Advanced Purification Technology for High-Purity L-alpha-GPC Pharmaceutical Intermediates
Advanced Purification Technology for High-Purity L-alpha-GPC Pharmaceutical Intermediates
The pharmaceutical industry continuously demands higher purity standards for neuroprotective agents, particularly for compounds like L-alpha-glycerophosphocholine (L-α-GPC), which serves as a critical precursor for acetylcholine synthesis. Patent CN102260286A introduces a transformative methodology for separating and purifying crude L-α-GPC, addressing the longstanding challenge of removing deep-colored impurities that plague conventional synthesis routes. This technology leverages a synergistic combination of activated alkaline aluminum oxide decolorization and precise silica gel column chromatography to elevate product purity from crude levels to over 99.5%. For R&D directors and procurement specialists, this represents a pivotal shift from complex crystallization protocols to a more robust, adsorption-based purification strategy that is inherently scalable. The ability to transform dark brown crude material into a colorless, transparent pharmaceutical-grade substance without excessive energy consumption marks a significant advancement in fine chemical processing.
Furthermore, the economic implications of this patent extend beyond mere purity metrics, offering a compelling value proposition for supply chain optimization. By utilizing reagents that can be regenerated and recycled, such as the alkaline aluminum oxide and absolute ethanol, the process drastically reduces the variable costs associated with consumable materials. This aligns perfectly with the strategic goals of a reliable pharmaceutical intermediate supplier aiming to deliver cost-effective solutions without compromising on quality. The method eliminates the need for expensive ion-exchange resins that often suffer from capacity degradation, replacing them with stable inorganic adsorbents. Consequently, this innovation not only enhances the chemical profile of the final product but also fortifies the supply chain against raw material volatility, ensuring consistent availability for downstream drug manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of L-α-GPC has been hindered by the inefficiencies of traditional decolorization and separation techniques, which often fail to meet the stringent requirements of modern pharmacopeia. Prior art, such as the method described in CN 102041281A, relies heavily on activated carbon decolorization coupled with ion exchange resin chromatography. While this approach can achieve purities around 98.8%, it frequently falls short of the >99% threshold required for direct clinical application and formulation studies. The residual impurities, often deeply colored organic byproducts, are difficult to remove completely using activated carbon due to non-specific adsorption and limited surface area efficiency. Moreover, ion exchange resins introduce additional complexities, including the need for rigorous pH control, extensive washing cycles to remove leached ions, and the eventual degradation of the resin matrix which necessitates frequent replacement.
Alternative methods involving crystallization from ethanol solutions, as seen in WO/2007/145476 and EP 0486100 A1, impose even stricter constraints on the quality of the starting crude material. These crystallization-dependent processes require the crude L-α-GPC to possess a relatively high initial purity to facilitate crystal growth, making them unsuitable for processing lower-grade bulk intermediates. The crystallization step itself is energy-intensive, requiring precise temperature cycling and long settling times, which bottlenecks production throughput. Additionally, the mother liquor remaining after crystallization often contains significant amounts of the target product, leading to yield losses unless complex recovery systems are implemented. These limitations collectively result in higher operational expenditures and a less flexible manufacturing process that struggles to adapt to varying qualities of incoming crude feedstock.
The Novel Approach
The methodology disclosed in CN102260286A circumvents these bottlenecks by introducing a two-stage purification protocol centered on activated alkaline aluminum oxide and silica gel chromatography. This novel approach begins with a highly efficient decolorization step where the crude product is dissolved in absolute ethanol at 50°C and treated with activated alkaline Al2O3. Unlike activated carbon, the alkaline aluminum oxide exhibits superior selectivity for polar colored impurities while minimizing the adsorption of the target zwitterionic molecule. The process operates at a mass ratio of 1:(5-10) between the crude product and the adsorbent, ensuring sufficient surface area for impurity capture without excessive loss of the active ingredient. Following centrifugation and concentration, the resulting viscous oil undergoes silica gel column chromatography using a specifically optimized ternary solvent system.
This second stage utilizes a mobile phase composed of methanol, ethanol, and chloroform in a precise volume ratio of 50:35:15, which creates an ideal polarity gradient for separating L-α-GPC from structurally similar phospholipid byproducts. The use of silica gel, with a mass loading ratio of 1:(40-50), provides high resolution separation capable of resolving trace impurities that previous methods left behind. The result is a colorless, transparent product with purity exceeding 99.5%, directly suitable for pharmaceutical formulation without further refinement. This approach not only simplifies the workflow by removing the need for crystallization but also enhances robustness, allowing for the processing of darker, lower-quality crude inputs that would otherwise be rejected by conventional methods. The integration of these two distinct physicochemical mechanisms creates a synergistic effect that maximizes both yield and purity.
Mechanistic Insights into Adsorption Decolorization and Chromatographic Separation
The core of this purification technology lies in the specific surface chemistry interactions between the impurities and the activated alkaline aluminum oxide. Aluminum oxide (Al2O3) possesses a amphoteric surface with Lewis acid and base sites; in its activated alkaline form, it presents a high density of basic hydroxyl groups that interact strongly with acidic or polar chromophores present in the crude synthesis mixture. When the crude L-α-GPC solution is stirred with the adsorbent at 50°C, the thermal energy increases the diffusion rate of impurities to the adsorbent surface, facilitating rapid binding. The mechanism likely involves hydrogen bonding and dipole-dipole interactions between the surface of the Al2O3 and the conjugated systems of the colored degradation products. This selective adsorption leaves the L-α-GPC, which is highly soluble in the ethanol medium and less prone to irreversible binding under these specific conditions, in the supernatant. The subsequent centrifugation effectively partitions the solid-bound impurities from the liquid phase, achieving a preliminary purity boost to over 85%.
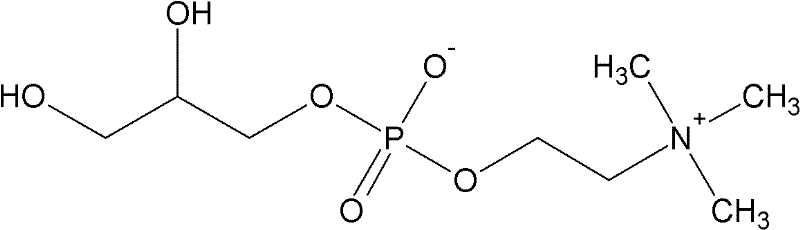
Following decolorization, the silica gel chromatography step exploits the differences in polarity and hydrogen-bonding capability between L-α-GPC and residual contaminants. Silica gel, composed of silicon dioxide with surface silanol groups, acts as a normal-phase stationary phase. The mobile phase, a carefully balanced mixture of methanol, ethanol, and chloroform, modulates the elution strength to ensure the target molecule migrates at a distinct rate. Methanol and ethanol act as polar modifiers that compete with the analyte for silanol binding sites, preventing tailing and ensuring sharp peak resolution, while chloroform provides the necessary organic character to maintain solubility of the phospholipid structure. The specific ratio of 50:35:15 is critical; deviations could lead to co-elution of impurities or excessive retention times. This precise control over the chromatographic environment ensures that the final eluate contains exclusively the target species, resulting in the observed single peak in HPLC analysis and the characteristic colorless appearance of the high-purity product.
How to Synthesize L-alpha-GPC Efficiently
Implementing this purification protocol requires strict adherence to the activation parameters of the adsorbents and the solvent ratios defined in the patent to ensure reproducibility at scale. The process begins with the thermal activation of the alkaline aluminum oxide at 250°C for 4 hours and silica gel at 105°C for 30 minutes to remove adsorbed water and maximize surface activity. Once prepared, the crude material is subjected to the decolorization and chromatography sequence as detailed in the technical specifications. This standardized approach minimizes batch-to-batch variability and ensures that the final product consistently meets the >99.5% purity benchmark required for regulatory compliance. For detailed operational parameters and safety guidelines, please refer to the full technical documentation.
- Dissolve crude L-alpha-GPC in absolute ethanol at 50°C and treat with activated alkaline Al2O3 (1: 5 to 1:10 ratio) for 2 hours to remove colored impurities.
- Centrifuge to remove the solid Al2O3 phase, then concentrate the supernatant to a viscous oil.
- Perform silica gel column chromatography using a methanol: ethanol:chloroform (50:35:15) eluent system to isolate pure L-alpha-GPC.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this purification technology offers substantial opportunities for cost optimization and supply chain stabilization. The primary driver of cost reduction is the regenerability of the key processing materials. Unlike ion-exchange resins which have a finite lifecycle and require costly disposal, the alkaline aluminum oxide used in this process can be restored to its original white powder state through a simple calcination process at 400°C after washing. This closed-loop usage of the adsorbent dramatically lowers the recurring cost of goods sold (COGS) associated with purification media. Furthermore, the solvent system relies on common industrial chemicals like ethanol, methanol, and chloroform, which are widely available and can be recovered via distillation. The ability to recycle absolute ethanol through condensation means that solvent consumption is minimized, reducing both purchase costs and waste treatment expenses.
Supply chain reliability is significantly enhanced by the robustness of the method against feedstock variability. Traditional crystallization methods often fail when the purity of the incoming crude drops below a certain threshold, leading to production stoppages or the need for expensive rework. In contrast, the adsorption-based decolorization step in this patent is highly effective even on dark brown, low-purity crude materials. This flexibility allows manufacturers to source crude L-α-GPC from a broader range of suppliers, including those offering lower-cost bulk grades, without risking the quality of the final API. This diversification of the supply base mitigates the risk of shortages and price spikes from single-source vendors. Additionally, the elimination of complex crystallization steps shortens the overall production cycle time, allowing for faster turnaround on orders and improved responsiveness to market demand fluctuations.
- Cost Reduction in Manufacturing: The elimination of expensive ion-exchange resins and the implementation of a regenerable aluminum oxide system fundamentally alters the cost structure of purification. By extending the lifespan of the adsorbent through thermal regeneration, the facility avoids the continuous capital outlay for fresh resin beds. Additionally, the recovery of solvents reduces the volume of hazardous waste requiring disposal, leading to lower environmental compliance costs. The process operates at moderate temperatures (50°C for dissolution, ambient for chromatography), avoiding the high energy demands of reflux or deep-freeze crystallization, which translates to lower utility bills per kilogram of product produced.
- Enhanced Supply Chain Reliability: The tolerance of this method for lower-quality crude inputs provides a strategic buffer against upstream supply disruptions. Procurement teams are not locked into purchasing premium-grade crude, which is often scarce and expensive. Instead, they can secure cost-effective bulk intermediates and rely on the robust purification process to upgrade the quality internally. This decoupling of crude quality from final product quality ensures a steady flow of finished goods even when the raw material market is volatile. The use of standard, commodity-grade solvents further ensures that production is never halted due to the unavailability of specialized reagents.
- Scalability and Environmental Compliance: The transition from batch crystallization to column chromatography, while traditionally seen as a scaling challenge, is managed here through the use of standard adsorbents that pack predictably. The process generates significantly less aqueous waste compared to ion-exchange methods, which require extensive acid/base regeneration cycles. The primary waste stream is the spent aluminum oxide, which is non-toxic and can be regenerated, or disposed of as inert solid waste. This reduced environmental footprint simplifies permitting and aligns with the increasing regulatory pressure on pharmaceutical manufacturers to adopt greener chemistry practices, enhancing the corporate sustainability profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this purification technology. They are derived directly from the experimental data and beneficial effects reported in patent CN102260286A, providing clarity on purity outcomes, material regeneration, and process parameters. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this method into existing production lines.
Q: What purity level can be achieved with this Al2O3 decolorization method?
A: According to patent CN102260286A, the combined process of activated alkaline aluminum oxide decolorization followed by silica gel chromatography achieves a final product purity exceeding 99.5%, which is suitable for direct pharmaceutical formulation.
Q: Can the adsorbents and solvents be recycled to reduce costs?
A: Yes, the patent explicitly states that the alkaline aluminum oxide can be regenerated by calcination at 400°C after washing, and the absolute ethanol solvent can be recovered via condensation, significantly lowering material costs.
Q: What is the optimal eluent ratio for the silica gel chromatography step?
A: The optimal eluent system identified is a mixed solvent of methanol, ethanol, and chloroform in a volume ratio of 50:35:15, which ensures effective separation of the target compound from residual impurities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-alpha-GPC Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the development of effective nootropic and neuroprotective therapies. Our technical team has extensively analyzed advanced purification pathways, including the Al2O3-silica gel methodology, to ensure we can deliver L-alpha-GPC that meets the most rigorous international standards. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order volume. Our facilities are equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every batch of L-alpha-GPC supplied is free from the colored impurities and residual solvents that compromise efficacy.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains for this vital pharmaceutical intermediate. By leveraging our technical expertise, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us partner with you to secure a stable, high-quality supply of L-alpha-GPC that drives your drug development projects forward with confidence and efficiency.
