Advanced Synthesis of Azacycloacene Compounds for High-Performance OLED Manufacturing
The rapid evolution of the organic light-emitting diode (OLED) industry demands intermediates that combine structural precision with manufacturing efficiency. Patent CN113563366A introduces a breakthrough in the synthesis of azacycloacene compounds, specifically 4,9-dibromo-6,7-dimethyl-[1,2,5]thiadiazolo[3,4-g]quinoxaline, which serves as a critical building block for large acene compounds used in next-generation display technologies. This technical disclosure addresses the longstanding industry challenge of balancing high purity with scalable production metrics. By leveraging a novel two-step pathway involving acidic reduction and catalytic condensation, the patent outlines a method that drastically improves yield consistency while maintaining rigorous quality standards required for electronic applications. For R&D directors and procurement specialists alike, understanding the mechanistic advantages of this route is essential for optimizing supply chains and reducing the total cost of ownership for high-performance luminescent materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for complex heterocyclic intermediates often suffer from severe inefficiencies that hinder commercial viability. Comparative data within the patent landscape reveals that conventional methods frequently rely on extreme reaction conditions, such as prolonged heating at 80°C or higher, which not only increases energy consumption but also accelerates equipment degradation and safety risks. Furthermore, these legacy processes often exhibit poor atom economy and generate significant amounts of difficult-to-remove impurities, leading to overall yields that struggle to exceed 50% after purification. The reliance on harsh alkaline workups and extended reaction times of over 12 hours creates bottlenecks in production scheduling, making it difficult for suppliers to meet the tight delivery windows expected by multinational electronics manufacturers. These technical deficiencies translate directly into higher operational costs and inconsistent batch-to-batch quality, posing a significant risk to the stability of the downstream supply chain for OLED panel producers.
The Novel Approach
In stark contrast, the methodology disclosed in CN113563366A offers a streamlined alternative that redefines process efficiency for azacycloacene synthesis. The novel approach utilizes a mild acidic environment facilitated by lower fatty acids like acetic acid, allowing the reaction to proceed effectively at temperatures as low as 45-55°C. This significant reduction in thermal demand preserves the integrity of the reaction vessel and minimizes the formation of thermal degradation byproducts. The integration of a specific reducing agent system in the first step, followed by a palladium-catalyzed cyclization in the second step, ensures a rapid conversion rate that completes the entire synthesis within approximately 8 to 9.5 hours. This acceleration in reaction kinetics allows for higher throughput without compromising the structural fidelity of the target molecule. By shifting away from harsh conditions to this controlled, mild protocol, manufacturers can achieve a more robust and predictable production cycle that aligns with the rigorous demands of modern fine chemical manufacturing.
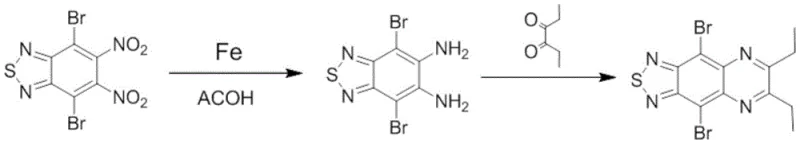
Mechanistic Insights into Iron-Mediated Reduction and Pd/C Catalyzed Cyclization
The core of this technological advancement lies in the precise control of the reduction mechanism during the initial synthesis stage. The process employs iron powder as a reducing agent within an acidic medium, where the presence of hydrogen ions plays a pivotal role in facilitating electron transfer to the nitro groups of the 4,7-dibromo-5,6-dinitrobenzo[c][1,2,5]thiadiazole precursor. This acidic environment activates the reducing agent, increasing the number of active sites available for the reaction and significantly enhancing the probability of successful nitro group reduction to amines. The careful regulation of temperature between 45°C and 55°C ensures that the reduction proceeds selectively without affecting other sensitive functional groups on the aromatic ring. This selectivity is crucial for maintaining the purity profile of the intermediate diamine, which serves as the foundation for the subsequent cyclization step. By optimizing the molar ratio of the substrate to the reducing agent, the process minimizes the presence of unreacted starting materials, thereby simplifying the downstream purification requirements and reducing waste generation.
Following the reduction, the cyclization mechanism leverages the catalytic activity of Pd/C to drive the condensation reaction with 3,4-hexanedione. This step is critical for forming the fused quinoxaline ring system that defines the electronic properties of the final azacycloacene compound. The use of a heterogeneous catalyst like Pd/C allows for efficient reaction progression at mild temperatures around 45-50°C, avoiding the need for high-energy inputs that could lead to polymerization or decomposition. The acidic conditions continue to play a supportive role by protonating intermediate species, making them more susceptible to nucleophilic attack and ring closure. This dual-stage mechanistic approach ensures that the final product achieves a purity level exceeding 90% directly from the reaction mixture, significantly reducing the burden on purification columns. The result is a highly efficient transformation that maximizes the yield of the target luminescent intermediate while strictly controlling the impurity spectrum to meet the exacting standards of the optoelectronic industry.
![Chemical structure of 4,9-dibromo-6,7-dimethyl-[1,2,5]thiadiazolo[3,4-g]quinoxaline showing the fused heterocyclic core](/insights/img/4-9-dibromo-thiadiazoloquinoxaline-oled-intermediate-supplier-20260315035835-01.png)
How to Synthesize 4,9-Dibromo-6,7-dimethyl-[1,2,5]thiadiazolo[3,4-g]quinoxaline Efficiently
Implementing this synthesis route requires careful attention to reagent preparation and environmental controls to ensure optimal outcomes. The process begins with the rigorous drying of organic solvents and fatty acids to moisture levels below 200 ppm, as water content can significantly inhibit the reduction rate and extend reaction times. Operators must maintain a nitrogen atmosphere throughout the procedure to prevent oxidation of sensitive intermediates and ensure the safety of the exothermic reduction step. The batch-wise addition of the nitro precursor is essential to manage heat generation and maintain the temperature within the narrow 45-55°C window. Detailed standard operating procedures regarding stirring speeds, filtration techniques, and recrystallization solvents are critical for reproducing the high yields reported in the patent examples. For a comprehensive guide on the specific operational parameters and safety protocols required for this synthesis, please refer to the standardized technical documentation provided below.
- Perform acidic reduction of 4,7-dibromo-5,6-dinitrobenzo[c][1,2,5]thiadiazole using iron powder in acetic acid at 45-55°C.
- Conduct condensation reaction with 3,4-hexanedione using Pd/C catalyst under acidic conditions at 45-50°C.
- Purify the final azacycloacene compound via recrystallization or column chromatography to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this synthesis methodology offers substantial benefits that extend beyond simple chemical yield. The shift to milder reaction conditions directly correlates with a reduction in energy consumption and equipment maintenance costs, as the process does not require high-pressure or high-temperature reactors. This operational simplicity allows for greater flexibility in manufacturing scheduling, enabling suppliers to respond more agilely to fluctuating market demands for OLED materials. Furthermore, the use of commercially abundant reagents such as iron powder and acetic acid mitigates the risk of raw material shortages that often plague supply chains dependent on exotic or specialized catalysts. The robustness of the process ensures a consistent supply of high-quality intermediates, reducing the likelihood of production delays caused by batch failures or quality deviations. For supply chain heads, this translates into a more reliable sourcing strategy that supports long-term production planning for electronic device manufacturing.
- Cost Reduction in Manufacturing: The elimination of extreme thermal requirements and the use of cost-effective reducing agents like iron powder significantly lower the variable costs associated with production. By avoiding expensive high-temperature equipment and reducing the energy load per batch, manufacturers can achieve a more favorable cost structure without sacrificing product quality. The simplified workup procedure, which avoids complex alkaline extractions, further reduces the consumption of auxiliary chemicals and waste disposal costs. These cumulative efficiencies contribute to a more competitive pricing model for the final intermediate, allowing downstream clients to optimize their own material costs in the highly price-sensitive display market.
- Enhanced Supply Chain Reliability: The reliance on readily available commodity chemicals ensures that the production of this intermediate is not vulnerable to the supply constraints often associated with specialized reagents. The short reaction cycle time of under 10 hours allows for increased production turnover, enabling suppliers to maintain higher inventory levels and shorter lead times for customer orders. This agility is crucial for maintaining continuity in the supply of critical materials for the electronics sector, where production stoppages can have cascading effects on global device availability. The proven scalability of the method provides confidence that supply volumes can be ramped up quickly to meet surges in demand without compromising on the stringent purity specifications required for electronic applications.
- Scalability and Environmental Compliance: The mild nature of the reaction conditions facilitates easier scale-up from laboratory to industrial production, as the thermal management requirements remain manageable even at larger volumes. The process generates less hazardous waste compared to traditional methods, aligning with increasingly strict environmental regulations and sustainability goals within the chemical industry. The use of recyclable solvents and the minimization of heavy metal waste through efficient catalyst usage further enhance the environmental profile of the manufacturing process. This compliance not only reduces regulatory risk but also appeals to end-users who are prioritizing sustainable sourcing practices in their supply chain audits and corporate responsibility initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this azacycloacene compound. These answers are derived directly from the experimental data and technical disclosures found in the patent literature, providing a reliable basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this intermediate into their specific product formulations. The information covers aspects ranging from reaction optimization to quality control metrics, ensuring a comprehensive understanding of the material's capabilities and limitations in a commercial context.
Q: What are the key advantages of this synthesis method over conventional routes?
A: The patent CN113563366A demonstrates a method that significantly reduces reaction time to under 10 hours and operates at mild temperatures below 55°C, avoiding the harsh conditions and low yields associated with traditional high-temperature processes.
Q: How does this process impact impurity profiles in OLED materials?
A: By utilizing controlled acidic reduction and specific catalytic cyclization, the process minimizes side reactions, resulting in a crude product purity exceeding 90% before final purification, which is critical for electronic performance.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the use of readily available reagents like iron powder and acetic acid, combined with low equipment requirements due to mild reaction conditions, makes this method highly scalable and cost-effective for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,9-Dibromo-6,7-dimethyl-[1,2,5]thiadiazolo[3,4-g]quinoxaline Supplier
As a leader in the fine chemical sector, NINGBO INNO PHARMCHEM is uniquely positioned to leverage advanced synthesis routes like CN113563366A to deliver superior value to our global partners. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 4,9-dibromo-6,7-dimethyl-[1,2,5]thiadiazolo[3,4-g]quinoxaline meets the exacting standards required for high-performance OLED applications. Our commitment to technical excellence ensures that our clients receive materials that not only perform reliably in their devices but also support their broader goals of cost efficiency and supply chain stability.
We invite procurement leaders and R&D directors to collaborate with us to explore how this optimized synthesis route can enhance your product portfolio. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and production timelines. We encourage you to reach out for specific COA data and route feasibility assessments to verify how our capabilities align with your project needs. Partnering with us means gaining access to a supply chain that is both robust and innovative, ready to support the next generation of electronic materials with speed and precision.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
