Advanced Glycerol Hydrochlorination Technology for Scalable Epichlorohydrin and Chlorohydrins Production
Introduction to Next-Generation Chlorohydrins Synthesis
The global chemical industry is currently witnessing a paradigm shift towards sustainable feedstock utilization, particularly in the synthesis of critical intermediates like chlorohydrins and epichlorohydrin. Patent CN103025692A discloses a groundbreaking method for preparing chlorohydrins compositions by reacting polyhydroxy aliphatic hydrocarbons, specifically glycerol, with a chlorinating agent in the presence of a catalyst. This technology addresses the longstanding economic and environmental inefficiencies associated with traditional propylene-based routes by leveraging the surplus glycerol generated from the booming biodiesel sector. By integrating a sophisticated sequence of unit operations including dual reaction stages and intermediate dehydration, this process achieves exceptional conversion rates and selectivity, positioning it as a cornerstone technology for modern fine chemical intermediates manufacturing.
For R&D directors and procurement strategists, understanding the nuances of this patent is vital for securing a reliable fine chemical intermediates supplier capable of delivering high-purity materials at scale. The disclosed method not only mitigates the volatility of propylene pricing but also drastically reduces the environmental footprint by eliminating the need for excessive industrial water usage during the chlorination phase. As we delve deeper into the technical specifics, it becomes evident that this approach offers a robust solution for cost reduction in fine chemical intermediates manufacturing while adhering to increasingly stringent global environmental regulations regarding waste discharge and carbon efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of chlorohydrins has relied heavily on the chlorination of propylene, a process fraught with significant logistical and economic challenges that impact the entire supply chain. The conventional method typically involves a two-step procedure where propylene is first subjected to high-temperature chlorination to produce allyl chloride, which is subsequently reacted with a chlorinating agent using large volumes of industrial water. This reliance on propylene exposes manufacturers to severe supply instability driven by fluctuating petrochemical markets, while the requirement for excess water generates substantial volumes of wastewater and solid waste that require costly treatment and disposal. Furthermore, the initial capital investment for constructing or retrofitting facilities to handle these hazardous and waste-intensive processes is prohibitively high, creating a barrier to entry for new market participants and limiting the scalability of existing operations.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes glycerol, a renewable and economically attractive byproduct of biodiesel production, as the primary feedstock for direct chlorohydrin synthesis. This one-step methodology fundamentally alters the economic equation by replacing volatile petrochemical inputs with stable, bio-based alternatives, thereby insulating the production process from oil price shocks. Crucially, the process design ingeniously circumvents the thermodynamic limitation of water inhibition by incorporating an intermediate dehydration step, allowing the reaction to proceed with high efficiency without the massive water consumption characteristic of the propylene route. This innovation not only lowers the initial investment costs by simplifying the reactor train but also aligns perfectly with green chemistry principles, offering a compelling value proposition for companies seeking to enhance their sustainability profiles while securing a consistent supply of high-quality intermediates.
Mechanistic Insights into Glycerol Hydrochlorination
The core of this technological breakthrough lies in its intricate reaction engineering, which orchestrates a series of catalytic events to maximize the yield of dichloropropanol, the direct precursor to epichlorohydrin. The process initiates in a first reactor where glycerol reacts with hydrogen chloride gas in the presence of an organic acid catalyst, such as acetic acid, at temperatures ranging from 50°C to 200°C. Under these conditions, the catalyst does not merely accelerate the reaction but actively participates by forming intermediate species like glyceryl acetate, which subsequently act as effective catalysts for the chlorination of the polyhydroxy backbone. This dual-function mechanism ensures that even as the reaction progresses and water is generated, the catalytic activity remains robust, preventing the stagnation often seen in single-stage batch processes.
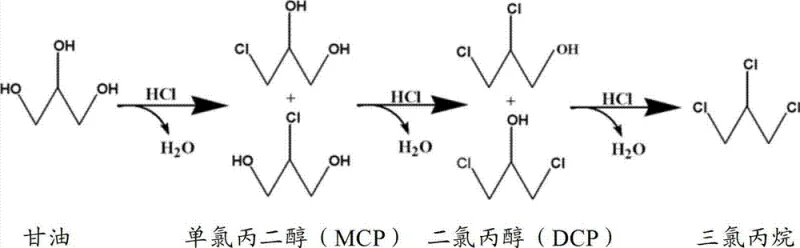
Following the initial reaction phase, the effluent undergoes a critical dehydration step via vacuum distillation, where water is separated as a water-rich layer while the water-deficient layer containing unreacted glycerol and chlorohydrins is fed into a second reactor. This second stage operates at elevated temperatures between 70°C and 200°C, facilitating the further substitution of hydroxyl groups with chlorine atoms to produce dichloropropanol with high selectivity. The patent data indicates that by carefully controlling the residence time and pressure conditions—specifically maintaining the dehydration unit between 10 mmHg and 760 mmHg—the process minimizes the formation of high-boiling byproducts and prevents pipeline blockages. The result is a chlorohydrin composition with a dichloropropanol selectivity reaching up to 94.8%, demonstrating the efficacy of this multi-stage continuous flow architecture in driving the equilibrium towards the desired product.
How to Synthesize Chlorohydrins Efficiently
Implementing this synthesis route requires precise control over reaction parameters and separation units to replicate the high yields observed in the patent examples. The process demands a seamless integration of continuous stirred tank reactors (CSTR) with high-efficiency vacuum distillation columns to manage the azeotropic behavior of the reaction mixture. Operators must ensure that the molar ratios of glycerol to氯化剂 are optimized to prevent over-chlorination to trichloropropane while maintaining sufficient acidity to drive the substitution reactions. For a detailed breakdown of the operational parameters and equipment specifications required to achieve these results, please refer to the standardized synthesis guide below.
- React glycerol with hydrogen chloride in a first reactor using an acetic acid catalyst to form mono- and dichlorohydrins along with glyceryl acetate intermediates.
- Separate water by-product via vacuum distillation to prevent reaction inhibition, then feed the dehydrated mixture into a second reactor for further chlorination.
- Treat the final chlorohydrin composition with an alkaline reagent to precipitate the catalyst as alkali metal salts, followed by distillation to isolate pure epichlorohydrin.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this glycerol-based technology offers profound advantages that extend far beyond simple raw material substitution, fundamentally reshaping the cost structure and reliability of the supply chain. By decoupling production from the petrochemical value chain, manufacturers can achieve significant cost reduction in fine chemical intermediates manufacturing through the utilization of low-cost, abundant glycerol derived from the global biodiesel surplus. This shift eliminates the exposure to the volatile pricing cycles of propylene, allowing for more accurate long-term budgeting and contract stability for downstream customers in the pharmaceutical and polymer sectors. Furthermore, the reduction in waste generation translates directly into lower operational expenditures related to environmental compliance and waste treatment, enhancing the overall margin profile of the production facility.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of recoverable organic acid catalysts significantly lower the variable costs associated with reagent consumption. Additionally, the process design allows for the recycling of unreacted intermediates and chlorinating agents back into the reactor system, minimizing raw material loss and maximizing atom economy. This closed-loop approach ensures that the cost per kilogram of produced chlorohydrins is substantially optimized compared to linear once-through processes, providing a competitive edge in price-sensitive markets.
- Enhanced Supply Chain Reliability: Sourcing glycerol from the biodiesel industry provides a diversified and geographically widespread supply base that is less susceptible to the geopolitical disruptions often affecting oil and gas feedstocks. The ability to operate continuous reactors with high throughput capabilities ensures a steady stream of output, reducing lead times for high-purity fine chemical intermediates and enabling Just-In-Time delivery models for key accounts. This reliability is crucial for pharmaceutical clients who require uninterrupted supply to maintain their own production schedules and regulatory filings.
- Scalability and Environmental Compliance: The modular nature of the reactor and distillation units described in the patent facilitates easy commercial scale-up from pilot plants to multi-ton annual production capacities without requiring exponential increases in footprint. Moreover, the drastic reduction in wastewater generation simplifies the permitting process and reduces the risk of regulatory shutdowns due to environmental non-compliance. This sustainability advantage not future-proofs the asset against tightening environmental laws but also appeals to end-consumers increasingly demanding green-certified supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this advanced chlorohydrins production technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific application needs. Understanding these details is essential for making informed decisions about adopting this technology for large-scale manufacturing.
Q: How does the glycerol route compare to the traditional propylene route for chlorohydrins?
A: Unlike the traditional propylene route which requires high-temperature chlorination and generates significant wastewater, the glycerol route utilizes biodiesel byproducts, eliminates the need for industrial water in the reaction step, and offers a more stable raw material supply chain.
Q: What is the role of the dehydration step in this specific patent process?
A: Water is a byproduct that inhibits the chlorination reaction. The patented process employs a vacuum distillation unit between two reactors to continuously remove water, thereby driving the equilibrium forward and maintaining high reaction rates and selectivity for dichloropropanol.
Q: How is the catalyst removed to ensure product purity?
A: The process utilizes an acetic acid catalyst which forms intermediates. In the final stage, the chlorohydrin composition is contacted with an alkaline reagent like sodium hydroxide, converting the catalyst into insoluble alkali metal salts which are easily separated via distillation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chlorohydrins Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the glycerol hydrochlorination pathway and have invested heavily in mastering the complexities of scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team possesses extensive experience in optimizing reaction conditions to meet stringent purity specifications, ensuring that every batch of chlorohydrins or epichlorohydrin delivered meets the rigorous quality standards required by the global pharmaceutical and specialty chemical industries. With our state-of-the-art rigorous QC labs and flexible manufacturing infrastructure, we are uniquely positioned to bridge the gap between innovative patent chemistry and reliable commercial supply.
We invite you to engage with our technical procurement team to discuss how we can tailor this technology to your specific volume and purity requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to our bio-based intermediates. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that will demonstrate our commitment to being your trusted partner in sustainable chemical manufacturing.
