Advanced NBS-Mediated Bromination for Scalable Production of 3-Bromo-N-Arylpropionamide Intermediates
Advanced NBS-Mediated Bromination for Scalable Production of 3-Bromo-N-Arylpropionamide Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic methodologies that balance efficiency with safety and environmental compliance. A significant breakthrough in this domain is detailed in patent CN115260050A, which introduces a novel method for preparing 3-bromo-N-arylpropionamides using N-bromosuccinimide (NBS) as a key additive. This technology represents a paradigm shift from traditional acylation strategies, utilizing 3-methylthio-N-arylpropionamide as a stable precursor instead of hazardous acid chlorides. For R&D directors and procurement managers alike, this innovation offers a compelling value proposition by simplifying the supply chain for critical building blocks used in drug discovery and agrochemical synthesis. The method operates under mild conditions in dichloromethane, achieving high conversion rates while mitigating the risks associated with handling corrosive reagents. As a leading manufacturer, we recognize the potential of this route to redefine the standard for producing high-purity pharmaceutical intermediates, ensuring consistent quality and reliability for downstream applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-bromo-N-arylpropionamides has relied heavily on the condensation of arylamines with 3-bromopropionyl chloride under basic conditions. While chemically straightforward, this conventional pathway is fraught with significant logistical and safety challenges that impact both cost and operational continuity. 3-Bromopropionyl chloride is a lachrymator and a highly corrosive liquid that poses severe health risks to personnel and requires specialized containment infrastructure. Furthermore, its high volatility and susceptibility to hydrolysis make long-term storage difficult, often leading to material degradation and batch-to-batch variability. From a supply chain perspective, the reliance on such hazardous reagents necessitates stringent regulatory compliance, expensive waste treatment protocols for acidic byproducts, and complex transportation logistics. These factors collectively inflate the total cost of ownership and introduce fragility into the manufacturing schedule, making the conventional method less desirable for modern, green chemistry-focused production facilities.
The Novel Approach
In stark contrast, the methodology disclosed in CN115260050A employs a thioether-to-bromide transformation driven by NBS, effectively bypassing the need for unstable acid chlorides. This innovative route utilizes 3-methylthio-N-arylpropionamide, a stable solid that is easy to handle, store, and transport without special precautions. The reaction proceeds efficiently in dichloromethane at elevated temperatures, leveraging the electrophilic nature of NBS to replace the methylthio group with a bromine atom. This switch not only enhances operator safety but also streamlines the purification process, as the byproducts are generally easier to manage than the HCl gas evolved in traditional acylations. By adopting this greener alternative, manufacturers can achieve substantial cost reduction in pharmaceutical intermediate manufacturing through simplified safety protocols and reduced waste disposal burdens. The robustness of this method ensures a more reliable supply of high-quality intermediates, crucial for maintaining uninterrupted production lines in competitive markets.

Mechanistic Insights into NBS-Mediated Electrophilic Substitution
The core of this technological advancement lies in the efficient electrophilic substitution mechanism facilitated by N-bromosuccinimide. In this transformation, the sulfur atom of the thioether moiety acts as a nucleophile towards the bromine source, generating a reactive sulfonium intermediate that subsequently undergoes displacement by a bromide ion. This mechanism is particularly advantageous because it avoids the harsh acidic conditions typically required for activating carboxylic acid derivatives. The reaction conditions are optimized to ensure that the amide bond remains intact while the alkyl chain is selectively functionalized. Detailed studies within the patent indicate that the reaction kinetics are highly dependent on the stoichiometry of the brominating agent, with 1.5 equivalents of NBS proving optimal for driving the reaction to completion without excessive formation of dibrominated side products. This precise control over reactivity is essential for maintaining high purity profiles, a critical parameter for R&D directors evaluating new synthetic routes for API synthesis.
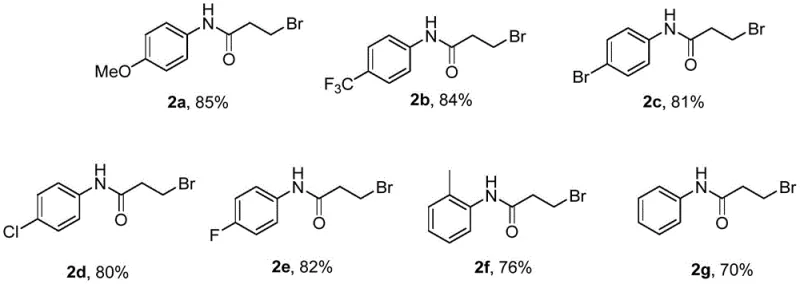
Furthermore, the protocol exhibits exceptional functional group tolerance, accommodating a wide array of electronic environments on the aromatic ring. Whether the substrate bears electron-donating groups like methoxy or methyl, or electron-withdrawing substituents such as trifluoromethyl and halogens, the reaction proceeds smoothly to afford the desired bromo-amides in yields ranging from 70% to 85%. This broad scope is demonstrated by the successful synthesis of various derivatives, including those with para-substituted methoxy, trifluoromethyl, bromo, chloro, and fluoro groups, as well as ortho-substituted methyl variants. Such versatility ensures that this method can be applied to the synthesis of diverse chemical libraries, accelerating the lead optimization phase in drug discovery. The ability to tolerate sensitive functional groups without protection-deprotection sequences further enhances the step economy of the overall synthesis, reducing both time and material costs.
How to Synthesize 3-Bromo-N-Arylpropionamide Efficiently
Implementing this synthesis requires careful attention to reaction parameters to maximize yield and purity. The standard protocol involves charging a sealed vessel with the thioether precursor and NBS in dichloromethane, followed by heating to 80°C for 12 hours. Optimization data reveals that temperature control is critical; while lower temperatures result in incomplete conversion, excessively high temperatures may lead to decomposition or side reactions. Similarly, the solvent choice is pivotal, with dichloromethane outperforming alternatives like acetonitrile or toluene in terms of solubility and reaction rate. The following guide outlines the standardized steps derived from the patent data to ensure reproducible results on both laboratory and pilot scales. Adhering to these optimized conditions allows for the efficient production of target compounds with minimal impurity formation.
- Charge a reaction vessel with 3-methylthio-N-arylpropionamide and N-bromosuccinimide (1.5 equivalents) in dichloromethane solvent.
- Heat the reaction mixture to 80°C and maintain vigorous stirring for 12 hours to ensure complete conversion.
- Upon completion, concentrate the reaction mixture under reduced pressure and purify the crude residue via column chromatography to isolate the target bromo-amide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this NBS-mediated protocol offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of raw material handling and storage logistics. By replacing volatile and corrosive 3-bromopropionyl chloride with stable solid reagents, companies can significantly reduce the overhead costs associated with hazardous material management. This shift eliminates the need for specialized corrosion-resistant equipment and reduces the frequency of safety audits related to toxic gas exposure. Moreover, the stability of the starting materials ensures longer shelf life, allowing for bulk purchasing opportunities that can further drive down unit costs. The streamlined workflow also translates to faster turnaround times, enhancing the overall agility of the supply chain in responding to market demands.
- Cost Reduction in Manufacturing: The elimination of toxic acid chlorides removes the necessity for expensive scrubbing systems and neutralization processes required to handle HCl off-gassing. Additionally, the use of NBS, a commercially abundant and cost-effective reagent, combined with the high atom economy of the transformation, leads to substantial cost savings. The simplified workup procedure, often requiring only concentration and chromatography, reduces solvent consumption and labor hours. These cumulative efficiencies result in a lower cost of goods sold (COGS), providing a competitive edge in pricing strategies for downstream pharmaceutical products.
- Enhanced Supply Chain Reliability: Sourcing stable solid intermediates mitigates the risk of supply disruptions caused by the degradation of sensitive liquid reagents during transit or storage. The robustness of the 3-methylthio-N-arylpropionamide precursor ensures consistent quality upon arrival, reducing the incidence of rejected batches due to原料 instability. This reliability fosters stronger relationships with suppliers and enables more accurate forecasting of production timelines. Consequently, manufacturers can maintain higher inventory turnover rates and reduce the capital tied up in safety stock, optimizing working capital utilization across the organization.
- Scalability and Environmental Compliance: The reaction conditions are inherently scalable, utilizing standard heating and stirring equipment available in most multipurpose chemical plants. The absence of highly corrosive reagents minimizes equipment wear and tear, extending the lifespan of reactor vessels and reducing maintenance downtime. From an environmental standpoint, the reduction in hazardous waste generation aligns with increasingly stringent global regulations on chemical manufacturing. This compliance not only avoids potential fines but also enhances the corporate sustainability profile, appealing to eco-conscious partners and investors who prioritize green chemistry initiatives in their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this NBS-mediated bromination technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this route into existing manufacturing portfolios. The answers provide clarity on safety, scalability, and product quality, ensuring that stakeholders have a comprehensive understanding of the process capabilities.
Q: Why is the NBS-mediated method superior to using 3-bromopropionyl chloride?
A: The traditional method utilizes 3-bromopropionyl chloride, which is highly toxic, corrosive, volatile, and difficult to store due to its sensitivity to moisture. The novel NBS-mediated approach uses stable solid reagents, significantly improving operational safety and shelf-life stability while maintaining high yields.
Q: What is the functional group tolerance of this bromination protocol?
A: The protocol demonstrates excellent compatibility with various substituents on the aryl ring, including electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups such as trifluoromethyl, halogens (fluoro, chloro, bromo), yielding products in 70-85% isolated yields.
Q: Can this process be scaled for commercial manufacturing?
A: Yes, the reaction utilizes common solvents like dichloromethane and standard heating conditions (80°C), making it highly amenable to scale-up. The avoidance of hazardous gaseous byproducts and the use of stable solid starting materials facilitate safer large-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Bromo-N-Arylpropionamide Supplier
At NINGBO INNO PHARMCHEM, we are committed to translating cutting-edge academic research into industrial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent CN115260050A can be seamlessly transferred to full-scale manufacturing. We understand that consistency is key in the pharmaceutical sector; therefore, our rigorous QC labs enforce stringent purity specifications for every batch of 3-bromo-N-arylpropionamide we produce. By leveraging our expertise in process optimization and hazard analysis, we guarantee a supply of high-purity intermediates that meet the exacting standards of global drug manufacturers. Our facility is equipped to handle the specific requirements of this NBS-mediated chemistry, ensuring safety and efficiency at every stage of production.
We invite you to collaborate with us to optimize your supply chain for these critical intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to reach out to request specific COA data and route feasibility assessments to determine how this advanced synthesis method can benefit your upcoming projects. Partnering with us means gaining access to a reliable network of chemical experts dedicated to driving innovation and efficiency in your production processes. Let us help you navigate the complexities of fine chemical sourcing with confidence and precision.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
