Optimizing Agrochemical Intermediate Production via Novel Atmospheric Fluorination Technology
The global demand for high-performance agrochemicals continues to drive innovation in the synthesis of fluorinated intermediates, where efficiency and safety are paramount. Patent CN1894198A introduces a transformative methodology for the production of dialkyl α-fluoromalonates, specifically addressing the longstanding bottlenecks associated with nucleophilic fluorination reactions. This technology enables the conversion of α-chloro-β-ketoesters into their corresponding α-fluoro derivatives under remarkably mild conditions, eliminating the need for hazardous high-pressure equipment while simultaneously accelerating reaction kinetics. For R&D directors and process chemists, this represents a significant leap forward in managing the reactivity of hydrogen fluoride, allowing for precise control over substitution without compromising yield or purity. The strategic implementation of this atmospheric process offers a robust pathway for scaling the production of critical building blocks used in next-generation plant protection agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of α-fluoro-β-dicarbonyl compounds has been plagued by severe operational constraints that hinder scalability and economic viability. Traditional approaches often necessitate the use of pressurized reactors to facilitate the reaction between α-chloro precursors and hydrogen fluoride sources at elevated temperatures ranging from 103°C to 130°C. This requirement for high-pressure infrastructure not only inflates capital expenditure due to the need for specialized metallurgy and safety systems but also introduces significant operational risks when handling corrosive fluorinating agents. Alternatively, existing atmospheric methods described in prior art, such as those referenced in DE-A-4237882, suffer from prohibitively long reaction cycles, often extending up to 72 hours even when utilizing large excesses of reagents. These inefficiencies result in poor space-time yields, tying up reactor capacity and increasing energy consumption, which ultimately translates to higher manufacturing costs and supply chain vulnerabilities for downstream agrochemical producers.
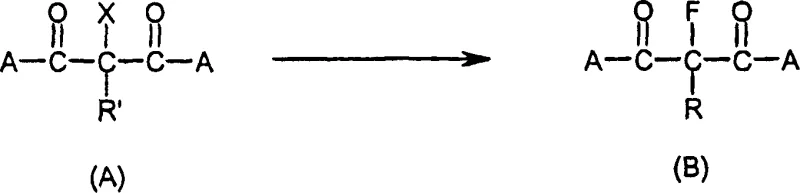
The Novel Approach
In stark contrast to these legacy techniques, the process disclosed in CN1894198A achieves a remarkable optimization of reaction parameters by utilizing a specific hydrogen fluoride-triethylamine addition product under strictly controlled atmospheric conditions. By operating within a narrow temperature window of 103°C to 115°C, preferably 105°C to 110°C, the method successfully drives the halogen exchange to completion in merely 15 hours, a drastic reduction compared to the multi-day cycles of conventional atmospheric routes. This acceleration is achieved without sacrificing yield, as the optimized stoichiometry ensures that only a slight excess of the fluorinating agent is required, thereby minimizing waste generation and raw material costs. The ability to conduct this transformation at normal pressure (800-1200 mbar) fundamentally alters the economic landscape of production, allowing manufacturers to utilize standard glass-lined or stainless steel reactors without the need for expensive autoclaves, thus facilitating easier commercial scale-up of complex agrochemical intermediates.

Mechanistic Insights into HF-Triethylamine Mediated Fluorination
The core of this technological advancement lies in the nuanced interaction between the hydrogen fluoride-triethylamine adduct and the electrophilic carbon center of the α-chloromalonate substrate. The triethylamine serves not merely as a base but as a crucial modifier that modulates the nucleophilicity and solubility of the fluoride ion, creating a reactive species capable of displacing the chlorine atom efficiently at moderate temperatures. Mechanistically, the reaction proceeds via a nucleophilic substitution pathway where the fluoride ion attacks the alpha-carbon, facilitated by the electron-withdrawing nature of the adjacent carbonyl groups which stabilize the transition state. Crucially, the patent highlights that maintaining the reaction temperature within the specified range prevents thermal decomposition of the sensitive fluorinated product, a common issue that typically plagues high-temperature fluorinations. This precise thermal control ensures that the integrity of the ester functionalities is preserved, resulting in a cleaner crude profile with fewer side products such as defluorinated species or hydrolysis byproducts.
From an impurity control perspective, the use of a defined molar ratio of hydrogen fluoride to triethylamine, specifically between 1.2 and 1.8 moles of HF per mole of amine, is instrumental in suppressing unwanted side reactions. Excess free acid can lead to ester hydrolysis or polymerization, while insufficient acidity may fail to activate the leaving group effectively. The process described ensures that the adduct remains stable throughout the 15-hour reflux period, providing a consistent concentration of active fluorinating species. Furthermore, the workup procedure involving extraction with solvents like xylene or toluene allows for the efficient separation of the organic product from the aqueous amine salts, simplifying downstream purification. This mechanistic robustness provides R&D teams with a reliable framework for adapting the process to various alkyl esters, ensuring consistent quality across different batches of high-purity agrochemical intermediates.
How to Synthesize Diethyl α-Fluoromalonate Efficiently
The practical execution of this synthesis involves a straightforward sequence of mixing, heating, and separation steps that are highly amenable to standard batch processing in a multipurpose chemical plant. The protocol begins with the preparation of the fluorinating agent, followed by the controlled addition of the chloro-precursor and a sustained heating period to drive the conversion. Detailed operational parameters regarding reagent ratios and temperature profiles are critical for replicating the high space-time yields reported in the patent data. For process engineers looking to implement this technology, the following guide outlines the standardized synthetic steps derived directly from the exemplary embodiments provided in the intellectual property documentation.
- Prepare the fluorinating agent by mixing triethylamine and hydrogen fluoride to form an addition product, ensuring a molar ratio of HF to TEA between 1.2 and 1.8.
- Meter the dialkyl α-chloromalonate substrate into the reaction vessel containing the HF-TEA adduct at a controlled temperature of approximately 80°C.
- Heat the reaction mixture to 105-110°C under atmospheric pressure and stir for approximately 15 hours to complete the halogen exchange.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this atmospheric fluorination process presents a compelling value proposition centered on operational efficiency and risk mitigation. By eliminating the dependency on high-pressure reactors, facilities can significantly reduce capital investment requirements and lower the barrier to entry for producing high-value fluorinated intermediates. The drastic reduction in reaction time from several days to less than one day means that existing reactor assets can be turned over much more frequently, effectively increasing production capacity without the need for physical expansion. This enhancement in throughput directly addresses supply chain bottlenecks, ensuring a more reliable flow of critical materials to downstream formulation plants and reducing the risk of stockouts during peak agricultural seasons.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the elimination of expensive high-pressure equipment and the substantial decrease in energy consumption associated with shorter reaction cycles. Since the method operates at atmospheric pressure, the need for specialized safety infrastructure and rigorous pressure testing is removed, leading to lower maintenance and compliance costs. Additionally, the stoichiometric efficiency of the reaction means that less raw material is wasted, and the reduced volume of waste streams lowers disposal expenses. These factors combine to create a leaner manufacturing model that delivers substantial cost savings in agrochemical intermediate manufacturing without compromising on product quality or yield.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route contributes to a more resilient supply chain by reducing the complexity of the manufacturing process. With shorter lead times for each batch, producers can respond more agilely to fluctuations in market demand, adjusting production schedules dynamically rather than being locked into long campaign runs. The use of commercially available starting materials and common solvents further mitigates the risk of raw material shortages, ensuring continuous operation. This reliability is crucial for maintaining long-term contracts with major agrochemical companies who require guaranteed delivery schedules to support their own global distribution networks.
- Scalability and Environmental Compliance: Scaling this process from pilot plant to commercial production is inherently safer and more straightforward due to the absence of high-pressure hazards. The simplified engineering requirements allow for faster technology transfer and quicker commissioning of new production lines. From an environmental standpoint, the reduced reaction time and improved atom economy result in a smaller carbon footprint per kilogram of product. The ability to recycle solvents like xylene and the manageable nature of the aqueous waste streams facilitate compliance with increasingly stringent environmental regulations, positioning manufacturers as responsible partners in the sustainable production of specialty chemicals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel fluorination technology. These insights are derived from the specific experimental data and comparative analysis presented in the patent literature, providing clarity on how this method outperforms traditional synthesis routes. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this process into their existing manufacturing portfolios.
Q: What are the safety advantages of this new fluorination process compared to traditional methods?
A: Unlike conventional high-pressure methods requiring specialized equipment and safety measures, this process operates at atmospheric pressure (800-1200 mbar), significantly reducing capital expenditure and operational risks associated with pressurized hydrogen fluoride handling.
Q: How does the reaction time of this novel method compare to existing atmospheric techniques?
A: While prior art atmospheric methods often require up to 72 hours to reach completion even with large reagent excesses, this optimized protocol achieves high yields in approximately 15 hours, drastically improving space-time yield and production throughput.
Q: What is the primary industrial application of dialkyl α-fluoromalonates produced by this route?
A: These compounds serve as critical intermediates for synthesizing 4,6-dichloro-5-fluoropyrimidines, which are essential precursors in the manufacturing of biologically active plant protection agents and other fluorinated heterocycles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diethyl α-Fluoromalonate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient fluorination technologies play in the modern agrochemical supply chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN1894198A are fully realized in industrial practice. We are committed to delivering high-purity dialkyl α-fluoromalonates that meet stringent purity specifications, supported by our rigorous QC labs which employ advanced analytical techniques to verify product identity and impurity profiles. Our capability to adapt and optimize such atmospheric processes allows us to offer a stable and cost-effective supply of these vital intermediates to global partners.
We invite procurement specialists and R&D leaders to collaborate with us on optimizing their supply chains for fluorinated building blocks. By leveraging our technical expertise, you can achieve a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that your project moves forward with the most efficient and reliable manufacturing strategy available in the market today.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
