Scalable Biocatalytic Production of Novel Amino-Succinic Anhydride Derivatives for Pharmaceutical Intermediates
The landscape of fine chemical manufacturing is undergoing a paradigm shift with the emergence of advanced biocatalytic pathways, as exemplified by the groundbreaking technology disclosed in patent CN107903227B. This intellectual property introduces a novel method for synthesizing amino-containing succinic anhydride derivatives, a class of compounds that serves as indispensable building blocks in the pharmaceutical and polymer industries. Unlike traditional petrochemical routes that often rely on hazardous reagents and extreme conditions, this innovation leverages the metabolic machinery of heterologous organisms, specifically utilizing a synergistic enzyme system derived from thermophilic fungi. For R&D directors and procurement strategists alike, this represents a significant opportunity to secure a reliable succinic anhydride derivative supplier capable of delivering high-purity intermediates through a sustainable, green chemistry approach. The patent details the successful co-expression of Type I PKS/NRPS enzymes and ER enzymes within a yeast host, resulting in the efficient biosynthesis of a specific molecular scaffold that was previously difficult to access with high selectivity. This technological leap not only addresses the growing demand for chiral intermediates but also aligns perfectly with global initiatives to reduce the carbon footprint of chemical manufacturing processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for producing succinic anhydride derivatives frequently encounter substantial bottlenecks related to reaction specificity and environmental impact. Conventional organic synthesis often necessitates the use of strong dehydrating agents, toxic solvents, and heavy metal catalysts to drive the cyclization and functionalization reactions required to form the anhydride ring. These harsh conditions can lead to the formation of numerous by-products and isomers, complicating the downstream purification process and significantly lowering the overall yield of the desired active pharmaceutical ingredient intermediate. Furthermore, the reliance on non-renewable feedstocks and the generation of hazardous waste streams pose increasing regulatory and cost challenges for manufacturers aiming to maintain compliance with stringent environmental standards. The lack of inherent stereocontrol in many chemical methods also means that additional resolution steps are often required, further inflating production costs and extending lead times for high-purity pharmaceutical intermediates. Consequently, the industry has long sought a more elegant solution that can bypass these inefficiencies while maintaining the structural integrity of the sensitive anhydride moiety.
The Novel Approach
In stark contrast to these legacy methods, the biocatalytic strategy outlined in the patent utilizes a highly specific enzymatic cascade to construct the target molecule with precision. By engineering Saccharomyces cerevisiae to express specific genes from Thermomyces dupontii, the process achieves the direct biosynthesis of the amino-succinic anhydride derivative under mild, aqueous conditions. This biological factory approach inherently avoids the use of toxic reagents and operates at ambient temperatures and pressures, drastically simplifying the safety requirements for the production facility. The core of this innovation is the ability of the Type I PKS/NRPS enzyme system to assemble the carbon skeleton, while the ER enzyme facilitates the necessary reduction steps, ensuring the correct oxidation state and functionality are achieved in a single fermentation pot. 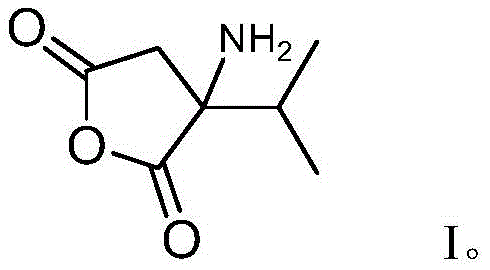 As illustrated in the structure above, the resulting compound possesses a unique arrangement of functional groups that makes it an ideal precursor for complex drug molecules. This novel approach not only enhances the purity profile of the final product but also opens new avenues for cost reduction in pharmaceutical intermediates manufacturing by streamlining the entire value chain from raw materials to finished goods.
As illustrated in the structure above, the resulting compound possesses a unique arrangement of functional groups that makes it an ideal precursor for complex drug molecules. This novel approach not only enhances the purity profile of the final product but also opens new avenues for cost reduction in pharmaceutical intermediates manufacturing by streamlining the entire value chain from raw materials to finished goods.
Mechanistic Insights into PKS/NRPS and ER Enzyme Synergistic Catalysis
The mechanistic foundation of this technology rests on the precise orchestration of two distinct enzymatic activities within a single microbial host. The Type I PKS/NRPS (Polyketide Synthase/Non-Ribosomal Peptide Synthetase) enzyme acts as a molecular assembly line, recruiting specific substrates and catalyzing the formation of carbon-carbon and carbon-nitrogen bonds to build the core framework of the succinic anhydride derivative. This mega-enzyme complex is encoded by a specific gene sequence (SEQ ID NO. 1) derived from thermophilic genera, which confers stability and activity even under varying fermentation conditions. Simultaneously, the ER (Enoyl Reductase) enzyme, encoded by SEQ ID NO. 2, plays a critical role in modulating the saturation level of the intermediate chains, ensuring that the final product retains the necessary chemical reactivity for downstream derivatization. The co-expression of these genes in a heterologous organism like yeast allows for the compartmentalization of these reactions, effectively isolating the biosynthetic pathway from competing metabolic processes that could otherwise degrade the yield. This dual-enzyme system creates a highly controlled micro-environment where the substrate is channelled efficiently from one active site to the next, minimizing the accumulation of incomplete intermediates and maximizing the flux towards the desired end product.
From an impurity control perspective, this enzymatic specificity offers a distinct advantage over random chemical transformations. Because enzymes are chiral catalysts, they naturally produce a single enantiomer or diastereomer, thereby eliminating the racemic mixtures that plague traditional synthesis. This intrinsic selectivity means that the crude fermentation broth already possesses a high degree of chemical purity, reducing the burden on subsequent purification steps. The patent describes a rigorous downstream processing protocol involving acetone extraction and multi-stage column chromatography (Sephadex LH-20 and RP18) to isolate the orange powdery compound. The use of TLC detection with specific Rf values (0.35-0.45) ensures that only fractions containing the pure target molecule are pooled, effectively filtering out any residual cellular debris or minor metabolic by-products. This level of control is crucial for meeting the stringent quality specifications required for API intermediates, where even trace impurities can affect the safety and efficacy of the final drug product.
How to Synthesize Amino-Succinic Anhydride Derivative Efficiently
The practical implementation of this biocatalytic route involves a series of well-defined genetic engineering and fermentation steps that can be scaled from laboratory benchtop to industrial bioreactors. The process begins with the construction of robust expression vectors, where the genes encoding the Type I PKS/NRPS and ER enzymes are ligated into compatible plasmids such as pGADT7 and pGBKT7. These vectors are then co-transformed into a specialized yeast strain, creating an engineered cell factory capable of producing the target compound continuously during the growth phase. Following fermentation, the recovery of the product relies on a solvent extraction strategy optimized for the polarity of the succinic anhydride derivative, followed by sophisticated chromatographic techniques to achieve pharmaceutical-grade purity. This standardized workflow ensures reproducibility and consistency, which are paramount for maintaining supply chain reliability in the competitive fine chemicals market.
- Construct expression vectors by linking Type I PKS/NRPS and ER enzyme genes from Thermomyces dupontii to pGADT7 and pGBKT7 plasmids respectively.
- Co-transform the engineered plasmids into Saccharomyces cerevisiae FY834 strain and culture in amino acid-deficient medium to obtain fermentation broth.
- Extract the target compound using acetone-water mixtures followed by ultrasonic treatment, and purify via Sephadex LH-20 and RP18 column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this biocatalytic technology translates into tangible strategic benefits that extend far beyond simple unit cost metrics. The shift from chemical synthesis to fermentation-based production fundamentally alters the risk profile of the supply chain by reducing dependence on volatile petrochemical feedstocks and hazardous reagent markets. This transition enables a more resilient sourcing strategy, as the raw materials for fermentation (such as glucose and nitrogen sources) are generally abundant, renewable, and subject to less price fluctuation than specialized organic starting materials. Furthermore, the simplified reaction conditions eliminate the need for expensive corrosion-resistant equipment and complex waste treatment facilities associated with harsh chemical processes, leading to substantial capital expenditure savings for manufacturing partners. The inherent safety of the biological process also reduces insurance premiums and regulatory compliance costs, contributing to a lower total cost of ownership for the final intermediate.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and toxic dehydrating agents removes the necessity for costly metal scavenging steps and extensive solvent recovery systems, directly lowering operational expenses. By utilizing renewable biological feedstocks instead of petroleum-derived precursors, manufacturers can insulate themselves from fossil fuel price volatility, ensuring more stable long-term pricing for their customers. Additionally, the high selectivity of the enzymatic process reduces the volume of waste generated per kilogram of product, significantly cutting disposal costs and environmental levies. This streamlined production model allows for a more competitive pricing structure without compromising on the quality or purity of the succinic anhydride derivative supplied to the market.
- Enhanced Supply Chain Reliability: Fermentation processes are highly scalable, allowing production capacity to be ramped up rapidly by increasing bioreactor volume rather than building entirely new chemical synthesis lines. This flexibility ensures that sudden spikes in demand for pharmaceutical intermediates can be met without the long lead times typically associated with expanding chemical infrastructure. The use of robust yeast strains that can be stored and propagated easily guarantees a continuous supply of the biological catalyst, mitigating the risk of production stoppages due to reagent shortages. Moreover, the decentralized nature of biomanufacturing allows for production to be located closer to key markets, reducing logistics costs and improving delivery speed for global clients seeking a reliable agrochemical intermediate supplier or pharma partner.
- Scalability and Environmental Compliance: The aqueous nature of the fermentation broth and the use of bio-based solvents like acetone align perfectly with green chemistry principles, facilitating easier permitting and regulatory approval in jurisdictions with strict environmental laws. The process generates significantly less hazardous waste compared to traditional organic synthesis, simplifying the effluent treatment process and reducing the environmental footprint of the manufacturing site. This sustainability profile is increasingly becoming a prerequisite for inclusion in the supply chains of major multinational corporations, providing a distinct competitive advantage for suppliers who can demonstrate verifiable eco-friendly production methods. The ability to scale this process from grams to tons while maintaining consistent quality makes it an ideal solution for the commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this novel succinic anhydride compound. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the technology's capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this intermediate into their existing drug development pipelines. We encourage potential partners to review these points carefully to appreciate the full scope of value this biocatalytic platform offers.
Q: What represents the primary advantage of this biocatalytic method over traditional chemical synthesis?
A: The primary advantage lies in the exceptional regioselectivity and stereoselectivity provided by the Type I PKS/NRPS and ER enzyme system, which eliminates the need for harsh chemical reagents and complex protection-deprotection steps typically required in traditional organic synthesis of succinic anhydride derivatives.
Q: Which downstream pharmaceutical products can utilize this novel succinic anhydride compound?
A: This compound serves as a critical precursor for the manufacture of high-value active pharmaceutical ingredients including N-hydroxysuccinimide, erythromycin ethyl succinate, fenbufen, artesunate, and oxaprozin, making it highly versatile for drug discovery pipelines.
Q: How does the fermentation process ensure high purity of the final product?
A: The process utilizes specific gene sequences from Thermomyces dupontii expressed in a controlled yeast host, which minimizes side reactions. Subsequent purification through gel column chromatography and medium-pressure RP18 ensures the removal of biological impurities, yielding a high-purity orange powdery compound suitable for sensitive applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Succinic Anhydride Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the biocatalytic synthesis route described in patent CN107903227B and are fully equipped to bring this technology to commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of succinic anhydride derivative meets the exacting standards required by the global pharmaceutical industry. Our commitment to quality is backed by state-of-the-art fermentation facilities and downstream processing units capable of handling the specific extraction and chromatography needs of this unique molecule.
We invite you to engage with our technical procurement team to discuss how this innovative intermediate can optimize your synthesis routes and reduce your overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain a detailed projection of the economic benefits specific to your application. We are ready to provide specific COA data and route feasibility assessments to support your R&D efforts, ensuring that you have all the necessary information to make informed sourcing decisions. Partner with us to secure a sustainable, high-quality supply of this critical building block for your next generation of therapeutic agents.
