Advanced Rhodium Catalysis for Efficient 3-Substituted Pyridazine Derivative Manufacturing
The pharmaceutical industry continuously seeks robust methodologies to access structurally diverse heterocyclic scaffolds, particularly those capable of serving as privileged structures in drug discovery. Patent CN115557900A introduces a groundbreaking synthetic strategy for the preparation of 3-substituted pyridazine derivatives, specifically targeting the challenging construction of quaternary carbon centers at the C3 position. This innovation addresses a significant gap in current medicinal chemistry toolkits, where traditional methods often fail to introduce bulky substituents with high regioselectivity. By leveraging a rhodium-catalyzed insertion reaction between pyridazin-3-one derivatives and aryl diazo compounds, this technology enables the efficient assembly of complex molecular architectures under remarkably mild conditions. For R&D directors and procurement specialists alike, this represents a pivotal advancement in the synthesis of high-purity pharmaceutical intermediates, offering a pathway to novel bioactive molecules that were previously difficult or impossible to access through conventional retrosynthetic disconnections.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of substituted pyridazine derivatives has relied heavily on [4+2] cyclization reactions or aza-Diels-Alder protocols. While these methods have achieved considerable success in generating simple substituted variants, they suffer from inherent limitations when attempting to construct sterically congested quaternary centers at the 3-position. The electronic and steric environment of the pyridazine ring often dictates poor regioselectivity, leading to mixtures of isomers that are costly and time-consuming to separate. Furthermore, direct functionalization of the heterocyclic ring is fraught with challenges due to the presence of multiple nitrogen atoms which can coordinate with catalysts or deactivate reactive sites. As demonstrated in comparative studies within the patent data, attempting to react unprotected pyridazin-3-ones directly results in chaotic reaction profiles with conversion rates around 50% but product yields less than 5%, generating a plethora of unidentified by-products that complicate downstream purification and waste management.
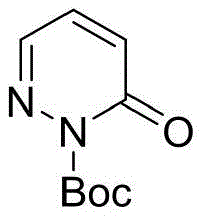
The Novel Approach
The novel approach detailed in CN115557900A circumvents these historical bottlenecks by employing N-protected pyridazin-3-one derivatives as stable precursors. This strategic protection of the nitrogen atom not only enhances the solubility and stability of the starting material but also directs the reactivity towards the desired C3 position. By utilizing aryl diazo compounds as carbene precursors in the presence of a dirhodium catalyst, the reaction proceeds through a highly controlled mechanistic pathway that avoids the random decomposition often seen with free diazo species. This method boasts wide substrate adaptability, tolerating various electron-withdrawing and electron-donating groups on both the pyridazine and the diazo components. The operational simplicity is another major advantage; the reaction requires no specialized high-pressure equipment or cryogenic conditions, functioning effectively at temperatures between 20°C and 35°C, which significantly lowers the energy footprint and safety risks associated with large-scale manufacturing of these valuable chemical building blocks.
Mechanistic Insights into Rhodium-Catalyzed Carbene Insertion and Rearrangement
The core of this transformative chemistry lies in the generation of a reactive rhodium-carbenoid species from the aryl diazo compound. Upon coordination with the dirhodium tetraacetate catalyst, the diazo compound undergoes denitrogenation to form a metal-bound carbene intermediate. This electrophilic species then attacks the carbonyl oxygen of the pyridazin-3-one derivative, forming a transient oxonium ylide. This key intermediate is pivotal as it sets the stage for the subsequent intramolecular rearrangement. Unlike simple insertion reactions that might lead to linear products, the specific geometry of the ylide facilitates an epoxidation-like followed by a rearrangement sequence that ultimately installs the quaternary carbon center at the C3 position of the pyridazine ring. This cascade process is highly atom-economical, incorporating nearly all atoms of the starting materials into the final product, which aligns perfectly with the principles of green chemistry and sustainable manufacturing practices demanded by modern regulatory frameworks.
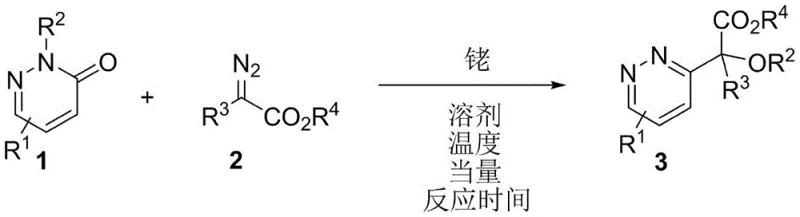
Impurity control in this system is inherently managed by the high selectivity of the rhodium catalyst. The use of N-protecting groups such as Boc prevents side reactions at the nitrogen atom, ensuring that the carbene insertion occurs exclusively at the carbonyl oxygen. This specificity minimizes the formation of N-alkylated by-products or polymeric tars that typically plague diazo chemistry. Furthermore, the mild reaction conditions prevent thermal degradation of the sensitive diazo reagents, which can otherwise decompose explosively or form non-productive dimers. The resulting crude reaction mixtures are generally clean, allowing for straightforward purification via standard silica gel column chromatography or recrystallization. This high level of chemoselectivity is crucial for pharmaceutical applications where strict limits on genotoxic impurities and heavy metal residues must be met, thereby reducing the burden on quality control laboratories and accelerating the release of batch records for clinical supply.
How to Synthesize 3-Substituted Pyridazine Derivatives Efficiently
To implement this synthesis effectively, one must adhere to the optimized protocol which balances catalyst loading with reaction kinetics. The process begins with the precise weighing of the N-protected pyridazin-3-one derivative and the aryl diazo compound, typically in a molar ratio ranging from 1:1 to 1:2.5 to ensure complete consumption of the limiting reagent. The choice of solvent is critical, with dichloromethane proving to be the optimal medium for solubilizing both organic partners while maintaining catalyst stability. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Prepare the reaction mixture by adding pyridazin-3-one derivatives, aryl diazo compounds, and a rhodium catalyst such as Rh2(OAc)4 into a reaction vessel under argon protection.
- Add an appropriate amount of organic solvent like dichloromethane and maintain the reaction temperature between 20°C and 35°C for 15 to 60 minutes.
- Upon completion, remove the solvent under reduced pressure and purify the crude product via silica gel column chromatography or recrystallization to obtain the target 3-substituted pyridazine derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this rhodium-catalyzed methodology offers substantial strategic benefits beyond mere chemical novelty. The primary advantage lies in the drastic simplification of the manufacturing process, which translates directly into cost reduction in pharmaceutical intermediate manufacturing. By eliminating the need for harsh reagents, extreme temperatures, or multi-step protection-deprotection sequences often required in alternative routes, the overall operational expenditure is significantly lowered. The high atom economy means less raw material waste, reducing disposal costs and environmental compliance burdens. Additionally, the short reaction times of 15 to 60 minutes allow for higher throughput in existing reactor vessels, effectively increasing plant capacity without capital investment in new infrastructure, thus enhancing supply chain reliability and responsiveness to market demands.
- Cost Reduction in Manufacturing: The use of commercially available and relatively inexpensive rhodium catalysts at low loadings of just 1 mol% ensures that catalyst costs do not become a prohibitive factor. Since the reaction proceeds at near-room temperature, there is no need for energy-intensive heating or cooling systems, leading to substantial utility savings. The high yields, often exceeding 80% even on scaled-up batches, mean that less starting material is required per kilogram of final product, optimizing the cost of goods sold. Furthermore, the simplified workup procedure reduces labor hours and solvent consumption during purification, contributing to a leaner and more cost-effective production model that improves overall profit margins.
- Enhanced Supply Chain Reliability: The starting materials, including various substituted pyridazin-3-ones and aryl diazo esters, are readily accessible from global chemical suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions ensures consistent batch-to-batch quality, which is essential for maintaining long-term supply contracts with major pharmaceutical clients. The ability to perform the reaction under ambient pressure and moderate temperatures reduces the risk of unplanned shutdowns due to equipment failure or safety incidents. This operational stability guarantees a continuous flow of high-quality intermediates, preventing bottlenecks in the downstream synthesis of active pharmaceutical ingredients and securing the continuity of the entire drug development pipeline.
- Scalability and Environmental Compliance: The patent explicitly demonstrates successful scale-up, with gram-scale reactions maintaining high efficiency, indicating strong potential for commercial scale-up of complex pharmaceutical intermediates. The process generates minimal hazardous waste, primarily consisting of nitrogen gas and spent catalyst which can be recovered or treated easily. The absence of toxic heavy metals other than the trace rhodium catalyst, which is tightly controlled, simplifies the environmental impact assessment. This alignment with green chemistry principles facilitates easier regulatory approval and supports corporate sustainability goals, making the supply chain more resilient against tightening environmental regulations and enhancing the company's reputation as a responsible manufacturer.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method, derived directly from the experimental data and beneficial effects described in the patent documentation. These insights are intended to clarify the practical implementation and strategic value of this technology for potential partners and stakeholders evaluating its integration into their supply chains.
Q: What is the key advantage of this rhodium-catalyzed method over traditional cyclization?
A: Unlike traditional [4+2] cyclization which struggles with regioselectivity, this method efficiently constructs a quaternary carbon center at the 3-position of the pyridazine ring under mild conditions.
Q: Is N-protection of the pyridazinone substrate necessary for this reaction?
A: Yes, comparative data indicates that unprotected pyridazin-3-ones result in complex mixtures and negligible yields, making N-protection critical for reaction success.
Q: Can this process be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the patent demonstrates successful scale-up with maintained high yields, indicating strong potential for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Substituted Pyridazine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this rhodium-catalyzed technology in accelerating drug discovery programs. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required for clinical and commercial grade materials. We understand the critical nature of timeline and quality in the pharmaceutical sector, and our dedicated technical team is prepared to optimize this specific route to meet your unique project requirements, guaranteeing a reliable supply of high-purity pharmaceutical intermediates.
We invite you to collaborate with us to leverage this advanced synthetic capability for your next-generation therapeutics. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating how this efficient route can lower your overall production costs. Please contact us today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets, and let us help you bring your innovative medicines to market faster and more economically.
