Advanced Synthesis of Fenbendazole Intermediate 2-Nitro-4-Thiophenyl Aniline for Industrial Scale
Advanced Synthesis of Fenbendazole Intermediate 2-Nitro-4-Thiophenyl Aniline for Industrial Scale
The pharmaceutical and veterinary industries are constantly seeking more efficient, sustainable, and cost-effective pathways for producing critical active pharmaceutical ingredients (APIs) and their precursors. A significant breakthrough in this domain is detailed in patent CN111499549A, which discloses a novel method for preparing the fenbendazole intermediate, 2-nitro-4-thiophenyl aniline. This specific intermediate is pivotal in the synthesis of fenbendazole, a broad-spectrum benzimidazole anthelmintic widely used for treating gastrointestinal nematodes in livestock. The traditional manufacturing processes have long been plagued by the use of expensive raw materials, harsh reaction conditions involving strong acids, and complex operational procedures that hinder large-scale industrial adoption. The new methodology presented in this patent addresses these critical bottlenecks by introducing a streamlined synthetic route that utilizes readily available starting materials such as N-chloroacetanilide and thiophenol. By shifting away from the conventional reliance on m-chloroaniline, this innovation not only drastically reduces raw material costs but also simplifies the purification process, resulting in a greener, safer, and more economically viable production strategy that aligns perfectly with modern sustainable chemistry principles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 2-nitro-4-thiophenyl aniline has relied heavily on routes starting from m-chloroaniline, a raw material that presents significant economic and logistical challenges due to its high market price and supply chain volatility. As illustrated in the traditional pathway, the synthesis typically involves a multi-step sequence including acylation, nitration using mixed acids (sulfuric and nitric acid), and finally a condensation reaction. 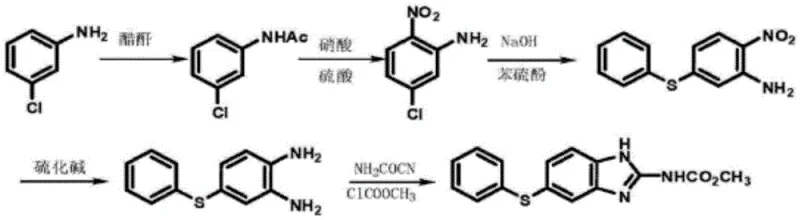 The use of strong mineral acids in the nitration step necessitates specialized corrosion-resistant equipment, driving up capital expenditure (CAPEX) for reactor maintenance and replacement. Furthermore, the generation of acidic wastewater and hazardous byproducts during nitration creates a substantial environmental burden, requiring complex and costly waste treatment protocols to meet regulatory compliance standards. Additionally, alternative literature methods utilizing 2-nitro-5-chloroaniline often require high-boiling polar solvents like DMF and elevated temperatures, which complicate solvent recovery and increase energy consumption, thereby diminishing the overall process efficiency and profitability for manufacturers aiming for cost reduction in veterinary drug manufacturing.
The use of strong mineral acids in the nitration step necessitates specialized corrosion-resistant equipment, driving up capital expenditure (CAPEX) for reactor maintenance and replacement. Furthermore, the generation of acidic wastewater and hazardous byproducts during nitration creates a substantial environmental burden, requiring complex and costly waste treatment protocols to meet regulatory compliance standards. Additionally, alternative literature methods utilizing 2-nitro-5-chloroaniline often require high-boiling polar solvents like DMF and elevated temperatures, which complicate solvent recovery and increase energy consumption, thereby diminishing the overall process efficiency and profitability for manufacturers aiming for cost reduction in veterinary drug manufacturing.
The Novel Approach
In stark contrast to the legacy methods, the innovative route disclosed in the patent employs a direct substitution strategy that fundamentally alters the economic landscape of producing this key intermediate. The new process initiates with the condensation of N-chloroacetanilide and thiophenol in the presence of triethylamine as an acid-binding agent, forming an N-phenylthioacetanilide intermediate in situ.  This intermediate then undergoes a nucleophilic substitution reaction with o-nitroaniline to yield the target 2-nitro-4-thiophenyl aniline without the need for isolation between steps. This "one-pot" or telescoped approach significantly reduces solvent usage, labor hours, and material handling risks. By eliminating the need for hazardous nitrating agents and expensive chlorinated aniline derivatives, the process achieves a remarkable improvement in atom economy and safety profiles. The reaction conditions are notably mild, operating effectively in common polar aprotic solvents like acetonitrile at moderate temperatures, which facilitates easier scale-up and reduces the thermal load on production facilities, making it an ideal candidate for reliable fenbendazole intermediate supplier operations seeking to optimize their manufacturing footprint.
This intermediate then undergoes a nucleophilic substitution reaction with o-nitroaniline to yield the target 2-nitro-4-thiophenyl aniline without the need for isolation between steps. This "one-pot" or telescoped approach significantly reduces solvent usage, labor hours, and material handling risks. By eliminating the need for hazardous nitrating agents and expensive chlorinated aniline derivatives, the process achieves a remarkable improvement in atom economy and safety profiles. The reaction conditions are notably mild, operating effectively in common polar aprotic solvents like acetonitrile at moderate temperatures, which facilitates easier scale-up and reduces the thermal load on production facilities, making it an ideal candidate for reliable fenbendazole intermediate supplier operations seeking to optimize their manufacturing footprint.
Mechanistic Insights into Triethylamine-Mediated Condensation and Substitution
The core of this technological advancement lies in the precise control of the nucleophilic substitution mechanisms facilitated by the choice of base and solvent system. In the initial condensation step, triethylamine acts as a proton scavenger, neutralizing the hydrochloric acid generated when the thiol group of thiophenol attacks the electrophilic carbon attached to the chlorine in N-chloroacetanilide. 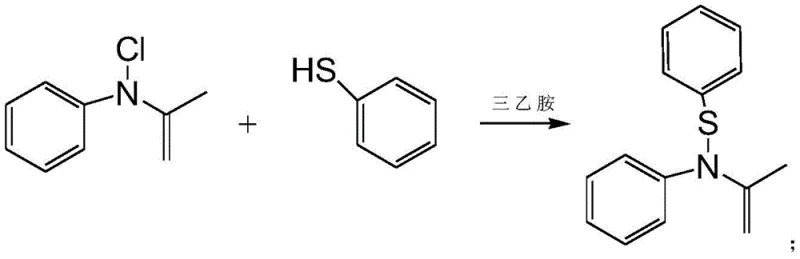 This step is critical for driving the equilibrium forward and preventing the protonation of the nucleophile, which would render it inactive. The selection of polar aprotic solvents such as acetonitrile, MTBE, or tetrahydrofuran is strategic; these solvents stabilize the transition state of the SN2-like substitution without solvating the nucleophile too strongly, thereby maintaining its reactivity. Experimental data suggests that maintaining the condensation temperature between 50°C and 90°C is crucial; temperatures below this range result in incomplete conversion, while excessively high temperatures may promote side reactions or decomposition of the sensitive thioether linkage. The subsequent addition of o-nitroaniline triggers the second substitution event, where the amino group displaces the acetanilide moiety, a transformation that is thermodynamically favored under the optimized thermal conditions of 70°C to 100°C.
This step is critical for driving the equilibrium forward and preventing the protonation of the nucleophile, which would render it inactive. The selection of polar aprotic solvents such as acetonitrile, MTBE, or tetrahydrofuran is strategic; these solvents stabilize the transition state of the SN2-like substitution without solvating the nucleophile too strongly, thereby maintaining its reactivity. Experimental data suggests that maintaining the condensation temperature between 50°C and 90°C is crucial; temperatures below this range result in incomplete conversion, while excessively high temperatures may promote side reactions or decomposition of the sensitive thioether linkage. The subsequent addition of o-nitroaniline triggers the second substitution event, where the amino group displaces the acetanilide moiety, a transformation that is thermodynamically favored under the optimized thermal conditions of 70°C to 100°C.
From an impurity control perspective, this mechanistic pathway offers superior selectivity compared to acid-catalyzed nitration routes. The absence of strong oxidizers minimizes the formation of nitro-group reduction byproducts or ring-oxidized impurities that are notoriously difficult to separate. The use of triethylamine ensures that the reaction medium remains basic throughout the process, which suppresses the formation of azo-compounds or other coupling byproducts that often arise in acidic environments. Furthermore, the telescoped nature of the reaction means that the intermediate N-phenylthioacetanilide does not need to be isolated, reducing the exposure of reactive species to air and moisture which could lead to oxidation of the sulfur atom to sulfoxides or sulfones. This inherent stability within the reaction matrix allows for a cleaner crude product profile, simplifying downstream purification and ensuring that the final high-purity veterinary intermediate meets stringent quality specifications required for animal health applications without extensive recrystallization cycles.
How to Synthesize 2-Nitro-4-Thiophenyl Aniline Efficiently
Implementing this synthesis requires careful attention to stoichiometry and thermal management to replicate the high yields reported in the patent examples. The process begins by charging a reactor with a polar aprotic solvent, typically acetonitrile, followed by the sequential addition of thiophenol and triethylamine. N-chloroacetanilide is then introduced, and the mixture is heated to initiate the condensation phase, which typically proceeds for several hours to ensure complete consumption of the starting materials. Once the condensation is verified, o-nitroaniline is added directly to the same reaction vessel, and the temperature is adjusted to facilitate the substitution reaction. The detailed standardized synthesis steps, including specific molar ratios and exact timing for each phase to maximize the 86%+ yield potential, are outlined in the technical guide below.
- Perform condensation reaction of N-chloroacetanilide and thiophenol in polar aprotic solvent with triethylamine at 50-90°C.
- Add o-nitroaniline directly to the reaction mixture without separation of the intermediate.
- Maintain substitution reaction temperature at 70-100°C to achieve high yield conversion to 2-nitro-4-thiophenyl aniline.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthetic route represents a strategic opportunity to de-risk the supply of critical veterinary intermediates while achieving substantial cost savings. The primary driver of economic value is the substitution of high-cost raw materials; replacing m-chloroaniline with N-chloroacetanilide and thiophenol leverages commodity chemicals that are produced at massive scales globally, ensuring price stability and abundant availability. This shift eliminates the dependency on niche suppliers of specialized chlorinated anilines, thereby enhancing supply chain resilience against market fluctuations. Moreover, the elimination of corrosive mineral acids like sulfuric and nitric acid removes the need for expensive glass-lined or Hastelloy reactors, allowing production to occur in standard stainless steel equipment, which significantly lowers capital depreciation costs and maintenance overheads associated with acid corrosion.
- Cost Reduction in Manufacturing: The economic benefits extend beyond raw material pricing to include significant operational expenditure (OPEX) reductions. By avoiding the use of hazardous nitrating agents, the facility saves considerably on waste disposal costs, as the effluent stream is far less toxic and easier to treat than acidic nitration waste. The telescoped process design reduces solvent consumption and energy usage by eliminating intermediate isolation and drying steps, which are typically energy-intensive. Additionally, the higher atom utilization rate means less material is lost to byproducts, directly improving the mass balance and reducing the cost per kilogram of the final API intermediate. These cumulative efficiencies translate into a more competitive pricing structure for the finished fenbendazole, allowing manufacturers to improve margins or offer more aggressive pricing in the global veterinary market.
- Enhanced Supply Chain Reliability: The robustness of this chemical process contributes directly to supply continuity. Because the reaction conditions are mild and the reagents are stable, the risk of batch failure due to thermal runaway or equipment corrosion is minimized. The use of common solvents like acetonitrile ensures that solvent supply chains are not a bottleneck, unlike specialized high-boiling solvents that might have limited suppliers. Furthermore, the simplified workflow reduces the total cycle time for production, enabling faster turnaround from order to delivery. This agility is crucial for meeting the fluctuating demands of the agricultural sector, particularly during peak dosing seasons, ensuring that downstream formulators have a consistent and reliable flow of high-quality intermediate material.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the lack of extreme pressure or cryogenic requirements. The reaction exotherms are manageable, and the use of triethylamine as a base is a well-understood unit operation in fine chemical manufacturing. From an environmental standpoint, the process aligns with green chemistry principles by reducing the E-factor (mass of waste per mass of product). The absence of heavy metal catalysts and strong acids simplifies regulatory compliance regarding wastewater discharge and worker safety. This environmental stewardship not only mitigates regulatory risk but also enhances the brand reputation of the manufacturer as a sustainable partner, which is increasingly a prerequisite for contracts with major multinational animal health companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on yield optimization, safety parameters, and scalability factors. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their own production lines or for procurement specialists assessing the quality and consistency of suppliers utilizing this technology.
Q: What are the primary advantages of this new synthetic route over traditional methods?
A: The new route replaces expensive m-chloroaniline with cost-effective N-chloroacetanilide and thiophenol, eliminates the use of strong corrosive acids like sulfuric and nitric acid, and operates under milder conditions, significantly reducing equipment corrosion and three-waste discharge.
Q: What represents the optimal reaction conditions for maximum yield?
A: Experimental data indicates that maintaining the condensation reaction between 50-90°C and the subsequent substitution reaction between 70-100°C, using acetonitrile as a solvent, yields the highest conversion rates, reaching up to 86.25%.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes common polar aprotic solvents like acetonitrile and avoids extreme pressure or temperature requirements, making it highly scalable with standard stainless steel reactors while ensuring high atom utilization and safety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Nitro-4-Thiophenyl Aniline Supplier
At NINGBO INNO PHARMCHEM, we recognize that the efficiency of your final veterinary product depends heavily on the quality and consistency of your starting materials. Our technical team has thoroughly analyzed the synthetic pathway described in patent CN111499549A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovative process to life. We are committed to delivering high-purity 2-nitro-4-thiophenyl aniline that meets stringent purity specifications, utilizing our rigorous QC labs to ensure every batch is free from the impurities associated with older nitration-based methods. Our facility is equipped to handle the specific solvent systems and thermal profiles required for this condensation-substitution sequence, guaranteeing a supply that is both chemically superior and commercially stable.
We invite you to collaborate with us to optimize your supply chain for fenbendazole production. By leveraging this advanced synthetic route, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how switching to our material can lower your overall COGS. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a sustainable, cost-effective, and high-quality source of this critical intermediate, ensuring your production lines run smoothly and efficiently throughout the year.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
