Advanced One-Step Synthesis of 5-Substituted-2-Hydroxybenzoates for Fungicide Production
The pharmaceutical and agrochemical industries are constantly seeking more efficient pathways to synthesize critical intermediates, particularly those serving as precursors for high-value active ingredients. Patent CN1160304C introduces a groundbreaking one-step process for the preparation of 5-and/or 6-substituted-2-hydroxybenzoates, compounds that serve as essential building blocks for natural product synthesis and, more critically, for the manufacture of benzophenone fungicides. This technology represents a significant leap forward in process chemistry, addressing long-standing inefficiencies in the production of these vital aromatic esters. By shifting away from hazardous multi-step protocols, this innovation offers a robust platform for the commercial scale-up of complex agrochemical intermediates, ensuring a more sustainable and cost-effective supply chain for global manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-hydroxybenzoate derivatives has been plagued by cumbersome operational procedures that pose significant challenges for industrial adoption. Traditional methods, such as those cited in Synthesis (1980) and Tetrahedron (1991), typically require a multi-step sequence involving the isolation of unstable intermediates, which drastically increases processing time and solvent consumption. Furthermore, these legacy processes often rely on the use of highly corrosive hydrogen chloride (HCl) gas and toxic oxidizing agents like bromine (Br2) or copper(II) chloride (CuCl2). The necessity for separate oxidation steps not only complicates the reactor setup but also generates substantial amounts of hazardous waste, creating a heavy environmental burden and escalating disposal costs. For a reliable agrochemical intermediate supplier, managing these risks while maintaining competitive pricing is increasingly difficult under modern regulatory frameworks.
The Novel Approach
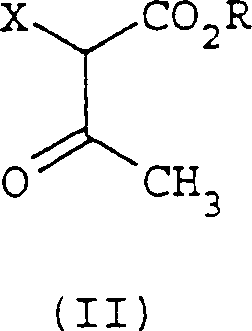
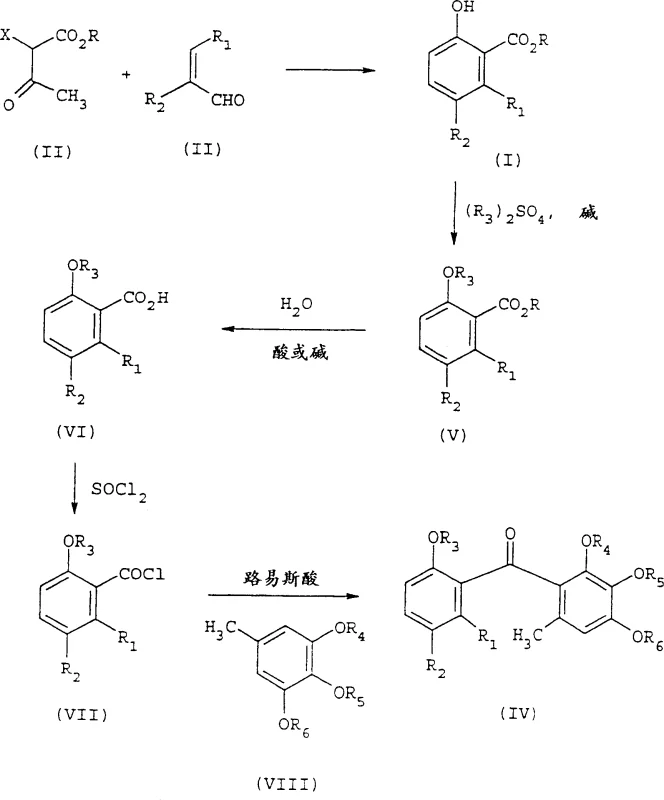
In stark contrast, the methodology disclosed in CN1160304C streamlines the entire synthesis into a single, efficient reaction vessel operation. This novel approach eliminates the need for corrosive gases and harsh oxidants by utilizing a mild cyclization strategy driven by C1-C4 carboxylate salts. The process involves reacting a beta-ketoester of formula II directly with an alpha,beta-unsaturated aldehyde of formula III in the presence of a simple carboxylate base, such as sodium acetate. This transformation occurs smoothly in common protic solvents like acetic acid or ethanol, avoiding the formation of excessive solvent waste associated with intermediate isolations. The result is a cleaner reaction profile that facilitates easier downstream processing, directly contributing to cost reduction in fungicide manufacturing by simplifying unit operations and improving overall atom economy.
Mechanistic Insights into Carboxylate-Promoted Cyclization
The core of this technological advancement lies in the elegant mechanistic pathway that allows for the direct construction of the aromatic ring system without external oxidation. The reaction initiates with the condensation of the beta-ketoester and the alpha,beta-unsaturated aldehyde, facilitated by the basic nature of the carboxylate salt. Unlike traditional routes that require discrete oxidation steps to aromatize the ring, this process likely proceeds through a cascade of aldol-type condensations and intramolecular cyclizations that spontaneously lead to the aromatic 2-hydroxybenzoate structure. The use of alkali metal acetates, such as sodium acetate or potassium acetate, serves a dual purpose: acting as a mild base to deprotonate the active methylene group and buffering the reaction medium to prevent the degradation of sensitive functional groups. This controlled environment is crucial for maintaining high selectivity and minimizing the formation of polymeric by-products often seen in aggressive acidic or oxidative conditions.
From an impurity control perspective, this mechanism offers distinct advantages for producing high-purity OLED material precursors or pharmaceutical intermediates where trace metal contamination is unacceptable. By avoiding transition metal oxidants like copper salts, the final product is inherently free from heavy metal residues that would otherwise require expensive scavenging treatments. The reaction temperature can be finely tuned between 25°C and 125°C, with optimal results observed between 75°C and 120°C, allowing operators to balance reaction rate against thermal decomposition. This thermal flexibility ensures that the process remains robust even when scaling up, as the exotherm can be managed effectively without risking the safety incidents associated with handling gaseous HCl or liquid bromine.
How to Synthesize 5-Substituted-2-Hydroxybenzoates Efficiently
Implementing this synthesis requires careful attention to stoichiometry and solvent selection to maximize yield and purity. The patent outlines a straightforward protocol where the beta-ketoester and unsaturated aldehyde are mixed with approximately 1.0 to 2.0 molar equivalents of a carboxylate salt in a polar protic solvent. The reaction mixture is then heated to reflux, typically for about 16 hours, to ensure complete conversion. Workup involves standard techniques such as partitioning between ethyl acetate and water, followed by washing with bicarbonate solution to remove acidic impurities. This simplicity makes the route highly attractive for contract development and manufacturing organizations looking to optimize their production lines. The detailed standardized synthetic steps for this specific transformation are outlined in the guide below.
- Mix a beta-ketoester of formula II with an alpha,beta-unsaturated aldehyde of formula III in a suitable solvent such as acetic acid or ethanol.
- Add a C1-C4 carboxylate salt, preferably sodium acetate or potassium acetate, acting as both base and catalyst.
- Heat the reaction mixture to reflux temperatures between 75°C and 120°C, then isolate the product via filtration or extraction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift to this one-step methodology translates into tangible operational benefits that extend beyond mere chemical efficiency. The elimination of hazardous reagents like bromine and HCl gas significantly reduces the regulatory compliance burden and lowers the cost of safety infrastructure required for storage and handling. Moreover, the use of commodity chemicals such as crotonaldehyde and ethyl chloroacetoacetate ensures a stable supply of raw materials, mitigating the risk of shortages that can plague specialty reagent markets. This stability is critical for maintaining continuous production schedules and meeting the demanding delivery timelines of global agrochemical clients.
- Cost Reduction in Manufacturing: The consolidation of multiple reaction steps into a single pot operation drastically reduces labor costs, energy consumption, and solvent usage. By removing the need for intermediate isolation and purification, manufacturers can achieve substantial savings in both time and resources. Furthermore, the avoidance of expensive oxidizing agents and the subsequent waste treatment associated with heavy metals leads to a leaner cost structure, allowing for more competitive pricing in the global market without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials and common solvents like acetic acid enhances the resilience of the supply chain. Unlike processes dependent on scarce or geopolitically sensitive reagents, this method utilizes bulk chemicals that are widely produced and easily sourced from multiple vendors. This diversification of supply sources reduces the risk of bottlenecks and ensures that production can continue uninterrupted even during periods of market volatility, providing a secure foundation for long-term supply agreements.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of toxic by-products make this process exceptionally scalable from pilot plant to full commercial production. The reduced environmental footprint aligns with increasingly stringent global regulations regarding industrial emissions and waste disposal. Facilities adopting this technology can demonstrate a commitment to green chemistry principles, potentially qualifying for environmental incentives and improving their corporate sustainability profiles, which is a key differentiator when partnering with major multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific application needs. Understanding these details is essential for R&D teams planning process validation and for procurement teams assessing vendor capabilities.
Q: What are the primary advantages of this one-step synthesis over conventional methods?
A: Unlike conventional multi-step methods requiring corrosive HCl gas and toxic oxidants like bromine, this process utilizes mild carboxylate salts in a single pot, significantly reducing waste and safety hazards.
Q: Can this process be scaled for industrial production of fungicide intermediates?
A: Yes, the process operates at moderate temperatures (25°C-125°C) using common solvents like acetic acid, making it highly suitable for large-scale commercial manufacturing without specialized high-pressure equipment.
Q: What is the purity profile of the resulting hydroxybenzoates?
A: The method yields products with high purity (e.g., >70% crude purity in examples) that can be easily purified via standard extraction or filtration, minimizing the need for complex chromatographic separation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Substituted-2-Hydroxybenzoates Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in the broader context of agrochemical and pharmaceutical development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 5-substituted-2-hydroxybenzoates meets the exacting standards required for downstream fungicide synthesis. Our commitment to quality and consistency makes us a trusted partner for companies seeking to secure their supply chains against market fluctuations.
We invite you to explore how this advanced synthesis route can optimize your production costs and enhance your product portfolio. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your decision-making process, ensuring that your project moves forward with confidence and clarity.
