fluctuations in the pricing of exotic chemical reagents.
- Cost Reduction in Manufacturing: The replacement of expensive borane reducing agents and complex amino alcohol catalysts with inexpensive yeast and glucose results in a significant decrease in direct material costs. Since the catalyst is biodegradable and does not require complex recovery systems, the downstream processing costs are also substantially lowered. This economic efficiency allows manufacturers to offer high-purity pharmaceutical intermediates at a more competitive price point, enhancing margin potential for downstream API producers.
- Enhanced Supply Chain Reliability: Sourcing yeast and glucose is far more stable and predictable than sourcing specialized chiral catalysts, which may have limited suppliers and long lead times. The robustness of the biological system means that production can be scaled up rapidly to meet demand surges without the bottleneck of catalyst synthesis. This reliability ensures continuous supply continuity for critical asthma medications, reducing the risk of stockouts and production delays in the global pharmaceutical market.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste, as the primary byproduct is biological biomass that can be easily disposed of or treated. This simplifies the environmental permitting process and reduces the liability associated with toxic waste management. The simplicity of the operation—essentially mixing and stirring—makes it highly scalable from pilot plants to multi-ton commercial reactors, facilitating rapid technology transfer and capacity expansion.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biocatalytic technology in industrial settings. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of using yeast catalysis over borane reduction for chiral intermediates?
A: Yeast catalysis eliminates the need for hazardous and expensive reagents like borane (BH3), significantly reducing raw material costs and safety risks while offering high enantiomeric excess (ee) values ranging from 80% to 98%.
Q: How does this process impact environmental compliance in pharmaceutical manufacturing?
A: The process utilizes biodegradable catalysts (yeast) and often employs water or benign solvents, avoiding heavy metal contamination and simplifying waste treatment compared to traditional chemical resolution methods.
Q: Is this synthesis method scalable for industrial production of Formoterol intermediates?
A: Yes, the mild reaction conditions (1-50°C) and simple operational steps (mixing and filtration) make the process highly amenable to scale-up from laboratory to commercial tonnage without complex equipment requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-2-halo-1-(substituted phenyl) ethanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development of life-saving respiratory medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of (R)-2-halo-1-(substituted phenyl) ethanol meets the exacting standards required for GMP API synthesis. Our commitment to technological excellence allows us to deliver products with consistent stereochemical integrity, supporting our partners in maintaining their own quality benchmarks.
We invite pharmaceutical companies and contract manufacturers to engage with our Customized Cost-Saving Analysis service to explore how this yeast-catalyzed route can optimize your specific supply chain. By partnering with us, you gain access to deep technical expertise and the ability to request specific COA data and route feasibility assessments tailored to your project needs. Contact our technical procurement team today to discuss how we can support your next-generation asthma therapy projects with reliable, cost-effective, and sustainable intermediate solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
