Advanced Chiral Boron-Nitrogen Ligands for Scalable Asymmetric Catalysis and Commercial Production
The landscape of asymmetric catalysis is undergoing a significant transformation driven by the need for more efficient and economically viable chiral ligands. Patent CN110746448A discloses a groundbreaking class of boron-nitrogen ligands featuring a chiral 1,2-ethylenediamine skeleton, which represents a substantial leap forward in ligand design for transition metal catalysis. Unlike traditional phosphine-based systems that often suffer from oxidation sensitivity and high costs, these novel boron-nitrogen structures offer enhanced stability and tunable steric environments. The core innovation lies in the direct formation of the B-N bond within the chiral framework, allowing for precise control over the stereochemical outcome of catalytic reactions. This technology is particularly relevant for the synthesis of high-value pharmaceutical intermediates where enantiomeric purity is critical. By leveraging this patented methodology, manufacturers can access a reliable chiral ligand supplier network that supports the production of complex molecules with superior selectivity. The implications for the fine chemical industry are profound, as this approach simplifies the supply chain for critical catalytic components.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral ligands for asymmetric catalysis has been plagued by cumbersome multi-step procedures that drive up costs and extend lead times. Conventional methods often rely on the resolution of racemic mixtures, a process that inherently wastes at least half of the starting material and requires additional purification steps to achieve high optical purity. Furthermore, many traditional ligands involve expensive noble metals or complex phosphine architectures that are sensitive to air and moisture, complicating their handling and storage in large-scale manufacturing environments. The reliance on such fragile systems often necessitates stringent inert atmosphere conditions, adding significant operational overhead to the production process. Additionally, the removal of residual metals from the final product to meet pharmaceutical standards can be a costly and technically challenging bottleneck. These factors collectively contribute to a high barrier to entry for the commercial scale-up of complex chiral intermediates, limiting the availability of cost-effective catalytic solutions for the broader market.
The Novel Approach
The methodology outlined in patent CN110746448A introduces a streamlined synthetic route that directly addresses these inefficiencies by utilizing a simple condensation reaction between 1,2-ethylenediamine derivatives and borosilicate reagents. This novel approach eliminates the need for complex resolution steps, as the chirality is effectively transferred from the readily available diamine starting materials to the final ligand structure. The reaction proceeds under relatively mild conditions using common organic solvents such as toluene, which are easily sourced and managed in standard chemical facilities. By avoiding the use of air-sensitive reagents and complex protection-deprotection sequences, this method significantly reduces the operational complexity and safety risks associated with ligand production. The resulting boron-nitrogen ligands exhibit robust stability, making them ideal candidates for long-term storage and transportation without degradation. This shift towards a more direct and robust synthesis pathway enables substantial cost savings in asymmetric catalysis manufacturing, providing a competitive edge for producers of high-purity pharmaceutical intermediates.
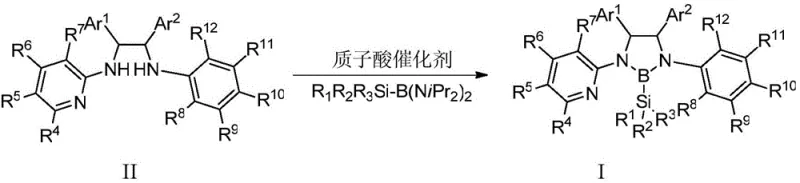
Mechanistic Insights into Boron-Nitrogen Ligand Formation and Catalytic Activity
The formation of the chiral boron-nitrogen ligand involves a precise interaction between the nitrogen atoms of the 1,2-ethylenediamine skeleton and the boron center of the silicate reagent, facilitated by a protic acid catalyst. This process creates a rigid chiral environment around the metal center when the ligand coordinates with transition metals such as iridium or rhodium. The steric bulk provided by the aryl substituents (Ar1 and Ar2) on the ligand framework plays a crucial role in discriminating between enantiotopic faces of the substrate during the catalytic cycle. This steric differentiation is essential for achieving high enantiomeric excess in asymmetric transformations, such as the C-H bond borylation of diarylmethylamines. The electronic properties of the boron-nitrogen bond also contribute to the stability of the metal-ligand complex, preventing premature decomposition under reaction conditions. Understanding these mechanistic details allows chemists to fine-tune the ligand structure by modifying the R groups, thereby optimizing the catalyst for specific substrate classes. This level of tunability is a key advantage for R&D teams seeking to develop bespoke catalytic solutions for challenging synthetic targets.
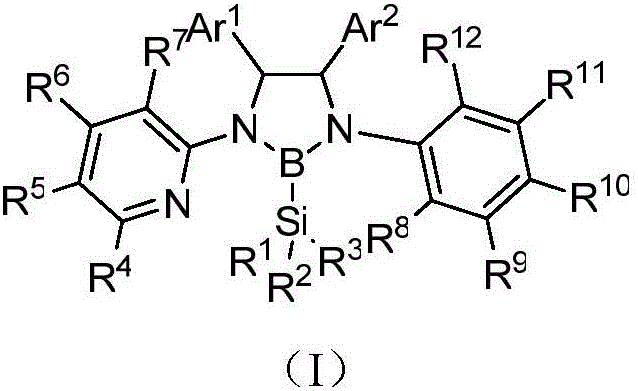
Impurity control is a critical aspect of this technology, as the presence of side products can significantly impact the performance of the catalyst in downstream applications. The patented process ensures high purity by utilizing specific molar ratios of the borosilicate reagent to the diamine precursor, typically ranging from 1.5:1 to 2:1, which drives the reaction to completion while minimizing the formation of oligomeric byproducts. The use of a protic acid catalyst, such as hydrochloric acid in dioxane, further promotes the selective formation of the desired B-N bond without inducing unwanted side reactions on the sensitive amine functionalities. Post-reaction purification via recrystallization or column chromatography effectively removes any residual starting materials or inorganic salts, yielding a ligand product that meets stringent quality specifications. This rigorous control over the synthesis parameters ensures that the final ligand is free from contaminants that could poison the catalytic cycle or compromise the purity of the final pharmaceutical intermediate. Consequently, this method supports the production of high-purity catalysts that are essential for regulatory compliance in the pharmaceutical industry.
How to Synthesize Chiral Boron-Nitrogen Ligands Efficiently
The synthesis of these advanced ligands is designed to be operationally simple, requiring standard laboratory equipment and readily available reagents. The process begins with the preparation of a uniformly mixed reaction system containing the specific 1,2-ethylenediamine derivative, the borosilicate reagent, a protic acid catalyst, and an organic solvent like toluene. This mixture is then heated to a temperature between 80°C and 180°C for a duration of 12 to 36 hours, allowing the B-N bond formation to proceed to completion. The detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Prepare a uniformly mixed reaction system containing the 1,2-ethylenediamine derivative, borosilicate reagent, protic acid catalyst, and organic solvent.
- Heat the reaction mixture to a temperature range of 80°C to 180°C and maintain for 12 to 36 hours to facilitate the formation of the B-N bond.
- Cool the reaction, remove the solvent, and purify the resulting chiral boron-nitrogen ligand via recrystallization or column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this ligand technology offers significant strategic benefits related to cost stability and supply continuity. The simplified synthesis route reduces the dependency on exotic or hard-to-source reagents, thereby mitigating the risk of supply chain disruptions caused by raw material shortages. Since the process utilizes common organic solvents and standard reaction conditions, it can be easily integrated into existing manufacturing infrastructure without the need for specialized equipment upgrades. This compatibility translates to faster implementation times and lower capital expenditure for facilities looking to adopt this technology. Furthermore, the robustness of the ligand structure ensures a longer shelf life, reducing waste associated with material degradation during storage. These factors collectively contribute to a more resilient and cost-effective supply chain for critical catalytic components.
- Cost Reduction in Manufacturing: The elimination of complex resolution steps and the use of cost-effective borosilicate reagents significantly lower the overall production cost of the ligand. By avoiding the waste associated with racemic resolution, the material efficiency of the process is greatly improved, leading to substantial cost savings in asymmetric catalysis manufacturing. Additionally, the simplified purification process reduces the consumption of solvents and energy, further enhancing the economic viability of large-scale production. This cost efficiency allows manufacturers to offer competitive pricing for high-purity pharmaceutical intermediates without compromising on quality or performance standards.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials such as 1,2-ethylenediamine derivatives and common boron reagents ensures a stable and predictable supply chain. Unlike specialized chiral pools that may be subject to market volatility, these raw materials are produced in high volumes by multiple suppliers globally. This diversity in sourcing options reduces the risk of single-supplier dependency and ensures consistent availability of the ligand for continuous manufacturing operations. The robust nature of the ligand also simplifies logistics, as it does not require stringent temperature control or inert atmosphere packaging during transportation, reducing shipping costs and complexity.
- Scalability and Environmental Compliance: The reaction conditions described in the patent are highly amenable to scale-up, utilizing standard batch reactors and common workup procedures. The use of toluene and other standard solvents allows for efficient recovery and recycling, minimizing environmental impact and waste disposal costs. Furthermore, the absence of heavy metal contaminants in the ligand synthesis itself simplifies the downstream waste treatment processes. This alignment with green chemistry principles supports corporate sustainability goals and ensures compliance with increasingly stringent environmental regulations. The ability to scale this process from gram to ton quantities without significant re-optimization makes it an ideal choice for the commercial scale-up of complex chiral ligands.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this boron-nitrogen ligand technology. These answers are derived directly from the technical specifications and experimental data provided in patent CN110746448A, ensuring accuracy and relevance for decision-makers. Understanding these details is crucial for evaluating the feasibility of integrating this catalyst system into your existing production workflows.
Q: What are the primary advantages of this boron-nitrogen ligand over conventional chiral ligands?
A: The primary advantage lies in the simplified preparation method which avoids complex resolution steps. The ligand can be prepared directly from racemic or optically pure 1,2-ethylenediamine derivatives, significantly reducing processing time and material waste compared to traditional multi-step chiral pool syntheses.
Q: How does this technology impact the cost structure of asymmetric catalytic processes?
A: By utilizing a simple one-pot reaction with readily available borosilicate reagents and common organic solvents like toluene, the manufacturing cost is substantially lowered. The elimination of expensive transition metal removal steps in downstream processing further enhances the economic viability for large-scale production.
Q: Is this ligand system suitable for industrial scale-up of complex pharmaceutical intermediates?
A: Yes, the reaction conditions described in patent CN110746448A, such as moderate temperatures and standard organic solvents, are highly compatible with existing industrial infrastructure. The robustness of the boron-nitrogen skeleton ensures stability during storage and handling, facilitating reliable supply chain continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Boron-Nitrogen Ligand Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced patent technologies like CN110746448A into commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial application is seamless. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of chiral boron-nitrogen ligand meets the highest industry standards. Our commitment to quality ensures that your catalytic processes perform consistently, delivering the high enantiomeric excess required for modern pharmaceutical synthesis.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through the adoption of this innovative ligand technology. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our solutions can enhance your operational efficiency. Let us partner with you to drive innovation and profitability in your asymmetric catalysis projects.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
