Advanced Synthesis of 2,4-Dichlorobenzoyl Chloride for Commercial Scale-Up of Complex Organic Intermediates
Advanced Synthesis of 2,4-Dichlorobenzoyl Chloride for Commercial Scale-Up of Complex Organic Intermediates
The chemical industry is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally sustainable synthesis routes for critical intermediates. A significant breakthrough in this domain is documented in patent CN109678698B, which outlines a novel preparation method for 2,4-dichlorobenzoyl chloride, a vital building block in the synthesis of pharmaceuticals, agrochemicals, and dyes. This technology addresses long-standing challenges associated with traditional acyl chlorination methods, offering a pathway that combines high yield with operational simplicity. As a reliable fine chemical intermediates supplier, understanding the nuances of such patented technologies is crucial for ensuring supply chain resilience and product quality. The method described leverages a two-step process involving free radical chlorination followed by hydrolysis, achieving a total product yield between 90% and 95% with a purity exceeding 99%. This level of efficiency is not merely a laboratory curiosity but represents a robust industrial solution capable of meeting the rigorous demands of global markets. By adopting this advanced synthesis route, manufacturers can significantly enhance their production capabilities while adhering to stricter environmental regulations.
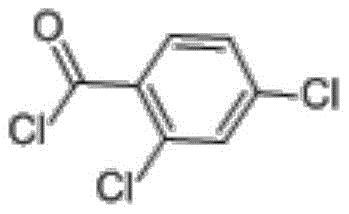
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of 2,4-dichlorobenzoyl chloride has relied heavily on the reaction of 2,4-dichlorobenzoic acid with acyl chlorinating agents such as thionyl chloride or phosphorus trichloride. While chemically feasible, these conventional methods present substantial drawbacks that hinder large-scale commercial viability. The use of thionyl chloride, for instance, generates significant amounts of sulfur dioxide and hydrogen chloride gases, necessitating complex and costly waste treatment systems to meet environmental compliance standards. Furthermore, the reaction conditions often require prolonged heating and refluxing, leading to higher energy consumption and increased operational costs. The presence of excess reagents also complicates the purification process, often resulting in lower overall yields and the potential for product contamination with residual sulfur or phosphorus impurities. These factors collectively contribute to a higher cost of goods sold and a larger environmental footprint, making traditional routes less attractive for modern, sustainability-focused manufacturing facilities seeking cost reduction in pharma intermediates manufacturing.
The Novel Approach
In stark contrast to the legacy methods, the novel approach detailed in the patent utilizes a side-chain chlorination strategy followed by hydrolysis, fundamentally shifting the chemical paradigm to one of greater efficiency and safety. This method begins with the chlorination of 2,4-dichlorotoluene, a readily available and inexpensive raw material, using chlorine gas in the presence of azobisisobutyronitrile (AIBN) as a catalyst. This shift eliminates the need for hazardous acyl chlorinating agents, thereby drastically simplifying the reaction setup and reducing the generation of toxic by-products. The subsequent hydrolysis step converts the chlorinated intermediate directly into the target acyl chloride with high selectivity. This streamlined process not only lowers the raw material costs but also minimizes the complexity of the downstream purification steps. By avoiding the use of corrosive reagents like thionyl chloride, the novel approach reduces equipment maintenance requirements and enhances operator safety. Consequently, this method offers a compelling value proposition for producers aiming to optimize their manufacturing processes while maintaining high-purity 2,4-dichlorobenzoyl chloride standards.
Mechanistic Insights into AIBN-Catalyzed Chlorination and Hydrolysis
The core of this innovative synthesis lies in the precise control of free radical chemistry during the chlorination phase. Azobisisobutyronitrile (AIBN) serves as a highly effective radical initiator, decomposing at elevated temperatures to generate nitrogen gas and carbon-centered radicals that propagate the chlorination chain reaction. The reaction is meticulously controlled at temperatures between 95°C and 105°C, ensuring that the chlorination occurs selectively at the methyl side chain of the toluene derivative rather than on the aromatic ring. This selectivity is paramount, as ring chlorination would lead to unwanted isomers that are difficult to separate and would degrade the quality of the final product. The process involves the gradual introduction of chlorine gas and the batched addition of the catalyst to maintain a steady reaction rate, preventing thermal runaways and ensuring consistent conversion. Gas chromatography is employed to track the reaction progress, with the endpoint defined by the residual amount of 2,4-dichlorotoluene dropping to less than 0.05%. This rigorous monitoring ensures that the intermediate, 2,4-dichlorotrichlorotoluene, is produced with high fidelity, setting the stage for the subsequent hydrolysis step.
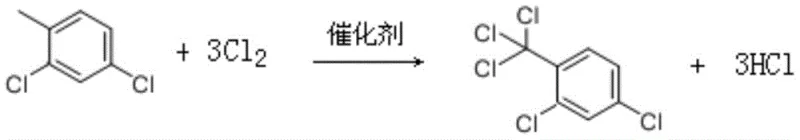
Following the chlorination, the hydrolysis mechanism plays a critical role in converting the trichloromethyl group into the desired acyl chloride functionality. This transformation is facilitated by the addition of water in the presence of a ferric trichloride catalyst at temperatures ranging from 110°C to 120°C. The ferric trichloride acts as a Lewis acid, coordinating with the chlorine atoms and enhancing the electrophilicity of the carbon center, thereby promoting nucleophilic attack by water molecules. This catalytic effect allows the hydrolysis to proceed rapidly and cleanly, minimizing the formation of carboxylic acid by-products which could otherwise form if the reaction conditions were too vigorous or uncontrolled. The reaction is again monitored via gas chromatography, terminating when the intermediate is consumed to below 0.05%. The resulting crude product is then subjected to reduced pressure rectification, a gentle purification technique that separates the target 2,4-dichlorobenzoyl chloride from any remaining impurities based on boiling point differences. This combination of catalytic hydrolysis and vacuum distillation ensures that the final product meets stringent purity specifications, often exceeding 99%, which is essential for its application in sensitive pharmaceutical and agrochemical syntheses.

How to Synthesize 2,4-Dichlorobenzoyl Chloride Efficiently
Implementing this synthesis route in a production environment requires careful attention to process parameters and safety protocols to maximize efficiency and yield. The procedure begins with the charging of 2,4-dichlorotoluene and a portion of the AIBN catalyst into a reactor equipped with efficient stirring and gas absorption systems. The system is maintained under a slight negative pressure to safely manage the evolution of hydrogen chloride gas, which is absorbed in an alkaline scrubber. As the reaction progresses, the remaining catalyst is added in batches to sustain the radical concentration without causing excessive heat generation. Once the chlorination is complete, the crude intermediate is transferred to the hydrolysis vessel where water is added dropwise under controlled heating. The use of ferric trichloride here is critical for accelerating the reaction without compromising selectivity. Finally, the crude acyl chloride is purified via vacuum distillation, collecting the fraction that boils at approximately 50°C under high vacuum. Detailed standardized synthesis steps see the guide below.
- Initiate free radical chlorination of 2,4-dichlorotoluene using azobisisobutyronitrile (AIBN) at 95-105°C to form 2,4-dichlorotrichlorotoluene.
- Perform hydrolysis of the crude chlorinated intermediate with water in the presence of ferric trichloride catalyst at 110-120°C.
- Purify the final product through reduced pressure rectification to achieve purity above 99% and yields between 90-95%.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis method offers profound benefits for procurement managers and supply chain leaders who are tasked with optimizing costs and ensuring material availability. The primary advantage stems from the use of 2,4-dichlorotoluene and chlorine gas as starting materials, which are commodity chemicals available in large volumes at stable prices, unlike specialized acyl chlorinating agents that can be subject to market volatility. This raw material accessibility translates directly into enhanced supply chain reliability, reducing the risk of production stoppages due to ingredient shortages. Furthermore, the elimination of thionyl chloride removes the logistical and disposal costs associated with hazardous waste management, leading to substantial cost savings in the overall manufacturing budget. The high yield of 90% to 95% means that less raw material is wasted per unit of product, further driving down the effective cost per kilogram. These factors combine to create a more resilient and cost-effective supply chain, enabling companies to offer competitive pricing to their downstream customers while maintaining healthy profit margins.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the substitution of expensive and hazardous reagents with inexpensive commodity chemicals, fundamentally altering the cost structure of production. By removing the need for thionyl chloride, manufacturers avoid the significant expenses related to the purchase, storage, and neutralization of this corrosive substance. Additionally, the high conversion efficiency ensures that the maximum amount of raw material is converted into saleable product, minimizing waste disposal fees. The simplified workflow also reduces labor hours and energy consumption, as the reaction conditions are less demanding than traditional reflux methods. These cumulative effects result in a significantly reduced cost of goods sold, allowing for more aggressive pricing strategies in the competitive fine chemicals market.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on niche reagents that have limited suppliers or long lead times. This novel method mitigates such risks by utilizing chlorine gas and toluene derivatives, which are produced by a vast global network of chemical manufacturers. This broad supplier base ensures that procurement teams can source materials quickly and reliably, even during periods of market disruption. The robustness of the process also means that production schedules are less likely to be impacted by equipment failures or safety incidents associated with handling highly corrosive materials. Consequently, companies can promise shorter lead times to their clients, enhancing their reputation as a dependable partner in the global supply chain.
- Scalability and Environmental Compliance: As regulatory pressures regarding industrial emissions and waste disposal intensify, the environmental profile of a manufacturing process becomes a critical business asset. This synthesis route generates significantly less hazardous waste compared to conventional methods, primarily producing hydrogen chloride which can be easily captured and converted into commercial grade hydrochloric acid. This not only reduces the environmental burden but also creates a potential revenue stream from by-product sales. The simplicity of the operation facilitates easy scale-up from pilot plants to full commercial production without the need for exotic equipment or complex safety systems. This scalability ensures that the technology can grow with market demand, supporting the commercial scale-up of complex organic intermediates without hitting technical bottlenecks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 2,4-dichlorobenzoyl chloride, based on the detailed specifications and benefits outlined in the patent literature. Understanding these aspects is vital for technical teams evaluating the feasibility of integrating this intermediate into their own synthesis pipelines. The answers provided reflect the consensus on best practices for handling and sourcing this chemical to ensure optimal performance in downstream applications.
Q: What are the primary advantages of the AIBN-catalyzed chlorination method over traditional thionyl chloride routes?
A: The AIBN-catalyzed route eliminates the need for corrosive acyl chlorinating agents like thionyl chloride, significantly reducing hazardous waste generation and simplifying the purification process while maintaining high product purity.
Q: How does the hydrolysis step ensure high purity in the final 2,4-dichlorobenzoyl chloride product?
A: By utilizing ferric trichloride as a catalyst during the hydrolysis of 2,4-dichlorotrichlorotoluene, the reaction proceeds with high selectivity, minimizing side reactions and allowing for efficient removal of by-products during subsequent vacuum distillation.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process is designed for industrial scalability, featuring simple operation, low energy consumption, and a total product yield between 90% and 95%, making it economically viable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4-Dichlorobenzoyl Chloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the technical potential of a synthesis route is only as valuable as its execution in a real-world manufacturing setting. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN109678698B are fully realized in practice. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 2,4-dichlorobenzoyl chloride meets the exacting standards required by the pharmaceutical and agrochemical industries. We understand that consistency is key, and our robust quality management systems are designed to detect and eliminate variability before it impacts your production. By partnering with us, you gain access to a supply chain that is not only reliable but also technically sophisticated, capable of supporting your most demanding projects.
We invite you to engage with our technical procurement team to discuss how we can tailor our production capabilities to your specific needs. Whether you require a Customized Cost-Saving Analysis for your current supply chain or need to verify specific COA data for a new project, we are ready to provide comprehensive support. We encourage you to request route feasibility assessments to explore how our advanced manufacturing techniques can enhance your product portfolio. Our commitment to transparency and technical excellence ensures that you receive not just a chemical product, but a strategic partnership that drives value and innovation in your operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
