Advanced Electrochemical Synthesis of 3-Aryl-2-propyne-1-alcohol Derivatives for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct complex molecular architectures, particularly those containing propargyl alcohol motifs which serve as critical building blocks for bioactive compounds. Patent CN112110800A introduces a groundbreaking electrochemical methodology for the synthesis of 3-aryl-2-propyne-1-alcohol derivatives, addressing significant limitations in existing synthetic routes. This innovation leverages electro-organic synthesis to facilitate the coupling of arylacetylenes with formaldehyde equivalents under exceptionally mild conditions, eliminating the need for harsh reagents. The general structural framework of these valuable derivatives is depicted below, showcasing the versatility of the aryl group which can encompass pyridine, thiophene, naphthalene, or substituted phenyl rings.  As a reliable pharmaceutical intermediate supplier, understanding the nuances of such patented technologies is essential for delivering high-purity materials that meet stringent regulatory standards. The ability to access these scaffolds through a green, metal-free electrochemical process represents a paradigm shift in how we approach the manufacturing of key drug precursors, offering substantial advantages in both safety and operational simplicity for large-scale production facilities.
As a reliable pharmaceutical intermediate supplier, understanding the nuances of such patented technologies is essential for delivering high-purity materials that meet stringent regulatory standards. The ability to access these scaffolds through a green, metal-free electrochemical process represents a paradigm shift in how we approach the manufacturing of key drug precursors, offering substantial advantages in both safety and operational simplicity for large-scale production facilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-aryl-2-propyne-1-alcohol derivatives has been plagued by severe operational constraints and safety hazards that hinder industrial scalability. Traditional protocols predominantly rely on the use of strong bases such as alkyllithium, dialkylzinc, or organomagnesium reagents to activate terminal alkynes, necessitating cryogenic conditions as low as minus 78 degrees Celsius to control reactivity and prevent side reactions. Furthermore, alternative routes involving aryl halides and propargyl alcohol derivatives often require expensive noble metal catalysts like Palladium, which not only inflate raw material costs but also introduce challenging heavy metal removal steps during purification. The use of formaldehyde sources is another critical bottleneck; traditional methods utilize paraformaldehyde, trioxane, or toxic gaseous formaldehyde, all of which present significant handling risks, solubility issues in organic media, or environmental pollution concerns. These cumulative factors result in high energy consumption, complex waste treatment requirements, and limited substrate scope, making conventional synthesis economically unviable for cost-sensitive commercial applications.
The Novel Approach
In stark contrast to these legacy methods, the electrochemical strategy disclosed in CN112110800A offers a concise, one-step solution that operates at room temperature under an air atmosphere, drastically simplifying the reaction setup. By utilizing electricity as a clean reagent and employing inexpensive platinum plate electrodes, this method completely bypasses the need for transition metal catalysts and cryogenic cooling systems. A key innovation lies in the use of aryltrimethylammonium trifluoromethanesulfonate salts, which serve as a safe, solid-state source for the in-situ generation of formaldehyde within the organic solvent, thereby avoiding the handling of toxic gases. This approach not only broadens the substrate scope to include various heterocycles and substituted aromatics but also aligns perfectly with green chemistry principles by reducing waste and energy usage. For procurement managers, this translates to a robust supply chain capable of delivering high-purity pharmaceutical intermediates without the volatility associated with hazardous reagent sourcing.
Mechanistic Insights into Electrochemical Hydroxymethylation
The core of this technological advancement lies in the unique electrochemical activation mechanism that facilitates the coupling of the alkyne and the formaldehyde equivalent without external oxidants or reductants. In the undivided cell setup, the application of a constant current drives the redox processes at the electrode surfaces, where the ammonium salt undergoes electrochemical decomposition to release formaldehyde directly into the reaction medium. Simultaneously, the terminal alkyne is activated, likely through an anodic oxidation or base-mediated deprotonation enhanced by the electric field, allowing it to attack the electrophilic formaldehyde species. This synergistic interaction ensures high atom economy and minimizes the formation of by-products commonly associated with stoichiometric metal reagents. The reaction proceeds efficiently in polar aprotic solvents like N,N-dimethylacetamide (DMA) with sodium carbonate acting as a mild base, creating a buffered environment that supports the electrochemical cycle while maintaining the integrity of sensitive functional groups on the aromatic ring.
From an impurity control perspective, this mechanism offers distinct advantages for producing high-purity pharmaceutical intermediates required for clinical trials and commercial API manufacturing. Since the process avoids transition metals like Copper or Palladium, the risk of metal leaching into the final product is virtually eliminated, removing the need for expensive scavenging resins or complex chelation steps during downstream processing. The mild reaction conditions also prevent thermal degradation of the product or polymerization of the alkyne, which are common issues in high-temperature catalytic methods. As demonstrated in the specific embodiments, the reaction yields are consistently high, ranging from approximately 53 percent to over 95 percent depending on the electronic nature of the substituents, with product purity reaching 99 percent after standard silica gel chromatography. 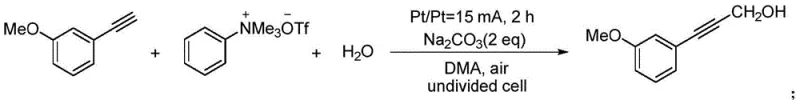 This level of purity and consistency is critical for reducing lead time for high-purity intermediates, ensuring that downstream synthesis steps are not compromised by trace contaminants.
This level of purity and consistency is critical for reducing lead time for high-purity intermediates, ensuring that downstream synthesis steps are not compromised by trace contaminants.
How to Synthesize 3-Aryl-2-propyne-1-alcohol Efficiently
The practical implementation of this synthesis route is designed for ease of operation, requiring standard laboratory glassware and a simple DC power supply, making it highly adaptable for both R&D screening and pilot plant operations. The procedure involves charging a three-necked flask with the arylacetylene substrate, the ammonium salt formaldehyde source, a mild inorganic base, water, and an organic solvent, followed by the insertion of platinum electrodes. The detailed standardized synthesis steps, including specific molar ratios, current densities, and workup procedures derived from the patent examples, are outlined in the guide below to assist technical teams in replicating this efficient process.
- Combine arylacetylene compounds, ammonium salts, alkali, water, and organic solvent in a three-necked flask equipped with platinum plate electrodes.
- Perform electrolysis at room temperature under an air atmosphere with stirring for 1 to 3 hours using a constant current of 10mA to 20mA.
- Extract the reaction mixture with ethyl acetate, dry over anhydrous sodium sulfate, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the adoption of this electrochemical technology presents a compelling value proposition centered around cost stability, safety compliance, and operational efficiency. By shifting away from volatile commodity chemicals like gaseous formaldehyde and expensive noble metal catalysts, manufacturers can achieve significant cost reduction in pharmaceutical intermediate manufacturing while mitigating supply chain disruptions caused by reagent scarcity. The elimination of cryogenic requirements further reduces utility costs associated with cooling and energy consumption, contributing to a lower overall cost of goods sold (COGS). Moreover, the use of stable solid salts instead of hazardous gases enhances workplace safety profiles, reducing insurance liabilities and regulatory burdens associated with storing toxic materials.
- Cost Reduction in Manufacturing: The removal of palladium and copper catalysts from the process flow eliminates a major cost driver and simplifies the purification workflow, leading to substantial cost savings. Without the need for specialized metal scavengers or extensive washing protocols to meet residual metal specifications, the downstream processing becomes faster and less resource-intensive. Additionally, the use of electricity as the primary driving force is generally more economical than purchasing stoichiometric amounts of strong organometallic bases, further optimizing the raw material expenditure for large-scale batches.
- Enhanced Supply Chain Reliability: Sourcing stable ammonium salts is significantly more reliable than managing the logistics of toxic gaseous formaldehyde or moisture-sensitive organolithium reagents. This stability ensures consistent production schedules and reduces the risk of batch failures due to reagent degradation or delivery delays. The robustness of the reaction under air atmosphere also means that strict inert gas blanketing is not required, simplifying the reactor setup and allowing for more flexible manufacturing scheduling in multi-purpose facilities.
- Scalability and Environmental Compliance: The simplicity of the undivided cell setup and the use of common solvents like DMA facilitate straightforward commercial scale-up of complex organic molecules from gram to ton scales. The process generates minimal hazardous waste compared to traditional methods, aligning with increasingly strict environmental regulations and corporate sustainability goals. This green profile not only future-proofs the manufacturing site against regulatory changes but also enhances the brand reputation of the final pharmaceutical products by associating them with sustainable production practices.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this patented technology, we have compiled a set of frequently asked questions based on the specific data and embodiments provided in the patent documentation. These answers address common concerns regarding safety, catalyst removal, and process scalability, providing a clear overview for technical decision-makers evaluating this synthesis route for their supply chain.
Q: How does this electrochemical method improve safety compared to traditional formaldehyde sources?
A: Traditional methods often require toxic gaseous formaldehyde or unstable paraformaldehyde. This patent utilizes aryltrimethylammonium trifluoromethanesulfonate salts which generate formaldehyde in situ within the organic solvent, eliminating the need to handle hazardous gases directly.
Q: Does this process require expensive transition metal catalysts like Palladium?
A: No, unlike conventional cross-coupling reactions that rely on costly Palladium or Copper catalysts, this electrochemical approach uses simple platinum plate electrodes and electricity as the primary driving force, significantly reducing raw material costs and metal contamination risks.
Q: What are the typical reaction conditions regarding temperature and atmosphere?
A: The process operates under remarkably mild conditions, specifically at room temperature and under a standard air atmosphere. This contrasts sharply with traditional methods requiring cryogenic conditions (-78°C) or inert gas protection, simplifying operational requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Aryl-2-propyne-1-alcohol Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in modernizing the production of critical pharmaceutical intermediates. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN112110800A can be successfully translated into robust industrial processes. We are committed to delivering high-purity materials that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards for residual solvents and impurities.
We invite global partners to collaborate with us to leverage this advanced technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can enhance your supply chain efficiency and reduce your overall manufacturing costs.
