Revolutionizing Sulfoxide Ylide Production: A Green Electrocatalytic Strategy for Commercial Scale-Up
Revolutionizing Sulfoxide Ylide Production: A Green Electrocatalytic Strategy for Commercial Scale-Up
The landscape of fine chemical synthesis is undergoing a paradigm shift towards greener, more sustainable methodologies, driven by the urgent need to reduce environmental footprints while maintaining high efficiency. A groundbreaking development in this arena is detailed in patent CN114411181A, which discloses a novel method for synthesizing alpha-carbonyl-alpha'-chlorosulfoxide ylides under electrocatalytic conditions. These specialized organosulfur compounds serve as critical building blocks in the construction of complex molecular architectures, finding extensive applications in the total synthesis of bioactive natural products, pharmaceutical agents, and advanced functional materials. The significance of this invention lies not merely in the creation of a new compound class, but in the establishment of a robust, environmentally benign pathway that circumvents the limitations of traditional stoichiometric oxidation and halogenation protocols. By leveraging electricity as a traceless reagent, this technology offers a compelling value proposition for R&D directors seeking cleaner routes and procurement managers aiming for cost-effective supply chains.
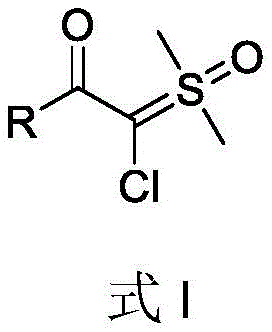
The structural versatility of these ylides is paramount for their utility in downstream applications. As illustrated in the general formula, the core scaffold accommodates a diverse array of substituents at the R position, ranging from simple phenyl rings to complex heterocycles and aliphatic systems. This adaptability allows chemists to fine-tune the electronic and steric properties of the final molecule, making them indispensable precursors for cyclopropanation reactions and other carbene-transfer processes that are foundational in modern medicinal chemistry. The ability to access these structures efficiently opens new doors for the rapid assembly of drug candidates and agrochemical intermediates, positioning this synthetic method as a key enabling technology for the next generation of high-value specialty chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha-halo carbonyl compounds and related ylides has relied heavily on the use of harsh chemical oxidants and chlorinating agents such as sulfuryl chloride, N-chlorosuccinimide (NCS), or molecular chlorine gas. These traditional reagents present significant challenges from both a safety and an economic perspective. The handling of toxic gases and corrosive liquids requires specialized infrastructure and rigorous safety protocols, which inherently drives up capital expenditure and operational costs for manufacturing facilities. Furthermore, stoichiometric amounts of these reagents often generate substantial quantities of hazardous waste, necessitating complex and expensive disposal procedures to meet increasingly stringent environmental regulations. From a purity standpoint, side reactions induced by aggressive chlorinating conditions can lead to the formation of difficult-to-remove impurities, complicating the purification process and reducing the overall yield of the desired high-purity intermediate.
The Novel Approach
In stark contrast to these legacy methods, the electrocatalytic strategy outlined in the patent introduces a transformative approach that utilizes electrical energy to drive the chlorination process. This method employs magnesium chloride, a cheap, abundant, and non-toxic salt, as the sole source of chlorine, thereby eliminating the need for dangerous molecular chlorine or expensive organic chlorinating agents. The reaction proceeds under remarkably mild conditions, typically at room temperature (20°C), which minimizes thermal degradation of sensitive substrates and reduces energy consumption associated with heating or cooling large reactors. The use of a binary solvent system comprising acetonitrile and water further enhances the green credentials of the process by reducing the reliance on purely organic solvents. This innovative workflow not only simplifies the operational complexity but also aligns perfectly with the principles of green chemistry, offering a sustainable alternative that does not compromise on reaction efficiency or product quality.
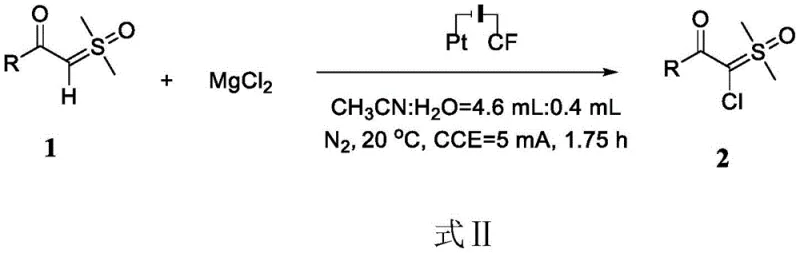
Mechanistic Insights into Electrocatalytic Chlorination
The success of this synthesis hinges on the precise control of the electrochemical environment within the reaction cell. The process utilizes a constant current electrolysis setup where a graphite felt serves as the anode and a platinum sheet acts as the cathode. At the anode surface, chloride ions derived from the dissolved magnesium chloride undergo oxidation to generate reactive chlorine species in situ. These electro-generated species then selectively attack the alpha-position of the carbonyl group in the sulfoxide ylide substrate. The choice of electrode materials is critical; graphite felt provides a high surface area for efficient electron transfer, while the platinum cathode ensures stable reduction potentials without introducing contaminating metal ions into the reaction mixture. This heterogeneous electrocatalytic system ensures that the reaction kinetics are optimized for the specific transformation, allowing for high conversion rates within a relatively short timeframe of approximately 1.75 hours.
Furthermore, the mechanism inherently supports superior impurity control compared to thermal radical chlorination. Because the reactive chlorine species are generated continuously at low concentrations at the electrode interface, the likelihood of over-chlorination or non-selective radical attacks on other parts of the molecule is significantly diminished. This controlled generation of reagents leads to a cleaner reaction profile, which is evidenced by the moderate to good yields observed across a wide range of substrates, including those with electron-rich and electron-deficient aromatic rings. The absence of transition metal catalysts also means that the final product is free from heavy metal residues, a critical quality attribute for pharmaceutical intermediates where strict limits on metal impurities are enforced by regulatory bodies. This intrinsic purity advantage simplifies the downstream purification steps, often requiring only standard silica gel chromatography to achieve analytical grade purity.
How to Synthesize Alpha-Carbonyl-Alpha'-Chlorosulfoxide Ylide Efficiently
Implementing this electrocatalytic protocol in a laboratory or pilot plant setting requires adherence to specific operational parameters to ensure reproducibility and safety. The standard procedure involves dissolving the alpha-carbonyl sulfoxide ylide substrate and magnesium chloride in the optimized acetonitrile-water solvent mixture. The molar ratio of substrate to salt is maintained at 1:2 to ensure an excess of chloride ions for efficient turnover. The reaction vessel is purged with nitrogen to exclude oxygen, which could interfere with the electrochemical process or lead to oxidative side products. Once the electrolysis is complete, the workup is straightforward: the mixture is dried, filtered to remove salts, and concentrated. For detailed operational specifics and safety guidelines regarding the setup of the electrolytic cell and optimization of current density, please refer to the standardized synthesis guide below.
- Prepare the reaction mixture by adding alpha-carbonyl sulfoxide ylide and magnesium chloride in a 1: 2 molar ratio to a reactor containing an acetonitrile-water solvent system (4.6 mL:0.4 mL).
- Set up the electrolytic cell using graphite felt as the anode and a platinum sheet as the cathode, then apply a constant current of 5 mA at 20°C for 1.75 hours under nitrogen atmosphere.
- Upon completion, dry the mixture with anhydrous magnesium sulfate, filter, concentrate via rotary evaporation, and purify the crude product using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this electrocatalytic technology translates into tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the raw material supply chain. By replacing specialized, hazardous chlorinating agents with commodity-grade magnesium chloride, manufacturers can mitigate supply risks associated with volatile chemical markets and reduce storage costs linked to dangerous goods. The elimination of toxic reagents also lowers the barrier for entry for contract manufacturing organizations (CMOs) that may lack the specialized infrastructure required to handle corrosive gases, thereby expanding the pool of potential suppliers and fostering a more competitive pricing environment. This shift towards benign reagents fundamentally alters the cost structure of production, making the manufacturing of these high-value intermediates more economically viable.
- Cost Reduction in Manufacturing: The economic impact of this method is profound due to the removal of expensive catalysts and oxidants. Traditional methods often rely on precious metal catalysts or stoichiometric amounts of costly organic reagents, which constitute a significant portion of the bill of materials. By substituting these with electricity and common salt, the direct material costs are significantly reduced. Additionally, the mild reaction conditions (20°C) eliminate the need for energy-intensive heating or cryogenic cooling systems, leading to lower utility bills. The simplified workup procedure, which avoids complex quenching steps and extensive washing required to remove toxic byproducts, further reduces labor costs and solvent consumption, resulting in a leaner, more cost-efficient manufacturing process.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of universally available reagents. Magnesium chloride and acetonitrile are bulk commodities with stable global supply chains, unlike specialized chlorinating agents that may be subject to production bottlenecks or regulatory restrictions. The robustness of the electrocatalytic method against variations in substrate structure means that a single production line can be easily adapted to manufacture a wide variety of derivatives without extensive retooling. This flexibility allows suppliers to respond rapidly to changing market demands and custom orders, ensuring consistent delivery schedules even in times of market volatility. The reduced hazard profile also simplifies logistics and transportation, as the reagents do not require special hazardous material handling, speeding up the inbound and outbound logistics processes.
- Scalability and Environmental Compliance: Scaling electrochemical processes is increasingly feasible with modern flow chemistry and large-scale electrolyzer technologies. The linear relationship between current and reaction rate allows for precise control over production throughput, facilitating a smooth transition from gram-scale R&D to tonne-scale commercial production. From an environmental compliance perspective, this method is a game-changer. The absence of heavy metal waste and toxic halogenated byproducts significantly reduces the burden on wastewater treatment facilities and lowers the cost of waste disposal. This alignment with green chemistry principles not only future-proofs the manufacturing process against tightening environmental regulations but also enhances the brand reputation of companies adopting sustainable practices, appealing to eco-conscious partners and end-users in the pharmaceutical and agrochemical sectors.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of integrating this technology into their existing workflows, we have compiled answers to common inquiries regarding the electrocatalytic synthesis of alpha-carbonyl-alpha'-chlorosulfoxide ylides. These responses are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance for process development scientists. Understanding these nuances is crucial for assessing the compatibility of this method with specific project requirements and for planning the necessary resource allocation for pilot studies.
Q: What are the advantages of using electrocatalysis over traditional chlorinating agents?
A: Electrocatalysis eliminates the need for hazardous external oxidants and expensive transition metal catalysts. It operates under mild conditions (20°C) and uses benign magnesium chloride as the chlorine source, significantly reducing waste generation and safety risks associated with toxic reagents.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the method demonstrates excellent scalability potential. The use of simple electrode materials like graphite felt and platinum, combined with a straightforward workup procedure involving filtration and column chromatography, facilitates the transition from laboratory benchtop to commercial manufacturing scales.
Q: What is the substrate scope for this electrocatalytic chlorination?
A: The protocol exhibits broad functional group tolerance. It successfully converts various alpha-carbonyl sulfoxide ylides bearing electron-donating groups (like methoxy and methyl), electron-withdrawing groups (like chloro), and heterocyclic moieties (furan, thiophene) into the corresponding chlorinated products with moderate to good yields.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Carbonyl-Alpha'-Chlorosulfoxide Ylide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrocatalytic synthesis in the realm of fine chemical manufacturing. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries like the one described in CN114411181A can be seamlessly translated into industrial reality. Our state-of-the-art facilities are equipped with advanced electrochemical reactors and rigorous QC labs capable of meeting stringent purity specifications required by global pharmaceutical standards. We are committed to delivering high-quality intermediates that empower our clients to accelerate their drug discovery and development pipelines with confidence.
We invite you to explore how our expertise in green synthesis can optimize your supply chain. Whether you require custom synthesis of complex sulfoxide ylides or a comprehensive Customized Cost-Saving Analysis for your existing routes, our technical team is ready to assist. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your project needs. Let us collaborate to build a more sustainable and efficient future for chemical manufacturing.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
