Advanced One-Pot Synthesis of Spiro-Xanthene Derivatives for High-Performance OLED Manufacturing
The landscape of organic optoelectronic materials is undergoing a significant transformation, driven by the demand for more efficient and scalable synthetic routes for high-performance host materials. Patent CN1821239A introduces a robust preparation method for xanthene compounds, specifically targeting the synthesis of spiro-structured architectures that are pivotal in the fabrication of organic light-emitting diodes (OLEDs). This technology represents a departure from traditional multi-step syntheses, offering a streamlined one-pot condensation strategy that utilizes strong acid catalysis to couple ketones or aldehydes with phenolic derivatives. The core innovation lies in its ability to construct complex spiro-frameworks, such as spiro[fluorene-9,9'-xanthene], with exceptional purity levels exceeding 99% as verified by GCMS and NMR analysis. For R&D directors and process chemists, this patent outlines a pathway that not only simplifies the molecular construction of rigid, amorphous materials but also addresses critical bottlenecks in yield optimization and impurity profiling.
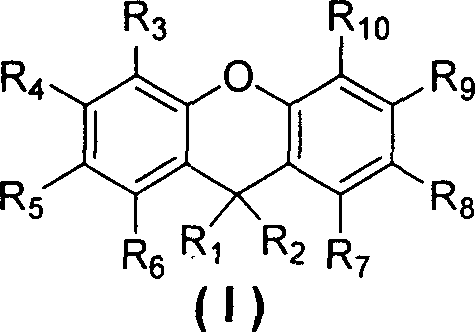
Furthermore, the versatility of this synthetic approach allows for extensive structural modulation through the selection of various substituents on the phenolic and ketone starting materials. The general formula presented in the patent encompasses a wide range of potential derivatives, including those substituted with halogens, alkyl chains, and diverse aryl groups, thereby enabling the fine-tuning of electronic properties such as HOMO-LUMO energy levels and thermal stability. This adaptability is crucial for developing next-generation display materials where precise control over charge transport and emission efficiency is paramount. By establishing a generalized protocol that accommodates steric bulk and electronic diversity, this method provides a foundational platform for the rapid prototyping and subsequent commercialization of novel electronic chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of spiro-xanthene derivatives has been plagued by procedural complexity and economic inefficiency, often requiring multiple discrete reaction steps that involve harsh conditions and expensive reagents. Traditional routes frequently rely on transition metal-catalyzed cross-coupling reactions or multi-stage cyclization processes that necessitate rigorous purification at each intermediate stage to prevent the accumulation of impurities. These conventional methodologies often suffer from low overall yields due to material loss during isolation and purification, significantly driving up the cost of goods sold (COGS) for the final API or electronic material. Additionally, the use of heavy metal catalysts introduces a substantial burden on the supply chain, as it mandates specialized equipment for metal scavenging and generates hazardous waste streams that require costly disposal protocols. The cumulative effect of these factors is a prolonged lead time and reduced agility in responding to market demands for high-purity optoelectronic intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in CN1821239A leverages a direct acid-catalyzed condensation that consolidates the entire synthetic sequence into a single reactor vessel, effectively eliminating the need for intermediate isolation. By employing strong acids such as methanesulfonic acid or trifluoromethanesulfonic acid in conjunction with an excess of phenol, the reaction proceeds efficiently at temperatures ranging from 80°C to 200°C to form the desired spiro-center. This one-pot strategy drastically reduces the operational footprint and solvent consumption, as the reaction mixture can be directly quenched and crystallized after completion. The patent examples demonstrate that this approach is highly effective for synthesizing both unsubstituted and halogenated spiro-compounds, achieving isolated yields between 70% and 82% with purity greater than 99% after simple recrystallization. This represents a paradigm shift towards greener chemistry principles, where atom economy and process intensification are prioritized to enhance manufacturing sustainability.
Mechanistic Insights into Acid-Catalyzed Spiro-Cyclization
The underlying mechanism of this transformation involves a sequential electrophilic aromatic substitution followed by an intramolecular dehydration cyclization, driven by the strong protonating capability of the sulfonic acid catalyst. Initially, the carbonyl group of the ketone or aldehyde substrate is activated by protonation, increasing its electrophilicity and facilitating nucleophilic attack by the electron-rich phenol ring. This initial addition forms a carbinol intermediate, which subsequently undergoes dehydration to generate a reactive carbocation species. The second equivalent of phenol then attacks this carbocation at the ortho-position relative to the hydroxyl group, setting the stage for the final ring-closing step. The formation of the spiro-center is thermodynamically favored due to the release of ring strain and the stabilization provided by the conjugated xanthene system, resulting in a rigid three-dimensional structure that inhibits molecular aggregation in the solid state.
From an impurity control perspective, the use of excess phenol serves a dual purpose: it drives the equilibrium towards product formation according to Le Chatelier's principle and acts as a scavenger for potential side reactions. The high purity observed in the final products (>99%) suggests that the reaction conditions are highly selective, minimizing the formation of regioisomers or oligomeric byproducts that are common in Friedel-Crafts type reactions. The subsequent recrystallization from ethyl acetate further refines the product quality by exploiting solubility differences between the target spiro-compound and any residual starting materials or minor impurities. This robust purification capability is essential for electronic applications, where trace impurities can act as quenching sites that degrade device performance and longevity, making the mechanistic understanding of this crystallization behavior vital for process validation.
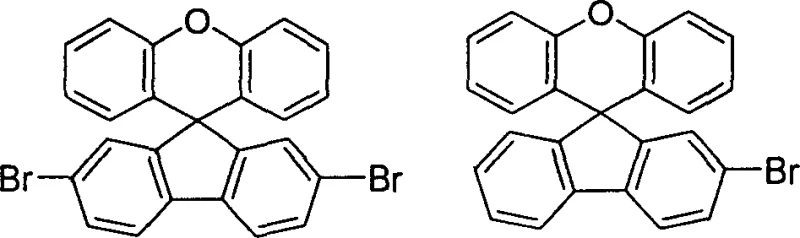
How to Synthesize Spiro[fluorene-9,9'-xanthene] Efficiently
The synthesis of spiro[fluorene-9,9'-xanthene] via this patented one-pot method offers a reproducible and scalable protocol suitable for both laboratory discovery and industrial production. The process begins by charging a reactor with fluorenone and a ten-fold molar excess of phenol under strictly anhydrous and oxygen-free conditions to prevent oxidative degradation of the sensitive intermediates. Following the addition of methanesulfonic acid as the catalyst, the mixture is heated to approximately 140°C and maintained for 24 hours to ensure complete conversion. Upon cooling, the reaction is quenched with water, and the organic product is extracted using dichloromethane, followed by solvent removal under reduced pressure. The resulting crude solid is then subjected to recrystallization using ethyl acetate, yielding the final white powder with high purity and excellent yield, ready for downstream device fabrication.
- Charge a reactor with ketone or aldehyde substrates and a significant excess of phenol derivatives under anhydrous conditions.
- Add a strong acid catalyst such as methanesulfonic acid or trifluoromethanesulfonic acid and heat the mixture to 80-200°C for 4-72 hours.
- Quench the reaction with water, extract the organic layer, and purify the crude product via recrystallization in ethyl acetate to achieve >99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this one-pot synthesis technology presents a compelling value proposition centered on cost efficiency and supply reliability. The elimination of transition metal catalysts removes a significant cost driver associated with precious metal procurement and the subsequent analytical testing required to certify low residual metal levels in the final product. Furthermore, the simplified workflow reduces the requirement for specialized reactor configurations and extensive purification infrastructure, allowing for faster turnaround times and lower capital expenditure per kilogram of produced material. The reliance on commodity chemicals like phenol and fluorenone ensures a stable and diversified supply base, mitigating risks associated with the scarcity of exotic reagents often found in complex pharmaceutical or agrochemical syntheses.
- Cost Reduction in Manufacturing: The consolidation of multiple reaction steps into a single pot significantly lowers operational expenses by reducing labor hours, energy consumption for heating and cooling cycles, and solvent usage for intermediate washes. By avoiding the use of expensive palladium or nickel catalysts, the process inherently lowers the raw material cost profile while simultaneously eliminating the need for costly metal scavenging resins or activated carbon treatments. This streamlined approach translates directly into improved margin potential for high-volume electronic chemical manufacturing, making it an attractive option for cost-sensitive display panel production lines.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate a wide temperature range and utilize stable acid catalysts, ensures consistent batch-to-batch reproducibility essential for long-term supply contracts. The use of readily available starting materials means that supply disruptions are less likely compared to routes dependent on custom-synthesized building blocks with long lead times. Additionally, the high purity achieved through simple recrystallization reduces the risk of batch rejection due to specification failures, thereby enhancing the overall reliability of the supply chain for critical OLED host materials.
- Scalability and Environmental Compliance: The one-pot nature of the synthesis facilitates straightforward scale-up from kilogram to tonnage quantities without the need for complex process re-engineering or additional unit operations. From an environmental standpoint, the reduction in solvent waste and the absence of heavy metal effluents simplify wastewater treatment protocols and align with increasingly stringent global environmental regulations. This compliance advantage minimizes the regulatory burden on manufacturing sites and supports corporate sustainability goals by reducing the overall environmental footprint of the chemical production process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this xanthene synthesis technology in industrial settings. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation, offering clarity on reaction scope, purification strategies, and scalability potential. Understanding these details is critical for technical teams evaluating the feasibility of integrating this route into existing manufacturing portfolios for optoelectronic materials.
Q: What are the primary advantages of the one-pot acid-catalyzed method for xanthene synthesis?
A: The primary advantages include process simplification by eliminating intermediate isolation steps, the use of inexpensive and readily available raw materials like phenol and fluorenone, and the ability to achieve high purity (>99%) through simple recrystallization without complex chromatography.
Q: Can this synthesis method be adapted for halogenated derivatives required for cross-coupling?
A: Yes, the patent explicitly demonstrates the successful synthesis of brominated derivatives such as 2-bromo and 2,7-dibromo spiro[fluorene-9,9'-xanthene], which are critical precursors for further functionalization in OLED material development.
Q: How does this method address environmental and safety concerns in large-scale production?
A: By utilizing a one-pot procedure, the method reduces solvent consumption and waste generation associated with multiple workup stages. Furthermore, the absence of transition metal catalysts eliminates the need for expensive and environmentally burdensome heavy metal removal processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spiro[fluorene-9,9'-xanthene] Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity spiro-compounds in the advancement of next-generation display technologies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering materials that meet stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the absence of impurities that could compromise device performance. Our expertise in acid-catalyzed condensations allows us to optimize this specific patent route for maximum yield and cost-efficiency, providing our partners with a competitive edge in the global electronic materials market.
We invite you to collaborate with us to explore how this innovative synthesis method can enhance your supply chain resilience and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our CDMO capabilities can accelerate your product development timeline while ensuring the highest standards of quality and reliability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
