Advanced Synthesis of Dapagliflozin Peroxide for High-Purity Pharmaceutical Intermediates
The global pharmaceutical landscape is increasingly focused on the rigorous control of genotoxic impurities to ensure patient safety, particularly for widely prescribed medications like SGLT-2 inhibitors. Patent CN116410163A discloses a groundbreaking preparation method for Dapagliflozin Peroxide, specifically identified as Impurity A ((2S,3R,4R,5S,6R)-2-(4-chloro-3-((4-ethoxyphenyl)(hydroperoxy)methyl)phenyl)-6-(hydroxymethyl)tetrahydro-2H-pyran-3,4,5-triol). As a critical genotoxic impurity, Impurity A poses significant risks even at trace levels, necessitating robust analytical methods and high-purity reference standards for quality control. This patent addresses a significant gap in the prior art, where no reliable synthetic routes existed to generate this specific peroxide derivative in sufficient quantity and purity. By establishing a clear, reproducible pathway, this technology empowers reliable pharmaceutical intermediate suppliers to support drug manufacturers in meeting stringent regulatory requirements set by agencies such as the FDA and EMA.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this invention, the synthesis of Dapagliflozin Peroxide was fraught with significant technical challenges and uncertainties. Existing literature lacked specific methodologies for introducing the hydroperoxy group at the benzylic position without degrading the sensitive glycosidic backbone or causing over-oxidation. Comparative examples within the patent data highlight these failures; for instance, attempts to oxidize related precursors like Compound X under various conditions (using hydrogen peroxide with or without acid catalysts at elevated temperatures) resulted in negligible yields of the target Impurity A. In many cases, the starting material remained largely unreacted, or the reaction produced complex mixtures of degradation products rather than the desired peroxide. These conventional approaches often suffered from poor selectivity, requiring harsh conditions that compromised the structural integrity of the molecule, thereby failing to provide the high-purity reference standards necessary for validating analytical methods in cost reduction in API manufacturing workflows.
The Novel Approach
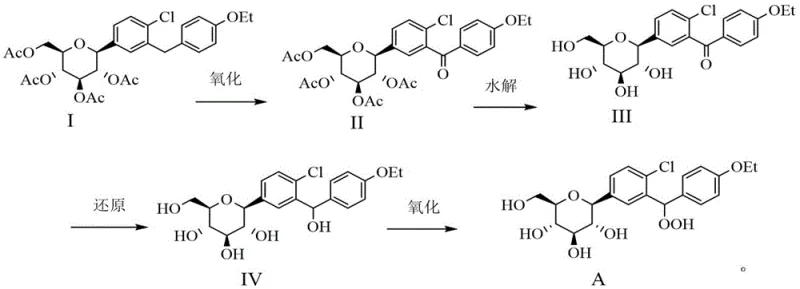
The patented methodology introduces a highly efficient, four-step synthetic strategy that overcomes the selectivity issues of previous attempts. The core innovation lies in the precise oxidation of the alcohol precursor (Compound IV) to the hydroperoxide (Compound A) using hydrogen peroxide in the presence of an acidic catalyst. This route begins with the oxidation of a protected precursor to a ketone, followed by deprotection and reduction to establish the correct stereochemistry before the final peroxidation. The process utilizes readily available reagents and operates under mild conditions, ensuring high product purity and yield. By avoiding exotic catalysts and extreme temperatures, this approach significantly simplifies the production workflow. The overall transformation is depicted below, showcasing the logical progression from the stable acetyl-protected starting material to the final genotoxic impurity standard.
Mechanistic Insights into Acid-Catalyzed Benzylic Peroxidation
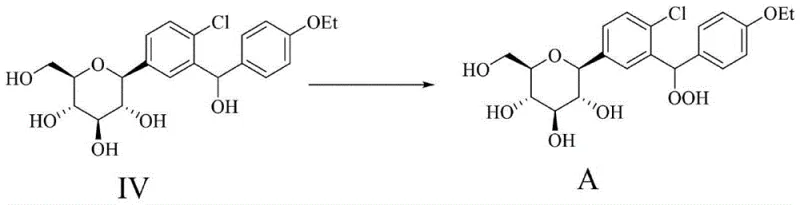
The success of this synthesis hinges on the mechanistic nuances of the final oxidation step, where Compound IV is converted to Compound A. This transformation involves the substitution of a benzylic hydroxyl group with a hydroperoxy moiety (-OOH). The reaction is facilitated by a strong acid catalyst, preferably sulfuric acid, which protonates the benzylic alcohol, generating a stabilized carbocation intermediate due to the resonance effect of the adjacent aromatic rings. In the presence of excess hydrogen peroxide, the nucleophilic attack by the peroxide anion or neutral hydrogen peroxide molecule occurs at this electrophilic center. The choice of solvent, specifically acetonitrile, plays a crucial role in stabilizing the transition state and solubilizing both the organic substrate and the aqueous oxidant. Furthermore, the stoichiometry is carefully controlled, with a molar ratio of Compound IV to hydrogen peroxide ranging from 1:10 to 1:40, ensuring that the equilibrium favors the formation of the peroxide while minimizing side reactions.
Impurity control is paramount in this mechanism, as peroxides can be unstable and prone to decomposition. The patent specifies a post-reaction treatment involving the addition of catalase to decompose excess hydrogen peroxide, followed by pH adjustment and freeze-drying. This careful quenching prevents the degradation of the newly formed hydroperoxide bond during workup. The specificity of the acid catalyst is also critical; strong mineral acids like sulfuric acid provide the necessary proton activity without introducing nucleophilic counterions that could compete with the peroxide. This mechanistic precision ensures that the resulting high-purity pharmaceutical intermediates meet the strict specifications required for use as reference standards in HPLC and LC-MS analysis, enabling accurate quantification of genotoxic risks in the final drug product.
How to Synthesize Dapagliflozin Peroxide Efficiently
The synthesis of Dapagliflozin Peroxide requires precise control over reaction parameters to maximize yield and safety. The process is divided into four distinct stages: initial oxidation of the benzylic methylene group, hydrolysis of protecting groups, stereoselective reduction of the ketone, and the final peroxidation. Each step builds upon the previous one to establish the correct functionalization and stereochemistry. The following guide outlines the operational framework derived from the patent examples, emphasizing the critical control points for temperature, stoichiometry, and workup procedures. For laboratory personnel and process engineers, adhering to these standardized protocols is essential for reproducing the high purity levels reported in the patent data.
- Oxidize Compound I using potassium permanganate in acetonitrile to form the ketone Compound II.
- Hydrolyze the acetyl groups of Compound II using lithium hydroxide in methanol to yield the triol Compound III.
- Reduce the ketone moiety of Compound III using sodium borohydride to generate the alcohol precursor Compound IV.
- Perform the critical acid-catalyzed oxidation of Compound IV with hydrogen peroxide and sulfuric acid to obtain Dapagliflozin Peroxide (Compound A).
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented route offers substantial strategic benefits by leveraging commodity chemicals and standard unit operations. The reliance on inexpensive, bulk-available reagents such as potassium permanganate, sodium borohydride, and hydrogen peroxide eliminates the dependency on scarce or proprietary catalysts that often bottleneck production. This accessibility translates directly into cost reduction in API manufacturing, as the raw material costs are minimized and supply volatility is reduced. Furthermore, the reaction conditions are温和 (mild), typically operating between 0°C and 55°C, which reduces energy consumption compared to processes requiring cryogenic cooling or high-temperature reflux. The simplicity of the workup procedures, involving standard filtration, extraction, and column chromatography, allows for seamless integration into existing manufacturing infrastructure without the need for specialized equipment.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of low-cost oxidants and reducing agents. Unlike methods requiring expensive transition metal catalysts or complex enzymatic systems, this route utilizes standard inorganic reagents that are globally sourced and price-stable. The elimination of costly purification steps, such as preparative HPLC for intermediate isolation (except for the final high-purity standard), further drives down the cost of goods sold. Additionally, the high selectivity of the reaction minimizes the formation of difficult-to-remove byproducts, reducing the waste disposal costs associated with complex impurity profiles.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of non-proprietary starting materials. Compound I and subsequent intermediates are synthesized from common building blocks, ensuring that raw material shortages are unlikely to disrupt production schedules. The robustness of the chemistry means that the process is less sensitive to minor variations in reagent quality, providing a buffer against supply chain fluctuations. This reliability is crucial for maintaining the continuous availability of reference standards, which are needed periodically for batch release testing throughout the lifecycle of the Dapagliflozin drug product.
- Scalability and Environmental Compliance: The process is inherently scalable, moving from gram-scale laboratory synthesis to multi-kilogram production with minimal modification. The use of acetonitrile and methanol as solvents allows for efficient recovery and recycling, aligning with green chemistry principles and reducing the environmental footprint. The waste streams generated are primarily aqueous salts and organic solvents, which can be treated using standard wastewater management protocols. This ease of commercial scale-up of complex organic molecules ensures that manufacturers can rapidly respond to increased demand for impurity standards without compromising on safety or regulatory compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of Dapagliflozin Peroxide. These insights are derived directly from the technical specifications and experimental data provided in the patent documentation. Understanding these details helps stakeholders assess the feasibility of adopting this method for their quality control laboratories or pilot production facilities.
Q: Why is the control of Dapagliflozin Peroxide (Impurity A) critical in API manufacturing?
A: Impurity A is classified as a genotoxic impurity, meaning it can damage human genetic material even at very low concentrations, potentially leading to mutations or tumorigenesis. Regulatory bodies like the FDA and EMA require strict monitoring and control of such impurities to ensure patient safety, making the availability of high-purity reference standards essential for method validation and quality control.
Q: What are the key advantages of this patented synthesis route over conventional methods?
A: The patented route utilizes commercially available and inexpensive reagents such as hydrogen peroxide, sulfuric acid, and sodium borohydride. Unlike prior art methods which often fail to produce the target peroxide or result in complex mixtures, this method offers a straightforward four-step sequence with manageable reaction conditions (0-55°C) and a simplified workup procedure involving filtration and extraction.
Q: Is this synthesis process scalable for industrial production?
A: Yes, the process is designed with industrial application in mind. The reactions utilize common organic solvents like acetonitrile and methanol, and the temperature ranges are moderate, avoiding cryogenic conditions or extreme heat. The workup steps, such as diatomaceous earth filtration and liquid-liquid extraction, are standard unit operations easily adaptable to large-scale reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dapagliflozin Peroxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality reference standards in ensuring the safety and efficacy of pharmaceutical products. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency. We are committed to delivering stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the identity and purity of every batch of Dapagliflozin Peroxide we supply. Our facility is equipped to handle sensitive peroxide chemistry safely, adhering to the highest international safety and quality standards.
We invite you to collaborate with us to optimize your supply chain for Dapagliflozin impurity controls. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your regulatory compliance and product quality goals efficiently.
