Scalable Synthesis of 1-(7-Halobenzo[D][1,3]Dioxol-4-Yl)Alkyl Ketones via Optimized Fries Rearrangement
Scalable Synthesis of 1-(7-Halobenzo[D][1,3]Dioxol-4-Yl)Alkyl Ketones via Optimized Fries Rearrangement
The pharmaceutical industry's relentless pursuit of efficient antiviral therapies has placed significant emphasis on the robust synthesis of key intermediates for Hepatitis C treatments. Patent CN114773309A introduces a transformative preparation method for 1-(7-halobenzo[D][1,3]dioxol-4-yl)alkyl ketone compounds, which serve as critical building blocks for drugs like Coblopasvir. This technology addresses longstanding challenges in the field by replacing hazardous, multi-step cryogenic processes with a streamlined, Lewis acid-catalyzed route. The core innovation lies in the strategic application of a Fries rearrangement to construct the ketone functionality directly on the aromatic ring, followed by a facile etherification to close the dioxole ring. This approach not only enhances the chemical efficiency but also aligns with modern green chemistry principles by reducing waste and energy consumption. For global supply chains, this represents a pivotal shift towards more reliable and cost-effective manufacturing of high-value pharmaceutical intermediates.
![Chemical structure of 1-(7-bromobenzo[d][1,3]dioxol-4-yl)ethanone, the target antiviral intermediate](/insights/img/1-7-halobenzo-dioxol-alkyl-ketone-fries-rearrangement-pharma-supplier-20260308170317-01.png)
The target molecule, specifically exemplified as 1-(7-bromobenzo[D][1,3]dioxol-4-yl)ethan-1-one, is a vital precursor in the assembly of direct-acting antiviral agents. As illustrated in the structural representation, the molecule features a halogenated benzo[d][1,3]dioxole core substituted with an acetyl group. The precise positioning of the halogen and the ketone moiety is crucial for subsequent coupling reactions in the drug synthesis pipeline. Traditional methods to access this scaffold often involve cumbersome protection-deprotection sequences or harsh metalation steps that compromise atom economy. The patented methodology circumvents these issues by starting from readily available 3-halogenated catechols. This strategic choice of starting material simplifies the supply chain logistics, as 3-halogenated catechols are commodity chemicals with stable pricing and abundant availability, unlike specialized organometallic reagents that require stringent storage and handling protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art synthetic routes for generating 1-(7-halobenzo[D][1,3]dioxol-4-yl)alkyl ketones are plagued by significant operational inefficiencies and safety hazards that hinder large-scale adoption. Conventional strategies typically rely on a sequence involving aldol condensation, followed by lithiation at cryogenic temperatures (often below -70°C), and subsequent Grignard additions or oxidations. These low-temperature requirements impose a massive energy burden on manufacturing facilities, necessitating specialized cooling infrastructure that drastically increases capital expenditure and operational costs. Furthermore, the use of n-butyllithium and other pyrophoric reagents introduces severe safety risks, requiring inert atmosphere handling and specialized training for operators. The multi-step nature of these legacy routes, often exceeding four distinct chemical transformations, inherently leads to cumulative yield losses and increased generation of solvent waste. Each additional step introduces potential points of failure, complicating quality control and extending the overall production lead time, which is detrimental in the fast-paced environment of generic drug development.
The Novel Approach
In stark contrast, the novel approach detailed in the patent leverages a concise three-step sequence that maximizes atom economy and operational simplicity. The process initiates with a straightforward acetylation of 3-halogenated catechol to form a diacetate intermediate, as depicted in the structural formula below. This step proceeds under mild alkaline conditions using inexpensive acetic anhydride, avoiding the need for exotic reagents. The cornerstone of this innovation is the subsequent Fries rearrangement, catalyzed by accessible Lewis acids such as boron trifluoride etherate or aluminum trichloride. This rearrangement effectively migrates the acyl group to the desired ortho-position relative to the phenolic hydroxyl, setting up the correct substitution pattern for the final ring closure. By eliminating the cryogenic lithiation step entirely, the new route operates at much higher and more manageable temperatures (50-130°C), significantly lowering the barrier to entry for contract manufacturing organizations (CMOs) and enhancing the overall safety profile of the synthesis.
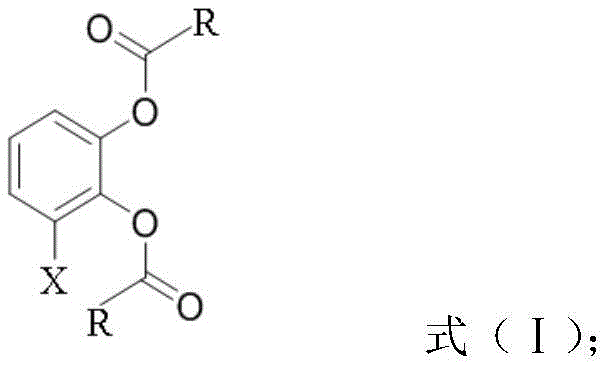
Mechanistic Insights into Lewis Acid-Catalyzed Fries Rearrangement
The success of this synthetic strategy hinges on the precise control of the Fries rearrangement, a reaction known for its sensitivity to reaction conditions and catalyst choice. In this specific application, the acetylated intermediate undergoes a Lewis acid-mediated cleavage of the ester bond, generating an acylium ion equivalent that attacks the aromatic ring. The patent specifies the use of Lewis acids like boron trifluoride etherate or titanium tetrachloride, which coordinate with the carbonyl oxygen to facilitate this migration. The regioselectivity is paramount; the reaction must favor the formation of the 4-acyl-3-halo-catechol derivative over other potential isomers. The patent data indicates that maintaining the reaction temperature between 100°C and 120°C is critical for optimizing this selectivity. At lower temperatures, the reaction kinetics may be too sluggish, leading to incomplete conversion, while excessively high temperatures could promote polymerization or degradation of the sensitive catechol backbone. The careful tuning of the molar ratio between the substrate and the Lewis acid (preferably 1:0.8 to 1:1.5) ensures that the catalytic cycle proceeds efficiently without generating excessive acidic waste that would complicate downstream workup.
Following the rearrangement, the resulting hydroxy-ketone intermediate, shown in the formula below, possesses the necessary functionality for the final cyclization. The presence of the free phenolic hydroxyl groups adjacent to the ketone allows for a rapid and high-yielding etherification with dihalomethane. This step effectively locks the structure into the rigid benzo[d][1,3]dioxole framework. The mechanistic advantage here is the intramolecular nature of the final ring closure, which is entropically favored compared to intermolecular reactions. The base-promoted etherification, typically using potassium carbonate in polar aprotic solvents like DMF, ensures that the phenoxide nucleophile is generated in situ to attack the dihalomethane electrophile. This sequence avoids the need for protecting groups on the phenols, which would otherwise add two extra steps (protection and deprotection) to the synthesis. The result is a highly convergent route where the complexity of the molecule is built up rapidly from simple precursors, minimizing the time the material spends in the reactor and reducing the opportunity for side reactions to occur.
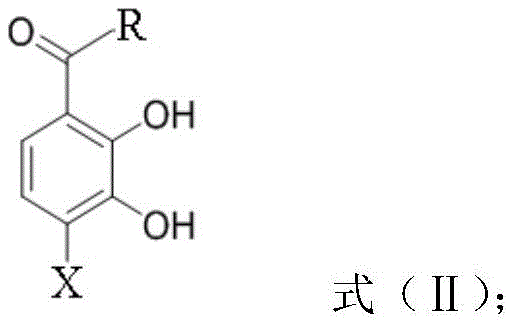
How to Synthesize 1-(7-Halobenzo[D][1,3]Dioxol-4-Yl)Alkyl Ketones Efficiently
The implementation of this synthesis requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and high purity. The process begins with the acetylation of 3-halogenated catechol in a solvent system such as dichloromethane or acetonitrile, using a slight excess of acetic anhydride to drive the reaction to completion. Once the diacetate is isolated, it is subjected to the Fries rearrangement in a high-boiling solvent like chlorobenzene or o-dichlorobenzene, or even under solvent-free conditions if heat transfer is managed correctly. The final etherification is conducted in DMF or acetone with a base, followed by a standard aqueous workup and recrystallization to achieve pharmaceutical-grade purity.
- Perform acetylation of 3-halogenated catechol using acetic anhydride in an organic solvent at 40-50°C to form the diacetate intermediate.
- Execute a selective Fries rearrangement on the acetylated product using a Lewis acid catalyst at 100-120°C to generate the hydroxy-ketone rearrangement product.
- Conduct an etherification reaction between the rearrangement product and dihalomethane under basic conditions at 70-80°C to cyclize and form the final benzo[d][1,3]dioxole ring.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this patented synthesis route offers profound strategic benefits that extend beyond simple chemical yield improvements. The elimination of cryogenic processing steps fundamentally alters the cost structure of the intermediate. By removing the requirement for specialized low-temperature reactors and the continuous consumption of cryogenic coolants, the manufacturing facility can operate with significantly reduced utility costs. Furthermore, the replacement of hazardous organolithium reagents with stable Lewis acids and acetic anhydride mitigates the regulatory and safety compliance burdens associated with handling pyrophoric materials. This reduction in hazard classification simplifies logistics, allowing for easier transportation of raw materials and safer storage within the plant, thereby reducing insurance premiums and safety overheads. The streamlined three-step process also shortens the overall cycle time, enabling faster turnover of production batches and improved responsiveness to market demand fluctuations.
- Cost Reduction in Manufacturing: The economic impact of this route is driven by the drastic simplification of the operational workflow. By consolidating the synthesis into fewer steps with higher individual yields, the total amount of solvent and reagent consumption per kilogram of product is substantially lowered. The use of commodity chemicals like acetic anhydride and dihalomethane, rather than custom-synthesized organometallics, ensures that raw material costs remain stable and predictable. Additionally, the higher selectivity of the Fries rearrangement reduces the formation of difficult-to-remove impurities, which in turn lowers the cost of purification and increases the recovery of valuable product mass. This cumulative effect results in a markedly lower cost of goods sold (COGS), providing a competitive edge in price-sensitive generic drug markets.
- Enhanced Supply Chain Reliability: Supply continuity is often jeopardized by reliance on niche reagents with limited supplier bases. This new method utilizes 3-halogenated catechols and common Lewis acids, which are produced by multiple global vendors, thereby diversifying the supply risk. The robustness of the reaction conditions means that the process is less susceptible to minor variations in environmental conditions or raw material quality, leading to more consistent batch-to-batch performance. This reliability is crucial for long-term supply agreements with major pharmaceutical companies, where any disruption can have cascading effects on clinical trial timelines or commercial launch schedules. The ability to scale this process from pilot plant to multi-ton production without fundamental changes to the chemistry further secures the supply chain against future capacity constraints.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route represents a significant improvement over legacy methods. The avoidance of heavy metal oxidants (like chromium trioxide used in some prior art) and pyrophoric reagents reduces the toxicity profile of the waste stream. The solvents employed, such as toluene and dichloromethane, are well-understood and can be efficiently recovered and recycled using standard distillation infrastructure. The higher atom economy of the Fries rearrangement means less organic waste is generated per unit of product, aligning with increasingly stringent global environmental regulations. This 'greener' profile not only facilitates easier permitting for new manufacturing lines but also enhances the corporate sustainability metrics of the producing entity, a factor that is becoming increasingly important in vendor selection criteria for top-tier pharmaceutical clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, highlighting the practical advantages of the Fries rearrangement approach. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their specific manufacturing needs.
Q: What are the primary advantages of this Fries rearrangement route over conventional lithiation methods?
A: The patented Fries rearrangement route eliminates the need for cryogenic conditions (-72°C) and hazardous organolithium reagents required in conventional methods. This shift significantly reduces energy consumption for cooling and lowers safety risks associated with handling pyrophoric materials, while simultaneously improving reaction selectivity and overall yield.
Q: How does the new process impact the purity profile of the Hepatitis C intermediate?
A: By utilizing a highly selective Lewis acid-catalyzed rearrangement followed by a controlled etherification, the process minimizes the formation of regioisomers and polymeric byproducts common in Grignard additions. The result is a crude product with inherently higher purity, reducing the burden on downstream purification steps like column chromatography.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is specifically designed for industrial scalability. It employs robust reaction conditions (0-130°C range) and commercially available starting materials like 3-halogenated catechol and acetic anhydride. The avoidance of sensitive low-temperature steps makes the process easier to manage in large reactors, ensuring consistent supply continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-(7-Halobenzo[D][1,3]Dioxol-4-Yl)Alkyl Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of antiviral therapies depends on the availability of high-quality, cost-effective intermediates. Our technical team has thoroughly analyzed the pathway described in CN114773309A and is fully prepared to execute this synthesis at commercial scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. Our state-of-the-art facilities are equipped to handle the specific thermal requirements of the Fries rearrangement and the solvent management needs of the etherification step. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 1-(7-halobenzo[D][1,3]dioxol-4-yl)alkyl ketone meets the exacting standards required for API synthesis. Our commitment to quality ensures that your downstream processes proceed without interruption due to impurity-related failures.
We invite you to collaborate with us to leverage this advanced synthetic technology for your Hepatitis C drug programs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage you to contact our technical procurement team today to request specific COA data from our recent pilot runs and to discuss detailed route feasibility assessments. Let us help you optimize your supply chain and reduce your time-to-market with our reliable, scalable, and economically superior manufacturing solutions.
