Scalable Synthesis of 1-N-Ethyl Gentamicin C1a: A High-Yield Route for Etimicin Production
The pharmaceutical industry continuously seeks robust and efficient pathways for the production of next-generation aminoglycoside antibiotics, particularly for critical intermediates like 1-N-ethyl gentamicin C1a, the key precursor to Etimicin sulfate. Patent CN103665068A introduces a transformative preparation method that addresses long-standing inefficiencies in aminoglycoside modification. This innovation shifts the paradigm from traditional, hazardous ethylation processes to a streamlined reductive alkylation strategy. By leveraging a specific combination of sodium borohydride, chloroform, and glacial acetic acid, the method achieves exceptional conversion rates while mitigating the formation of complex impurity profiles. For R&D directors and procurement managers alike, this represents a significant opportunity to optimize the supply chain for high-value antibiotic intermediates. The technical breakthrough lies not just in the yield improvement, but in the fundamental simplification of the reaction mechanism, which reduces the reliance on unstable intermediates and toxic reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
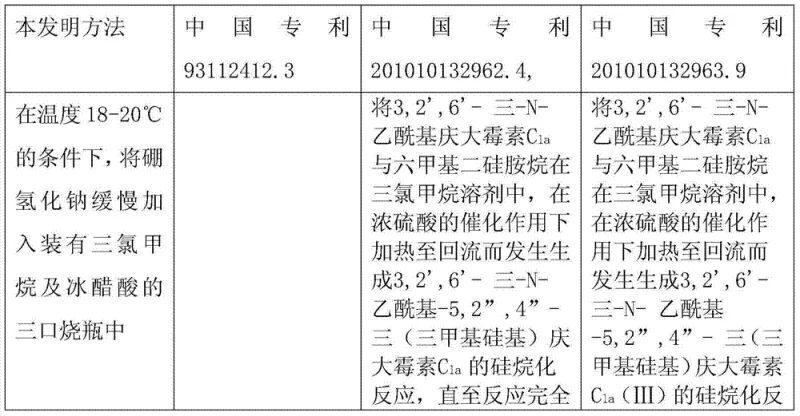
Historically, the synthesis of 1-N-ethyl gentamicin C1a has been plagued by significant technical and operational hurdles. Prior art, such as the methods disclosed in Chinese patents 93112412.3 and 201010132962.4, typically relies on the direct ethylation of the gentamicin backbone using acetaldehyde. This approach is inherently problematic because the formation of the imine intermediate is highly sensitive to pH fluctuations and reaction conditions. The instability of this imine often leads to extensive side reactions, resulting in a cluttered impurity profile that is notoriously difficult to purify. Furthermore, the use of acetaldehyde introduces severe safety and environmental concerns due to its volatility and toxicity, necessitating expensive containment systems and rigorous waste treatment protocols. Consequently, these conventional routes often suffer from mediocre yields, typically hovering around 30% to 45%, which drastically inflates the cost of goods sold and limits the scalability of the final API production.
The Novel Approach
In stark contrast, the methodology outlined in CN103665068A circumvents these pitfalls by employing a direct reductive alkylation strategy that bypasses the isolation of unstable intermediates. By conducting the reaction in a chloroform and glacial acetic acid medium, the process creates an optimal environment for the in-situ generation and immediate reduction of the reactive species. This eliminates the need for harsh ethylating agents and stabilizes the reaction trajectory, leading to a much cleaner product profile. The data indicates a dramatic leap in efficiency, with yields consistently reaching between 83% and 88%, nearly doubling the output of traditional methods. This substantial improvement in mass balance directly translates to reduced raw material consumption and lower waste generation. For a reliable pharmaceutical intermediate supplier, adopting this route means offering a product with superior cost-efficiency and consistent quality, addressing the critical pain points of cost reduction in API manufacturing.
Mechanistic Insights into Borohydride-Mediated Reductive Alkylation
The core of this technological advancement lies in the precise orchestration of the reductive amination mechanism within a non-aqueous, acidic organic phase. Unlike aqueous systems where borohydride decomposes rapidly, the use of chloroform and glacial acetic acid allows for the controlled generation of the active reducing species. The acetic acid serves a dual purpose: it protonates the carbonyl oxygen of the aldehyde equivalent (generated in situ or added) to facilitate nucleophilic attack by the amine, forming the iminium ion, and it moderates the reactivity of the sodium borohydride. This moderation is crucial; it prevents the violent decomposition of the hydride source while ensuring sufficient reducing power to convert the iminium ion to the secondary amine. The reaction is maintained at a mild temperature range of 40-50°C, which provides enough thermal energy to drive the kinetics forward without promoting thermal degradation of the sensitive glycosidic bonds in the gentamicin structure. This delicate balance ensures high regioselectivity for the 1-N position, minimizing N-alkylation at other susceptible amino groups on the sugar rings.
Furthermore, the impurity control mechanism is intrinsically linked to the stability of the reaction medium. In traditional aqueous or alcoholic reductions, competing hydrolysis or over-reduction can occur. However, the specific solvent system employed here suppresses these side pathways. The subsequent workup involves a neutralization step followed by a rigorous hydrolysis using 1N sodium hydroxide under reflux. This step is critical for removing the acetyl protecting groups from the 3, 2', and 6' positions without compromising the newly formed 1-N-ethyl bond. The robustness of the 1-N-ethyl bond under these basic hydrolysis conditions confirms the stability of the alkylation. The final purification via column chromatography, while standard, is rendered more effective because the preceding reaction steps generate fewer structural analogs. This results in a final product with purity levels exceeding 95%, meeting the stringent specifications required for parenteral antibiotic formulations.
How to Synthesize 1-N-Ethyl Gentamicin C1a Efficiently
The practical execution of this synthesis is designed for reproducibility and ease of operation in a standard chemical manufacturing setting. The process begins with the careful preparation of the reducing environment, where temperature control is paramount during the addition of sodium borohydride to prevent exothermic runaway. Following the addition of the protected gentamicin substrate, the reaction is allowed to proceed under vigorous stirring to ensure homogeneous mass transfer, which is vital for maximizing yield in heterogeneous or viscous mixtures. The extended reaction time of 20 hours ensures complete conversion of the starting material, minimizing the burden on downstream purification. After the reaction is quenched and the organic layer separated, the deprotection step is carried out under reflux, a unit operation familiar to any process chemist.
- Prepare the reaction system by slowly adding sodium borohydride to a mixture of chloroform and glacial acetic acid at 18-20°C.
- Dissolve 3,2',6'-tri-N-acetyl gentamicin C1a in chloroform and add it dropwise to the reaction flask, maintaining temperature at 40-50°C for 20 hours.
- Neutralize with saturated sodium hydroxide, extract with chloroform, concentrate, and reflux the residue with 1N NaOH for 24 hours to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis route offers profound advantages for supply chain resilience and cost management. The elimination of acetaldehyde not only improves safety but also removes a volatile commodity from the bill of materials, stabilizing procurement against market fluctuations. The significant increase in yield from roughly 30% to over 85% implies that less than half the amount of starting material is required to produce the same quantity of final product. This drastic reduction in material intensity directly lowers the variable cost per kilogram, providing a substantial margin buffer for manufacturers. Additionally, the simplified workflow reduces the number of unit operations and the complexity of waste streams, leading to lower operational expenditures related to effluent treatment and energy consumption.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the dramatic improvement in reaction yield. By achieving yields of 85% compared to the historical average of 30-45%, the consumption of the expensive gentamicin C1a starting material is effectively halved. This efficiency gain cascades through the entire production line, reducing the load on solvent recovery systems and minimizing the volume of chemical waste that requires disposal. Furthermore, the avoidance of toxic acetaldehyde eliminates the need for specialized scrubbing systems and reduces insurance and compliance costs associated with hazardous air pollutants. These factors combined result in a significantly leaner cost structure for the production of high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The reagents utilized in this novel method—sodium borohydride, chloroform, and acetic acid—are bulk commodity chemicals with stable global supply chains. Unlike specialized catalysts or unstable intermediates that may face sourcing bottlenecks, these materials are readily available from multiple vendors worldwide. This diversification of supply sources mitigates the risk of production stoppages due to raw material shortages. Moreover, the robustness of the reaction conditions (mild temperatures and atmospheric pressure) means that the process can be easily transferred between different manufacturing sites without requiring bespoke equipment modifications, ensuring continuity of supply even in the event of regional disruptions.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of extreme conditions or sensitive catalytic systems. The reaction exotherm is manageable, and the workup procedures involve standard liquid-liquid extraction and distillation techniques common in fine chemical plants. From an environmental standpoint, the process generates less hazardous waste by avoiding heavy metal catalysts and toxic alkylating agents. The solvent system, primarily chloroform, can be efficiently recovered and recycled, aligning with green chemistry principles and reducing the overall environmental footprint. This compliance readiness facilitates faster regulatory approvals and smoother audits from international pharmaceutical clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. They are derived from the specific experimental data and comparative analysis provided in the patent documentation. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer.
Q: How does this new method improve yield compared to traditional acetaldehyde ethylation?
A: Traditional methods utilizing acetaldehyde often suffer from unstable imine intermediates and side reactions, resulting in yields around 30-45%. The novel reductive alkylation method described in patent CN103665068A achieves yields of 83-88% by stabilizing the reaction environment with acetic acid and chloroform.
Q: What are the safety advantages of avoiding acetaldehyde in this synthesis?
A: Acetaldehyde is a volatile and toxic reagent that poses significant handling risks and requires strict containment. By replacing the ethylation step with a direct reductive alkylation using sodium borohydride, the process eliminates the need for toxic acetaldehyde, significantly improving operational safety and reducing environmental compliance burdens.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes common industrial solvents like chloroform and reagents like sodium borohydride. The reaction conditions are mild (40-50°C) and do not require exotic catalysts or extreme pressures, making it highly adaptable for commercial scale-up from pilot plants to multi-ton production facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-N-Ethyl Gentamicin C1a Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep process engineering expertise. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this high-yield route are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 1-N-ethyl gentamicin C1a meets the exacting standards required for antibiotic synthesis. Our commitment to quality assurance means that we can reliably support your clinical and commercial needs with a consistent supply of this critical intermediate.
We invite you to collaborate with us to optimize your supply chain for Etimicin and related aminoglycosides. By leveraging our technical capabilities, you can achieve significant operational efficiencies and secure a stable source of high-quality materials. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing excellence can drive value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
