Scalable Synthesis of 16-Beta-Methyl Steroid Intermediates for Corticosteroid Production
The pharmaceutical industry continuously seeks robust and scalable pathways for the production of potent corticosteroids, particularly those featuring the critical 16β-methyl substitution pattern found in drugs like Betamethasone. Patent CN1252081C discloses a sophisticated series of intermediates and a corresponding synthetic methodology designed to construct this complex steroidal architecture with high precision. This technology addresses the longstanding challenges associated with introducing the 16-methyl group while maintaining the integrity of the sensitive 17-alpha-hydroxy and 20-keto functionalities. By leveraging a strategic sequence of dehydration, organometallic methylation, and selective epoxidation, the disclosed route offers a viable alternative to traditional methods that often suffer from poor stereocontrol or harsh reaction conditions. For R&D teams focused on next-generation anti-inflammatory agents, understanding the nuances of this pathway is essential for optimizing yield and purity profiles in early-stage development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
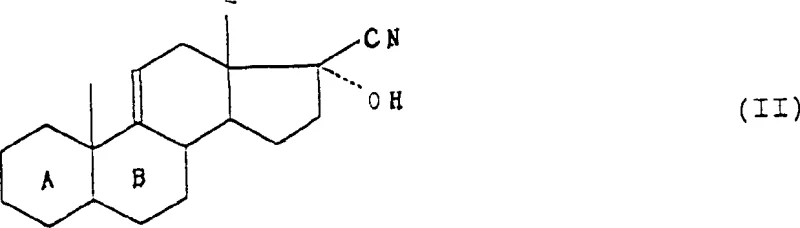
Historically, the synthesis of 16-methylated steroids has been plagued by issues related to regioselectivity and the stability of intermediates under acidic or basic conditions. Conventional routes often require extensive protection and deprotection strategies that add significant step counts and reduce overall atom economy. Traditional methods might rely on microbiological fermentation or complex rearrangement reactions that are difficult to control on a large industrial scale, leading to batch-to-batch variability. Furthermore, the introduction of the methyl group at the C16 position without affecting the neighboring C17 hydroxyl group typically demands highly specific reagents that are either cost-prohibitive or hazardous to handle in bulk quantities. These inefficiencies create bottlenecks in the supply chain, driving up the cost of goods sold (COGS) for the final active pharmaceutical ingredient and limiting the ability of manufacturers to respond quickly to market demand fluctuations.
The Novel Approach
The methodology outlined in the patent data presents a streamlined solution by utilizing a dehydration-first strategy that activates the D-ring for subsequent functionalization. Instead of struggling with direct alkylation on a saturated ring, the process converts the 17-hydroxy precursor into a reactive triene system, specifically the 5,9(11),16(17)-triene derivative. This activation allows for the facile introduction of the C20 methyl ketone via Grignard addition, setting the stage for a highly stereoselective epoxidation. The novelty lies in the sequential protection of the C20 ketone as an acetal prior to the final methylation step, which effectively shields the carbonyl from unwanted side reactions while directing the incoming methyl nucleophile to the C16 position from the beta-face. This logical progression minimizes the need for chromatographic purifications between steps, thereby enhancing the throughput and economic viability of the entire manufacturing campaign.
Mechanistic Insights into Dehydration and Epoxidation Strategy
The core of this synthetic route relies on the precise manipulation of the steroid D-ring electronics. The initial dehydration using phosphoryl chloride in pyridine proceeds via an E2-like elimination mechanism, removing the 17-alpha-hydroxyl group to generate the delta-16 double bond. This unsaturation is crucial as it extends the conjugation of the steroid nucleus, facilitating the subsequent nucleophilic attack by methylmagnesium halides. The formation of the methyl ketone at C20 is followed by an alkaline epoxidation using hydrogen peroxide, which selectively targets the electron-rich delta-16 double bond. The presence of the adjacent carbonyl group influences the electrophilicity of the alkene, ensuring that the peracid or hydroperoxide species attacks specifically at the 16,17-position to form the alpha-epoxide. This epoxide serves as a masked electrophile, primed for ring-opening by a second equivalent of methylating agent.
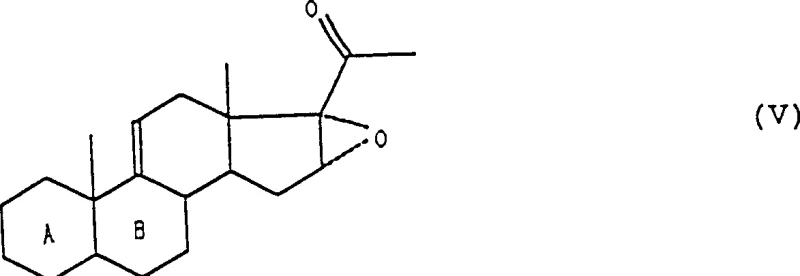
Controlling impurities during these transformations is paramount for meeting regulatory standards for pharmaceutical intermediates. The use of ethylene glycol to protect the C20 ketone as a ketal prior to the final Grignard reaction is a masterstroke in impurity control. Without this protection, the highly reactive Grignard reagent could attack the C20 carbonyl, leading to tertiary alcohol byproducts that are structurally similar to the desired product and difficult to separate. By masking the ketone, the process ensures that the nucleophilic attack occurs exclusively at the less hindered C16 position of the epoxide ring. Furthermore, the hydrolysis conditions used in the final deprotection step are mild enough to prevent the migration of the double bonds or the degradation of the sensitive 17-alpha-hydroxy group, resulting in a crude product profile that is significantly cleaner than those obtained via non-protected routes.
How to Synthesize 16-Beta-Methyl Steroid Intermediates Efficiently
The synthesis of these high-value intermediates requires careful attention to reaction parameters such as temperature control and stoichiometry to ensure optimal conversion. The process begins with the activation of the starting material, followed by a series of functional group interconversions that build complexity efficiently. Operators must maintain inert atmospheres during organometallic steps to prevent quenching of the reagents, and precise pH control is necessary during the epoxidation and hydrolysis phases to avoid ring opening at the wrong position. While the general workflow is robust, the specific timing of reagent addition and the choice of solvent systems (such as THF versus toluene mixtures) can influence the crystal habit and filtration properties of the intermediates. For a detailed breakdown of the standardized operating procedures and safety protocols required to execute this chemistry safely, please refer to the technical guide below.
- Dehydrate the starting 17-alpha-hydroxy-17-beta-cyano-androstane derivative using phosphoryl chloride in pyridine to form the triene intermediate.
- React the triene with an organo-metallic methylating reagent such as methylmagnesium chloride to introduce the C20 methyl ketone functionality.
- Perform alkaline epoxidation followed by C20 ketone protection and final Grignard methylation to establish the 16-beta-methyl stereochemistry.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers substantial advantages due to its reliance on commodity chemicals rather than specialized proprietary catalysts. The primary reagents, including phosphoryl chloride, hydrogen peroxide, and methylmagnesium bromide, are available globally in bulk quantities, reducing the risk of supply chain disruptions caused by single-source dependencies. The elimination of expensive transition metal catalysts not only lowers the raw material costs but also simplifies the downstream processing, as there is no need for costly heavy metal scavenging steps to meet residual metal specifications. This translates directly into a more favorable cost structure for the final API, allowing pharmaceutical companies to maintain competitive pricing in generic markets while preserving healthy margins. Additionally, the use of standard solvents like methylene chloride and ethyl acetate facilitates solvent recovery and recycling, further contributing to operational cost reductions.
- Cost Reduction in Manufacturing: The process design inherently reduces manufacturing costs by minimizing the number of isolation steps and avoiding the use of precious metal catalysts. By employing a protection strategy that prevents side reactions, the yield of the desired isomer is maximized, reducing the volume of waste material that must be disposed of. The ability to crystallize intermediates directly from the reaction mixture without extensive chromatography significantly lowers labor and consumable costs associated with purification. This efficiency is critical for high-volume production where even small improvements in yield can result in substantial financial savings over the lifecycle of the product.
- Enhanced Supply Chain Reliability: Utilizing widely available reagents ensures that production schedules are not held hostage by the lead times of exotic chemicals. The robustness of the chemistry means that the process is less sensitive to minor variations in raw material quality, providing a buffer against supply chain volatility. Manufacturers can source materials from multiple qualified vendors, creating a resilient supply network that can adapt to geopolitical or logistical challenges. This reliability is essential for maintaining continuous production lines and meeting the strict delivery commitments required by downstream pharmaceutical partners.
- Scalability and Environmental Compliance: The reaction conditions described are amenable to large-scale batch processing, with thermal profiles that can be managed using standard industrial reactor equipment. The avoidance of highly toxic reagents and the potential for solvent recycling align with modern green chemistry principles, simplifying the environmental permitting process. Waste streams generated are primarily aqueous and organic solvents that can be treated using conventional wastewater treatment facilities, reducing the environmental footprint of the manufacturing site. This scalability ensures that the technology can grow with market demand, from clinical trial batches to commercial tonnage production.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic pathway. Understanding these details is crucial for process chemists and engineers evaluating the feasibility of adopting this technology for their own production lines. The answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making.
Q: What is the key advantage of the dehydration step in this patent?
A: The use of phosphoryl chloride in pyridine allows for efficient dehydration of the 17-alpha-hydroxy group without compromising the sensitive cyano or ketone functionalities, streamlining the formation of the conjugated triene system essential for subsequent functionalization.
Q: How is the 16-beta stereochemistry controlled during synthesis?
A: The stereochemistry is established through a sequence involving epoxidation of the 16,17-double bond followed by nucleophilic attack with a methylating agent on the protected epoxide intermediate, ensuring the methyl group adopts the desired beta-orientation.
Q: Can this process be scaled for industrial API production?
A: Yes, the process utilizes standard industrial reagents like hydrogen peroxide, ethylene glycol, and Grignard reagents in common solvents such as THF and toluene, making it highly suitable for commercial scale-up without requiring exotic catalysts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 16-Beta-Methyl Steroid Intermediates Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful launch of life-saving corticosteroid therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab to plant is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch meets the highest industry standards. Our commitment to quality assurance means that you can rely on us to deliver consistent material that supports your regulatory filings and clinical timelines without compromise.
We invite you to discuss how our expertise in steroid chemistry can optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities align with your strategic goals. Let us be your partner in bringing advanced pharmaceutical solutions to the market faster and more economically.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
