Advanced Manufacturing of Dyclonine Hydrochloride via Novel Cu-Catalyzed C-O Cross-Coupling
Advanced Manufacturing of Dyclonine Hydrochloride via Novel Cu-Catalyzed C-O Cross-Coupling
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high purity with operational safety, particularly for established active pharmaceutical ingredients like local anesthetics. Patent CN113735799A introduces a transformative methodology for the synthesis of dyclonine hydrochloride, shifting away from hazardous acylation protocols toward a sophisticated two-step sequence involving a Mannich reaction and a copper-catalyzed C-O cross-coupling. This technical breakthrough addresses long-standing inefficiencies in prior art, such as the reliance on volatile acyl chlorides and the generation of excessive chemical waste, thereby offering a streamlined route that is inherently safer for large-scale operations. By leveraging specific ligand-catalyst systems, the inventors have achieved a level of process control that ensures consistent product quality while mitigating the environmental footprint typically associated with traditional etherification processes. For global procurement teams and R&D directors, this patent represents a significant opportunity to optimize supply chain resilience through a more sustainable and chemically elegant manufacturing strategy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of dyclonine hydrochloride has been plagued by significant technical hurdles that compromise both economic efficiency and workplace safety. Early synthetic routes, such as those disclosed in Indian patent IN172270A, relied on alkylation strategies that suffered from inherently low yields, often capping at approximately 75% for key intermediates like butoxyacetophenone, which drastically impacts the overall throughput of the manufacturing line. Furthermore, subsequent improvements attempted to utilize anhydrides or chlorinated acylating agents, but these methods introduced severe safety liabilities; for instance, the use of 3-chloropropionyl chloride presents a substantial risk due to its flammability and violent reactivity upon contact with moisture, necessitating expensive containment infrastructure and rigorous safety protocols. Additionally, many conventional processes require harsh purification steps, such as high-temperature distillation under reduced pressure, which not only consumes excessive energy but also degrades heat-sensitive intermediates, leading to complex impurity profiles that are difficult to remove. The reliance on two-phase reaction systems in some prior art further complicates matters by requiring phase transfer catalysts and creating emulsion issues during workup, ultimately increasing the volume of wastewater and organic solvents that must be treated before disposal.
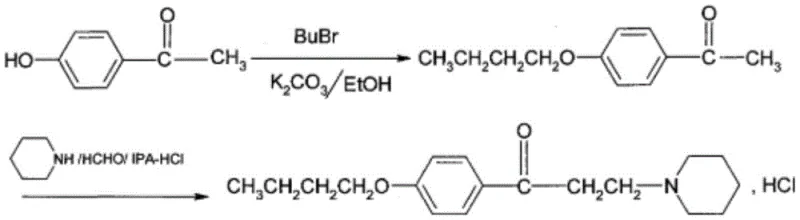
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in patent CN113735799A fundamentally reimagines the bond construction strategy by decoupling the carbon skeleton assembly from the ether linkage formation. Instead of attempting to install the butoxy group early in the synthesis using dangerous alkyl halides or acyl chlorides, this method first constructs the ketone-piperidine backbone via a highly efficient Mannich reaction using p-bromoacetophenone as a stable and commercially available starting material. This strategic pivot allows the critical C-O bond formation to occur in a dedicated second step under mild, catalytic conditions, completely eliminating the need for corrosive acetyl chloride or 3-chloropropionyl chloride reagents. The result is a process that operates at controllable temperatures with significantly reduced exothermic risks, facilitating a smoother transition from laboratory scale to multi-ton commercial production. By avoiding the generation of hydrochloric acid gas and other corrosive byproducts typical of acylation routes, this new methodology simplifies the engineering requirements for reactor materials and ventilation systems, directly translating to lower capital expenditure and operational overhead for manufacturing facilities.
Mechanistic Insights into Cu-Catalyzed C-O Cross-Coupling
The core innovation of this synthesis lies in the second step, where a copper-catalyzed C-O cross-coupling reaction is employed to link the piperidine-containing intermediate with n-butanol. This transformation is mediated by a specific catalytic system comprising cuprous iodide (CuI) as the metal source and trans-4-hydroxy-L-proline as a bidentate ligand, which plays a pivotal role in stabilizing the active copper species and facilitating the oxidative addition and reductive elimination cycles. The presence of the hydroxy-proline ligand enhances the solubility of the copper catalyst in the reaction medium and prevents the aggregation of metal particles, ensuring a homogeneous catalytic environment that maximizes turnover frequency. Mechanistically, the copper center activates the aryl bromide bond on the intermediate, allowing the nucleophilic attack by the butanol oxygen to proceed with high regioselectivity and minimal side reactions such as dehalogenation or homocoupling. This precise control over the reaction pathway is critical for maintaining the structural integrity of the sensitive piperidine ring and the ketone functionality, which might otherwise be compromised under the strongly basic or acidic conditions required by non-catalytic nucleophilic substitution methods.
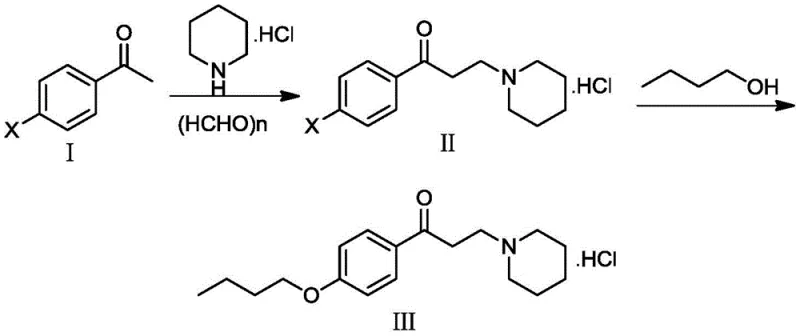
Furthermore, the impurity profile of the final product is rigorously controlled through the selectivity of this catalytic system, which minimizes the formation of over-alkylated byproducts or polymeric tars often seen in thermal etherification processes. The use of potassium carbonate as a base in conjunction with tetrabutylammonium bromide as a phase transfer agent creates an optimized ionic environment that promotes the deprotonation of the alcohol without inducing elimination reactions on the alkyl chain. Experimental data from the patent indicates that omitting the copper catalyst results in no product formation, while using alternative ligands like 1,10-phenanthroline without the optimal copper source leads to significantly diminished yields, underscoring the synergistic effect of the CuI/trans-4-hydroxy-L-proline pair. This mechanistic understanding allows process chemists to fine-tune reaction parameters such as temperature and stoichiometry to push the conversion to completion, achieving HPLC purities exceeding 99.9% after simple crystallization, which is a remarkable feat for a molecule containing multiple functional groups prone to side reactions.
How to Synthesize Dyclonine Hydrochloride Efficiently
The synthesis of dyclonine hydrochloride via this patented route involves a sequential two-step protocol that prioritizes safety and yield optimization at every stage. The process begins with the condensation of p-bromoacetophenone with piperidine hydrochloride and paraformaldehyde in an ethanol solvent, a classic Mannich reaction that proceeds smoothly at moderate temperatures to generate the key bromo-intermediate. Following isolation and purification of this intermediate, the material is subjected to the copper-catalyzed coupling with n-butanol under nitrogen protection to prevent oxidation of the catalyst, ensuring high reproducibility and batch-to-batch consistency. The detailed standardized synthetic steps, including specific molar ratios, temperature gradients, and workup procedures required to replicate these high-yield results, are outlined in the guide below.
- Perform Mannich reaction on p-bromoacetophenone with piperidine hydrochloride and paraformaldehyde in ethanol at 60°C.
- Conduct C-O crosslinking reaction on the intermediate with n-butanol using CuI catalyst and trans-4-hydroxy-L-proline ligand.
- Purify the final product through crystallization and vacuum drying to achieve high purity specifications.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain executives, the adoption of this novel synthesis route offers compelling economic and logistical benefits that extend far beyond simple yield improvements. By eliminating the requirement for high-toxicity reagents like acetyl chloride and 3-chloropropionyl chloride, manufacturers can significantly reduce the costs associated with hazardous material storage, specialized handling equipment, and regulatory compliance reporting. The removal of these dangerous chemicals from the supply chain also mitigates the risk of production stoppages due to supply shortages of controlled precursors, thereby enhancing the overall reliability and continuity of the manufacturing schedule. Moreover, the simplified workup procedure, which avoids complex distillations and extensive aqueous washes, leads to a drastic reduction in solvent consumption and waste treatment volumes, directly lowering the variable costs per kilogram of finished product. This leaner operational model allows suppliers to offer more competitive pricing structures while maintaining healthy margins, creating a win-win scenario for both the manufacturer and the downstream pharmaceutical client.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous acylating agents removes a major cost driver from the bill of materials, while the high efficiency of the copper-catalyzed step reduces the need for costly recycling of unreacted starting materials. The ability to achieve high purity directly from crystallization minimizes the number of purification cycles required, saving both energy and labor hours that would otherwise be spent on chromatography or repeated recrystallizations. Consequently, the overall cost of goods sold is optimized through a combination of cheaper raw material inputs and reduced processing intensity, making the final API intermediate more price-competitive in the global market.
- Enhanced Supply Chain Reliability: Starting materials such as p-bromoacetophenone and n-butanol are commodity chemicals with robust global supply networks, reducing the dependency on niche vendors who might face production bottlenecks. The stability of the intermediates involved in this route allows for flexible inventory management, enabling manufacturers to stockpile key precursors without the degradation risks associated with moisture-sensitive acyl chlorides. This inherent stability translates to shorter lead times for order fulfillment and greater agility in responding to sudden spikes in demand from the pharmaceutical sector, ensuring that critical medication supplies remain uninterrupted.
- Scalability and Environmental Compliance: The process generates significantly less hazardous waste compared to traditional methods, aligning perfectly with increasingly stringent environmental regulations and corporate sustainability goals. The absence of chlorinated byproducts simplifies wastewater treatment protocols, reducing the burden on effluent treatment plants and lowering the fees associated with industrial waste disposal. Furthermore, the mild reaction conditions and lack of exothermic runaway risks make the process inherently safer to scale up from pilot plant to full commercial production, minimizing the engineering challenges and capital investment typically required for process intensification.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis method for dyclonine hydrochloride. These answers are derived directly from the experimental data and technical disclosures within patent CN113735799A, providing a factual basis for evaluating the technology's viability. Understanding these details is crucial for stakeholders assessing the potential for technology transfer or licensing agreements.
Q: What are the safety advantages of the new synthesis route for dyclonine hydrochloride?
A: The novel method avoids highly toxic and reactive reagents such as acetyl chloride and 3-chloropropionyl chloride, significantly reducing storage hazards and improving operational safety for industrial mass production.
Q: How does the Cu-catalyzed system improve yield compared to traditional methods?
A: By utilizing cuprous iodide combined with trans-4-hydroxy-L-proline as a ligand, the reaction promotes forward progress effectively, achieving yields up to 81% in the coupling step and overall higher purity compared to older acylation routes.
Q: Is this synthesis method suitable for large-scale commercial manufacturing?
A: Yes, the process features controllable reaction temperatures, easily obtained starting materials, and reduced three-waste generation, making it highly favorable for scalable, environment-friendly industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dyclonine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic methodologies to ensure the highest standards of quality and safety in pharmaceutical manufacturing. Our team of expert process chemists has extensively evaluated the route described in patent CN113735799A and possesses the technical capability to implement this Cu-catalyzed strategy with precision and efficiency. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of high-purity intermediates that meet stringent purity specifications. Our state-of-the-art facilities are equipped with rigorous QC labs capable of performing comprehensive impurity profiling and stability testing, guaranteeing that every batch of dyclonine hydrochloride we produce adheres to the most demanding international pharmacopeial standards.
We invite global partners to collaborate with us to leverage this superior manufacturing technology for their supply chains. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this greener, more efficient route. We encourage you to reach out today to obtain specific COA data and route feasibility assessments tailored to your specific volume requirements, ensuring a seamless integration of our high-quality intermediates into your final drug product formulation.
