Advanced Stiripentol Manufacturing: Transforming Synthetic By-Products into High-Value Epilepsy Intermediates
The pharmaceutical landscape for neurotherapeutics is constantly evolving, with a specific focus on optimizing the manufacturing of antiepileptic drugs like Stiripentol. Patent CN102690252B introduces a groundbreaking methodology that redefines the economic and chemical efficiency of producing this critical medication. Unlike traditional approaches that rely on expensive, dedicated starting materials, this invention leverages a "waste-to-wealth" strategy by utilizing 4-(2-methylallyl)-1,2-phenyldiphenol, a compound typically discarded as a by-product in benzofuranol synthesis. This strategic pivot not only addresses environmental concerns regarding chemical waste but also fundamentally alters the cost structure of the supply chain. The patent outlines a robust five-step sequence involving etherification, isomerization, oxidation, condensation, and reduction, culminating in the high-purity formation of Stiripentol. For R&D directors and procurement specialists, this represents a significant opportunity to secure a more sustainable and cost-effective source of this vital epilepsy treatment intermediate.

Historically, the synthesis of Stiripentol has been approached through several distinct pathways, each carrying its own set of logistical and chemical burdens. Early methodologies, such as those described by Lepage et al., utilized bromo-piperonyl cyclonene as a precursor. This route, while chemically valid, involves the handling of halogenated intermediates which can complicate purification and increase the environmental footprint due to halogen waste streams. Furthermore, the reliance on specific brominated starting materials often ties the supply chain to volatile halogen markets and introduces potential heavy metal contamination risks during subsequent coupling steps.
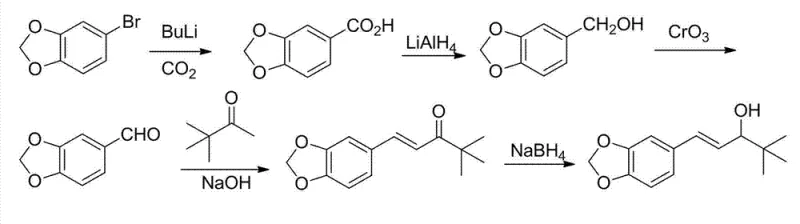
Alternative conventional methods, such as those reported by the Vallet group, commence directly from piperonylaldehyde. While this appears simpler on paper, it presents significant challenges in terms of raw material stability and quality control. Piperonylaldehyde is prone to oxidation and polymerization upon storage, necessitating rigorous quality assurance protocols that can delay production timelines. Additionally, direct condensation of commercially sourced piperonylaldehyde can lead to inconsistent batch-to-batch reproducibility due to trace impurities inherent in bulk chemical supplies. The novel approach detailed in CN102690252B circumvents these issues by generating the aldehyde functionality in situ, ensuring freshness and reactivity while bypassing the stability issues associated with storing sensitive aldehyde intermediates.
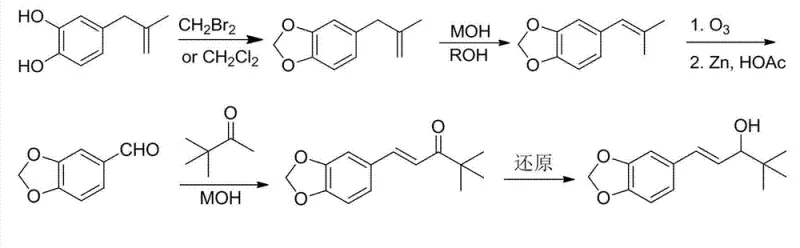
The mechanistic elegance of the new process lies in its sequential transformation of the phenolic by-product into the target pharmacophore. The initial etherification step utilizes methylene bromide to protect the catechol moiety, forming the methylenedioxy ring essential for the drug's activity. This is followed by a base-catalyzed isomerization, likely proceeding through an allylic transposition mechanism, which shifts the terminal double bond into conjugation with the aromatic system. This conjugation is critical for the subsequent oxidative cleavage. The use of ozone for oxidation is particularly noteworthy; it offers a highly selective method for cleaving the alkene to generate the requisite aldehyde without over-oxidizing the sensitive aromatic ring or the newly formed acetal. This precision minimizes the formation of carboxylic acid by-products, thereby simplifying downstream purification.
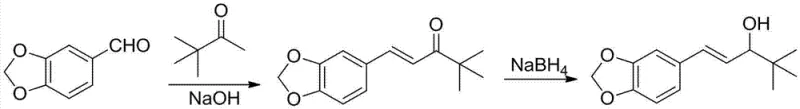
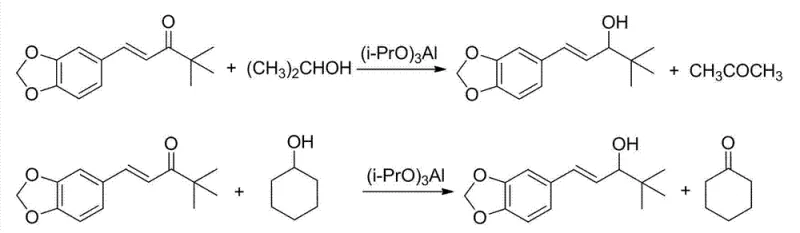
The final stages of the synthesis involve an aldol condensation with pinacolone followed by a stereoselective reduction. A key innovation here is the controlled addition of the aldehyde to the basic pinacolone solution. By dripping the aldehyde into the base rather than mixing all components at once, the process mitigates the risk of Cannizzaro disproportionation reactions, which would otherwise consume the valuable aldehyde and reduce overall yield. The subsequent reduction step offers a choice between sodium borohydride and aluminum isopropoxide. The patent highlights the superiority of the aluminum isopropoxide-mediated reduction, a variation of the Meerwein-Ponndorf-Verley (MPV) reduction. This method is exceptionally gentle, selectively reducing the ketone to the alcohol without affecting the conjugated double bond, preserving the geometric integrity of the molecule.
The commercial implications of adopting this synthesis route are profound for procurement and supply chain management teams. The primary driver for cost reduction is the utilization of 4-(2-methylallyl)-1,2-phenyldiphenol as a feedstock. Since this material is characterized as a by-product of another industrial synthesis, its acquisition cost is theoretically negligible compared to purchasing dedicated fine chemical building blocks. This "upcycling" of waste material translates directly into a substantially reduced Cost of Goods Sold (COGS). Furthermore, by integrating the synthesis of the piperonylaldehyde intermediate directly into the main workflow, manufacturers eliminate the markup and logistics costs associated with sourcing this unstable aldehyde from third-party vendors. The process effectively consolidates two supply chains into one, streamlining procurement operations.
- Cost Reduction in Manufacturing: The elimination of expensive halogenated starting materials and the substitution of high-cost dedicated precursors with low-value by-products creates a structural advantage in manufacturing economics. The avoidance of disproportionation side reactions during the condensation step further enhances material efficiency, ensuring that a higher percentage of input mass is converted into saleable product. Additionally, the use of aluminum isopropoxide for the final reduction step reportedly achieves yields significantly higher than standard borohydride reductions, minimizing the loss of high-value intermediates in the final, most expensive stage of the synthesis.
- Enhanced Supply Chain Reliability: Dependence on single-source suppliers for specialized intermediates like bromo-piperonyl derivatives poses a significant risk to production continuity. By shifting to a route that begins with a phenolic by-product, the supply chain becomes more resilient and less susceptible to market fluctuations in niche chemical sectors. The in situ generation of piperonylaldehyde removes the dependency on external aldehyde suppliers, who may face their own production bottlenecks or quality inconsistencies. This vertical integration of the early synthetic steps ensures a consistent flow of reactive intermediates, stabilizing the overall production schedule and reducing the risk of stockouts.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard unit operations such as etherification, distillation, and crystallization that are easily transferable from pilot plant to commercial scale. The replacement of harsh oxidants with ozone, followed by a mild zinc workup, aligns with modern green chemistry principles by reducing the generation of toxic heavy metal waste. The high selectivity of the MPV reduction reduces the burden on purification systems, lowering solvent consumption and energy usage during chromatography or recrystallization. These factors collectively simplify the regulatory approval process for the manufacturing site and ensure long-term environmental compliance.
- Etherify 4-(2-methylallyl)-1,2-phenyldiphenol using methylene bromide to form the methylenedioxy ring.
- Perform base-catalyzed isomerization to shift the double bond into conjugation with the aromatic ring.
- Execute oxidative cleavage via ozonolysis followed by zinc reduction to generate piperonylaldehyde.
- Conduct aldol condensation with pinacolone under controlled dripping conditions to prevent disproportionation.
- Reduce the resulting enone using aluminum isopropoxide to achieve high-yield Stiripentol.
Frequently Asked Questions (FAQ)
Q: What is the primary raw material advantage in patent CN102690252B?
A: The process utilizes 4-(2-methylallyl)-1,2-phenyldiphenol, which is identified as a by-product from benzofuranol synthesis, effectively turning waste material into a valuable pharmaceutical intermediate.
Q: Why is aluminum isopropoxide preferred for the final reduction step?
A: Using aluminum isopropoxide in a Meerwein-Ponndorf-Verley (MPV) reduction avoids side reactions common with polyfunctional compounds and reportedly achieves yields up to 95%, compared to lower yields with sodium borohydride.
Q: How does this method improve upon conventional piperonal-based routes?
A: By generating piperonylaldehyde in situ from the isomerized intermediate via ozonolysis, the process integrates the aldehyde synthesis directly into the Stiripentol workflow, reducing the need for external sourcing of unstable aldehydes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Stiripentol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthesis route described in CN102690252B for the global epilepsy treatment market. As a premier CDMO partner, we possess the technical expertise to translate this patented methodology into a robust, GMP-compliant manufacturing process. Our facilities are equipped to handle the specific requirements of this pathway, from the safe handling of ozone in oxidation steps to the precise temperature control needed for the MPV reduction. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize advanced analytical techniques to verify the identity and purity of every batch of Stiripentol intermediate produced.
We invite pharmaceutical partners to collaborate with us to leverage this innovative technology for your next project. By optimizing this route, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this by-product valorization strategy can impact your bottom line. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a sustainable, high-quality supply of Stiripentol that meets the rigorous demands of the modern neurotherapeutic industry.
