Scalable Production of High-Purity Halogenated Cyclopropanes via Novel Diazotization Route
Introduction to Next-Generation Halogenated Cyclopropane Synthesis
The strategic incorporation of cyclopropyl moieties into small molecule therapeutics has become a cornerstone of modern medicinal chemistry, driven by the unique conformational rigidity and metabolic stability these structures impart to drug candidates. Patent CN112573992A introduces a transformative two-step synthetic methodology that addresses long-standing bottlenecks in the production of halogenated cyclopropanes, specifically targeting bromo-, chloro-, and iodo- derivatives which serve as critical building blocks for complex API synthesis. This innovative approach leverages a diazotization-metalation sequence that bypasses the need for expensive carboxylic acid precursors, thereby unlocking a more economically viable pathway for generating these high-value intermediates. By utilizing cyclopropylamine as the starting material, the process not only simplifies the supply chain but also enhances the overall atom economy of the transformation. The significance of this technology lies in its ability to deliver products with exceptional purity profiles, often exceeding 97% GC purity, which is paramount for downstream pharmaceutical applications where impurity control is strictly regulated.
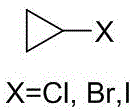
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of halogenated cyclopropanes has relied heavily on the decarboxylative halogenation of cyclopropanecarboxylic acids, a route fraught with significant economic and environmental disadvantages. As illustrated in prior art such as the method reported by Asahara T, this traditional pathway necessitates the use of cyclopropanecarboxylic acid, a raw material that is notoriously difficult to prepare and commands a high market price due to limited commercial availability. Furthermore, these legacy processes typically require stoichiometric amounts of toxic heavy metal salts as catalysts, which not only pose severe safety hazards during handling but also generate substantial quantities of hazardous waste streams containing heavy metals and halogens. The disposal of such halogen-rich wastewater presents a formidable challenge for chemical manufacturers, often requiring complex and costly treatment protocols to meet environmental compliance standards. Additionally, the reaction conversion rates in these conventional methods are frequently suboptimal, leading to lower overall yields and necessitating extensive purification steps that further erode profit margins and extend production timelines.
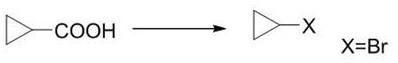
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the novel process disclosed in CN112573992A establishes a streamlined two-step protocol that fundamentally redefines the efficiency of halogenated cyclopropane manufacturing. The first stage involves the diazotization of readily available cyclopropylamine using nitrosating agents like tert-butyl nitrite in the presence of copper halides, effectively converting the amine into a 1,1-dihalocyclopropane intermediate with high selectivity. This is followed by a sophisticated metalation step where the dihalide intermediate reacts with organometallic reagents such as n-butyllithium, facilitating a controlled substitution that yields the target mono-halogenated product upon hydrolysis. This route eliminates the dependency on scarce carboxylic acid starting materials, replacing them with abundant amines, and operates under relatively mild thermal conditions that are far easier to manage on an industrial scale. The result is a robust synthetic platform that minimizes waste generation, avoids the use of persistent heavy metal catalysts in the final product, and delivers superior yields that make commercial production financially attractive.
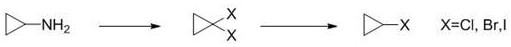
Mechanistic Insights into Copper-Mediated Diazotization and Lithiation
The core of this technological breakthrough lies in the precise orchestration of the diazotization and subsequent organometallic substitution mechanisms, which together ensure high fidelity in product formation. In the initial step, the interaction between cyclopropylamine and the nitrosating agent generates a reactive diazonium species in situ, which is immediately trapped by the halogenated copper salt to form the gem-dihalocyclopropane structure. This transformation is critical because it installs the necessary halogen handles while maintaining the integrity of the strained three-membered ring, a feat that requires careful control of reaction kinetics to prevent ring-opening side reactions. The choice of solvent, typically ethers like tetrahydrofuran or methyl tert-butyl ether, plays a pivotal role in stabilizing the intermediates and ensuring homogeneous reaction conditions throughout the process. Following isolation or in-situ processing, the 1,1-dihalocyclopropane undergoes selective metalation where one halogen atom is exchanged for a lithium species, creating a highly reactive organolithium intermediate that is poised for the final quenching step.
Impurity control in this system is achieved through rigorous management of the hydrolysis phase, where the organolithium species is carefully protonated to yield the final halogenated cyclopropane. The patent data indicates that maintaining the reaction temperature below -20°C during the metalation phase is essential to suppress competing elimination pathways that could lead to cyclopropene byproducts or ring-opened olefins. Furthermore, the use of specific molar ratios, such as a slight excess of the organometallic reagent (1.05 equivalents), ensures complete conversion of the dihalide precursor without promoting over-lithiation or degradation of the sensitive cyclopropyl ring. The resulting product profile is exceptionally clean, with gas chromatography analysis confirming purities of 98% for bromocyclopropane and similar high standards for chloro and iodo analogs. This level of chemical precision is vital for pharmaceutical clients who require intermediates with tightly defined impurity spectra to facilitate regulatory approval of their final drug substances.
How to Synthesize Halogenated Cyclopropane Efficiently
The operational execution of this synthesis requires adherence to strict protocol parameters regarding temperature, reagent addition rates, and solvent quality to maximize yield and safety. The process begins with the preparation of the 1,1-dihalocyclopropane intermediate, followed by the critical low-temperature lithiation step which demands specialized equipment capable of maintaining cryogenic conditions. Detailed standard operating procedures for scaling this reaction from laboratory benchtop to multi-ton production are essential for ensuring consistency and safety across different manufacturing sites. For a comprehensive breakdown of the specific reagent quantities, temperature ramps, and workup procedures validated in the patent examples, please refer to the standardized synthesis guide below.
- React cyclopropylamine with a nitrosating agent and a halogenated metal salt to form 1,1-dihalocyclopropane.
- Treat the 1,1-dihalocyclopropane intermediate with an organometallic reagent like n-butyllithium at low temperatures.
- Perform controlled hydrolysis of the metallated intermediate to yield the final high-purity halogenated cyclopropane.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented methodology offers profound advantages that directly address the pain points of cost volatility and supply chain fragility often associated with complex cyclic intermediates. By shifting the raw material base from expensive cyclopropanecarboxylic acid to commodity-grade cyclopropylamine, manufacturers can achieve a drastic reduction in direct material costs, insulating the supply chain from the price fluctuations typical of niche carboxylic acid suppliers. Moreover, the elimination of toxic heavy metal catalysts from the reaction matrix simplifies the downstream purification process, removing the need for expensive scavenging resins or complex extraction protocols designed to meet strict residual metal specifications. This simplification translates into shorter cycle times and reduced consumption of auxiliary chemicals, further driving down the total cost of ownership for the final intermediate. The robustness of the process also implies a higher reliability of supply, as the reaction is less prone to batch-to-batch variability caused by sensitive catalytic systems.
- Cost Reduction in Manufacturing: The substitution of high-cost starting materials with economically accessible amines creates a fundamental shift in the cost structure of halogenated cyclopropane production. By avoiding the procurement of specialized carboxylic acids and eliminating the expense associated with heavy metal waste disposal, the overall manufacturing overhead is significantly lowered. This economic efficiency allows for more competitive pricing models in the global market for pharmaceutical intermediates, providing a distinct advantage in tender situations where cost is a primary differentiator.
- Enhanced Supply Chain Reliability: The reliance on widely available bulk chemicals such as cyclopropylamine and standard organolithium reagents ensures that the supply chain is not vulnerable to the bottlenecks often seen with exotic or single-source reagents. The process uses common solvents like THF and MTBE, which are produced at massive scales globally, guaranteeing continuity of supply even during periods of market tightness. This resilience is crucial for long-term project planning, allowing procurement teams to secure multi-year contracts with confidence in the manufacturer's ability to deliver consistent volumes without interruption.
- Scalability and Environmental Compliance: The mild reaction conditions, operating primarily between -20°C and 50°C, are well within the capabilities of standard glass-lined or stainless steel reactors found in most multipurpose chemical plants, facilitating rapid technology transfer and scale-up. The reduction in hazardous waste generation, particularly the absence of heavy metal sludge, aligns perfectly with increasingly stringent global environmental regulations, reducing the regulatory burden and permitting risks associated with new process implementations. This environmental compatibility not only safeguards the manufacturer's license to operate but also appeals to end-clients who are prioritizing green chemistry metrics in their supplier selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this novel synthesis route. These answers are derived directly from the experimental data and technical disclosures within the patent documentation to provide accurate guidance for potential partners. Understanding these nuances is key to evaluating the feasibility of integrating this technology into existing production portfolios.
Q: What are the primary advantages of this synthesis method over traditional decarboxylation?
A: This method utilizes inexpensive cyclopropylamine instead of costly cyclopropanecarboxylic acid, avoids toxic heavy metal catalysts, and significantly reduces halogen-containing wastewater generation.
Q: What represents the critical control point for ensuring high purity in the final product?
A: Strict temperature control during the organometallic step (typically below -20°C) and precise hydrolysis conditions are essential to minimize side reactions and ensure purity exceeding 97%.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process employs mild reaction conditions, common solvents like THF and MTBE, and achieves high yields (up to 95%), making it highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Halogenated Cyclopropane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality halogenated cyclopropanes play in the development of next-generation therapeutics and agrochemicals. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify every batch against the highest industry standards. Our facility is equipped to handle the specific safety requirements of organometallic chemistry, including cryogenic operations and inert atmosphere processing, guaranteeing a safe and reliable supply of these valuable intermediates.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can optimize your supply chain and reduce your overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this superior process. We encourage you to contact us today to obtain specific COA data for our halogenated cyclopropane inventory and to receive detailed route feasibility assessments tailored to your specific project requirements.
