Scalable Catalytic Preparation of Aromatic Azoxy Compounds for Advanced Material Manufacturing
Scalable Catalytic Preparation of Aromatic Azoxy Compounds for Advanced Material Manufacturing
The chemical industry is constantly seeking more efficient and environmentally benign pathways for synthesizing high-value intermediates, particularly aromatic azoxy compounds which serve as critical precursors for dyes, indicators, and liquid crystal materials. Patent CN113019449A introduces a groundbreaking catalytic preparation method that leverages the unique redox properties of polyoxometallates to achieve high-yield oxidative coupling of aromatic amines. This technology represents a significant departure from conventional stoichiometric methods, offering a streamlined process that operates under mild thermal conditions while maintaining exceptional atom economy. For R&D directors and procurement specialists, this patent outlines a robust framework for producing complex azoxy structures with minimal environmental footprint, addressing the growing demand for sustainable fine chemical manufacturing processes that do not compromise on purity or scalability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aromatic azoxy compounds has relied heavily on reduction coupling strategies involving nitro or nitroso compounds, often utilizing harsh reducing agents that pose significant safety and environmental challenges. For instance, prior art such as Chinese patent CN 201010136406.4 describes methods using potassium borohydride (KBH4) in alkaline media, which, while effective, incurs high raw material costs and generates substantial inorganic waste streams that require complex treatment protocols. These traditional routes often suffer from poor atom economy due to the stoichiometric consumption of expensive reductants and the generation of salt byproducts that complicate downstream purification. Furthermore, the handling of strong reducing agents necessitates specialized equipment and rigorous safety measures, increasing the overall capital expenditure and operational complexity for manufacturers aiming to scale these processes to multi-ton levels.
The Novel Approach
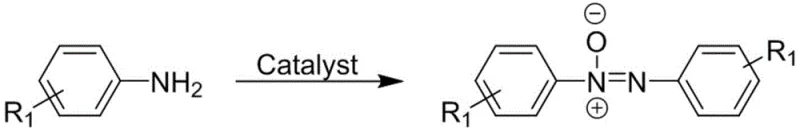
In stark contrast, the methodology disclosed in CN113019449A utilizes a catalytic oxidative coupling strategy driven by polyoxometallates, which act as versatile electron reservoirs to facilitate the transformation of simple aromatic amines directly into azoxy compounds. This novel approach eliminates the need for stoichiometric reducing agents, replacing them with benign oxidants like hydrogen peroxide or tert-butyl hydroperoxide that produce water or tert-butanol as the primary byproducts. The process operates at moderate temperatures ranging from 68°C to 72°C, significantly reducing energy consumption compared to high-temperature pyrolysis or reflux conditions often seen in older methodologies. By shifting from a reduction-based paradigm to an oxidation-based catalytic cycle, the invention achieves a cleaner reaction profile with higher selectivity, effectively bypassing the formation of azo or hydrazo impurities that typically plague non-catalytic routes.
Mechanistic Insights into Polyoxometallate-Catalyzed Oxidative Coupling
The core innovation of this technology lies in the deployment of Anderson-type and Lindqvist-type polyoxometallates, which possess a unique structural ability to function simultaneously as Lewis acids and Lewis bases depending on the reaction environment. These polyanionic clusters feature a surface rich in oxygen atoms capable of donating electrons to acceptors, while their metal centers possess vacant orbitals that can accept electrons, creating a dynamic redox system. This dual functionality allows the catalyst to mediate the transfer of oxygen atoms from the oxidant to the amine substrate with high precision, stabilizing reactive intermediates that would otherwise decompose or polymerize. The reversible redox properties of the polyoxometallate skeleton enable it to cycle between different oxidation states without degradation, ensuring sustained catalytic activity throughout the prolonged reaction times of 12 to 48 hours required for complete conversion.
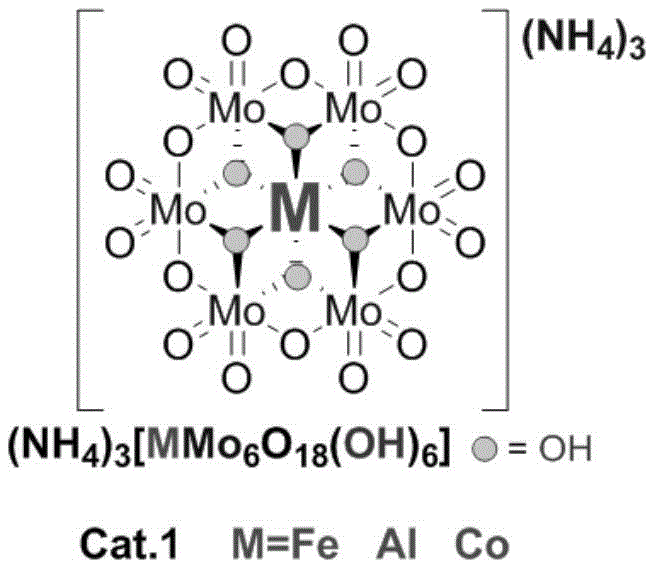
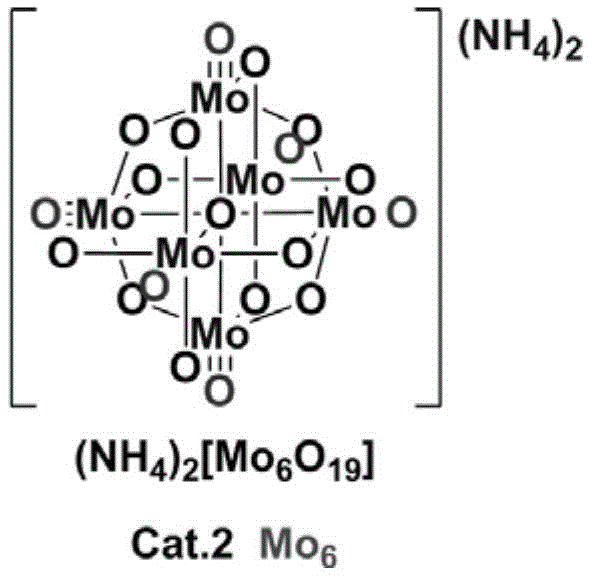
The structural integrity of the catalyst is paramount for maintaining high turnover numbers, and the patent specifies two distinct classes of polyoxometallates that have been optimized for this transformation. The Anderson-type catalysts, characterized by a central heteroatom surrounded by molybdenum-oxygen octahedra, allow for fine-tuning of electronic properties by varying the central metal ion such as Iron, Aluminum, or Cobalt. Conversely, the Lindqvist-type structure offers a compact, highly symmetric arrangement of six molybdenum atoms that provides a dense array of active sites for substrate activation. The choice between these structures depends on the specific electronic nature of the aromatic amine substrate, with electron-deficient amines potentially benefiting from the stronger Lewis acidity of the Iron-centered Anderson variant, while electron-rich substrates may proceed efficiently with the Molybdenum-centered Lindqvist cluster.
Impurity control in this system is achieved through the precise modulation of the oxidant-to-substrate ratio and the inclusion of specific additives like sodium sulfite or sodium bisulfite. These additives play a crucial role in buffering the reaction medium and preventing the over-oxidation of the azoxy product to the corresponding nitro compound or azo derivative, which are common side reactions in oxidative couplings. The patent data indicates that maintaining the molar ratio of oxidant within a specific window ensures that the reaction stops selectively at the azoxy stage, yielding products with purities exceeding 90% even before chromatographic purification. This inherent selectivity reduces the burden on downstream processing units, allowing for simpler workup procedures that rely on standard extraction and crystallization techniques rather than complex distillation columns.
How to Synthesize Aromatic Azoxy Compounds Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing the sequential addition of reagents to maximize catalyst efficiency and safety. The process begins with the charging of the reactor with the polyoxometallate catalyst and the sulfite additive, followed by the introduction of the organic solvent and the aromatic amine substrate to ensure homogeneous mixing before the oxidant is introduced. This order of addition is critical to prevent localized hot spots or rapid exotherms that could degrade the catalyst or lead to runaway reactions, particularly when using concentrated hydrogen peroxide. Detailed standardized synthetic steps for implementing this protocol are provided in the section below to guide process engineers in replicating these high-yielding results.
- Charge the reactor with the specific polyoxometallate catalyst (Anderson or Lindqvist type), additive (sodium sulfite/bisulfite), organic solvent, aromatic amine, and oxidant.
- Heat the mixture to 68-72°C and stir continuously for 12 to 48 hours to ensure complete conversion of the amine substrate.
- Upon completion, separate the product and recover the catalyst by adding volatile organic solvents like ether to precipitate the polyoxometallate for reuse.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this polyoxometallate-catalyzed route offers compelling economic and logistical benefits that extend far beyond simple yield improvements. The primary driver for cost reduction is the elimination of expensive stoichiometric reducing agents like borohydrides, which are not only costly to purchase but also expensive to dispose of safely due to their hazardous nature. By replacing these with catalytic amounts of reusable polyoxometallates and commodity oxidants like hydrogen peroxide, the raw material cost structure is drastically simplified, leading to substantial savings in both direct material costs and waste treatment expenses. Furthermore, the ability to recover and recycle the catalyst multiple times without significant loss of activity means that the effective cost per kilogram of catalyst consumed is negligible, enhancing the overall margin profile of the final product.
- Cost Reduction in Manufacturing: The transition to a catalytic oxidative process fundamentally alters the cost basis of aromatic azoxy compound manufacturing by removing the dependency on high-cost reducing agents and minimizing waste disposal fees. The use of inexpensive and widely available oxidants such as hydrogen peroxide, combined with the recyclability of the polyoxometallate catalyst, ensures that variable production costs remain low and predictable. Additionally, the mild reaction temperatures reduce energy consumption for heating and cooling, contributing to lower utility bills and a smaller carbon footprint for the manufacturing facility.
- Enhanced Supply Chain Reliability: Sourcing stability is significantly improved as the key reagents—aromatic amines, hydrogen peroxide, and simple inorganic salts—are commodity chemicals available from multiple global suppliers, reducing the risk of single-source bottlenecks. The robustness of the catalyst system against variations in substrate quality further insulates the supply chain from fluctuations in raw material purity, ensuring consistent output even when feedstock specifications vary slightly. This resilience is crucial for maintaining continuous production schedules and meeting tight delivery windows for downstream customers in the pharmaceutical and agrochemical sectors.
- Scalability and Environmental Compliance: The process is inherently scalable due to its operation in common organic solvents like toluene or MTBE and its lack of hazardous gas evolution, making it suitable for large-scale batch reactors without requiring specialized pressure vessels. From an environmental compliance perspective, the generation of water as the primary byproduct aligns perfectly with increasingly stringent green chemistry regulations, simplifying the permitting process for new production lines. The absence of heavy metal contamination in the final product also reduces the need for expensive metal scavenging steps, streamlining the path to commercial release for sensitive applications like API intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this polyoxometallate catalytic technology, derived directly from the experimental data and claims within the patent documentation. These insights are intended to clarify the operational parameters and potential advantages for stakeholders evaluating this technology for integration into their existing manufacturing portfolios. Understanding these nuances is essential for making informed decisions about process adoption and resource allocation.
Q: What are the primary advantages of using polyoxometallate catalysts over traditional reducing agents?
A: Unlike traditional methods utilizing expensive and environmentally hazardous reducing agents like KBH4, polyoxometallate catalysts offer a green oxidative coupling pathway. They function as reusable electron reservoirs, significantly lowering production costs and eliminating heavy metal waste streams associated with stoichiometric reductants.
Q: Can the polyoxometallate catalyst be recycled for industrial scale-up?
A: Yes, the patent explicitly details a recovery process where the catalyst is precipitated using volatile organic solvents such as ether or ethanol after the reaction. This recyclability is a critical factor for commercial viability, ensuring consistent catalyst performance across multiple batches without significant loss of activity.
Q: What types of aromatic amines are compatible with this catalytic system?
A: The system demonstrates broad substrate scope, successfully converting aniline and various substituted aromatic amines including those with methyl, fluoro, chloro, and bromo groups. This versatility makes it highly suitable for producing diverse intermediates required in dye and liquid crystal manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aromatic Azoxy Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the catalytic methods described in CN113019449A and are fully equipped to translate this academic innovation into commercial reality for our global partners. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of aromatic azoxy compound meets the exacting standards required for high-performance applications in liquid crystals and specialty dyes.
We invite you to engage with our technical procurement team to discuss how this advanced catalytic route can be tailored to your specific production needs and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that will demonstrate our capability to deliver high-quality intermediates with unmatched reliability and speed.
