Advanced Catalytic Hydrogenation for Beta-Tricyclic Amine Hydrochloride: Scaling High-Purity Analgesic Intermediates
The pharmaceutical industry continuously seeks robust synthetic pathways for critical analgesic intermediates, and Patent CN102060720A represents a significant technological leap in the production of beta-tricyclic amine hydrochloride. This specific compound serves as a pivotal building block in the synthesis of potent opioid analgesics, where stereochemical purity and process efficiency are paramount for regulatory compliance and commercial viability. The disclosed methodology replaces traditional, cumbersome reduction techniques with a streamlined catalytic hydrogenation protocol utilizing a palladium-carbon (Pd/C) system. By optimizing reaction parameters such as hydrogen pressure, temperature, and solvent composition, this innovation addresses long-standing challenges regarding yield optimization and impurity profiles. For R&D directors and process chemists, understanding the nuances of this transition from older nickel-based systems to modern palladium catalysis is essential for evaluating supply chain resilience and manufacturing cost structures. This report analyzes the technical merits of this patent to demonstrate how it enables the reliable supply of high-purity pharmaceutical intermediates while drastically simplifying the operational workflow.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of tricyclic amine derivatives relied heavily on Raney-Nickel catalysts, a technology fraught with significant operational and economic drawbacks that hindered efficient large-scale production. The conventional process typically necessitated a two-step reduction sequence where the tricyclic ketoxime was first reduced to an intermediate imine or amine, followed by a secondary reduction step to achieve the final target molecule. This multi-stage approach inherently compounded material losses, resulting in overall yields that frequently struggled to exceed forty-seven percent, thereby inflating the cost of goods sold. Furthermore, Raney-Nickel is a pyrophoric material that requires careful handling under inert atmospheres, introducing substantial safety hazards and requiring specialized equipment for storage and disposal. The inability to effectively recover and reuse the nickel catalyst meant that every batch incurred fresh catalyst costs, while the generation of heavy metal waste posed environmental compliance challenges. Additionally, the crude product from these older methods often contained complex impurity profiles that necessitated energy-intensive purification techniques, such as molecular distillation under high vacuum, which further eroded profit margins and limited throughput capacity.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a 5-10% palladium-carbon catalyst to effect a direct, one-pot reduction of the tricyclic ketoxime to the desired beta-tricyclic amine hydrochloride. This methodological shift eliminates the need for intermediate isolation and the associated yield penalties, allowing the reaction to proceed with a single charging of reagents in an ethanol and strong ammonia water solvent system. The use of Pd/C not only enhances the reaction kinetics but also significantly improves the stereoselectivity towards the beta-isomer, achieving contents greater than ninety-five percent as verified by high-performance liquid chromatography. A critical advantage of this system is the mechanical stability and reusability of the palladium catalyst, which can be recovered and reapplied for multiple cycles without significant loss of activity, thereby driving down raw material expenses. The process operates under moderate temperatures of 40 to 50 degrees Celsius and hydrogen pressures around 25 to 35 atmospheres, conditions that are readily achievable in standard industrial autoclaves. By bypassing the need for molecular distillation and relying on simple crystallization for purification, this route offers a scalable, cost-effective solution for the commercial scale-up of complex pharmaceutical intermediates.
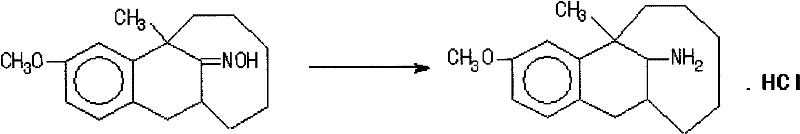
Mechanistic Insights into Pd/C-Catalyzed Hydrogenation
The mechanistic pathway of this transformation involves the adsorption of the tricyclic ketoxime onto the active sites of the palladium surface, where it undergoes hydrogenolysis of the nitrogen-oxygen bond. The presence of strong ammonia water in the reaction medium plays a dual role: it acts as a base to neutralize any acidic byproducts generated during the reduction and, more importantly, it suppresses the formation of secondary amines which often arise from the condensation of the primary amine product with remaining ketone or aldehyde impurities. The ammonia likely coordinates with the catalyst surface or the intermediate species, modulating the electronic environment to favor the formation of the primary amine over oligomerization side reactions. This suppression of side reactions is crucial for maintaining a clean impurity profile, which simplifies downstream processing and ensures that the final API meets stringent pharmacopeial standards. The hydrogen gas dissociates on the palladium surface into atomic hydrogen, which then attacks the C=N bond of the oxime or the intermediate imine, sequentially adding hydrogen atoms to form the saturated amine linkage. The stereochemical outcome, favoring the beta-isomer, is dictated by the steric constraints of the tricyclic framework and the specific interaction of the substrate with the catalyst surface in the ammoniacal ethanol solution.
Impurity control in this system is achieved through the precise regulation of reaction time and hydrogen uptake. Over-reduction or prolonged exposure to hydrogenation conditions can lead to the hydrogenolysis of other sensitive functional groups, such as the methoxy ether present on the aromatic ring, although the patent indicates high selectivity under the specified conditions. The filtration step post-reaction effectively removes the heterogeneous catalyst, preventing metal leaching into the final product, a common concern in pharmaceutical manufacturing. Subsequent treatment with concentrated hydrochloric acid converts the free amine into its hydrochloride salt, which exhibits lower solubility in the ethanol solvent system, prompting crystallization. This salt formation step serves as an additional purification stage, as many organic impurities remain in the mother liquor while the desired beta-tricyclic amine hydrochloride precipitates as high-purity white crystals. The ability to achieve moisture content below 0.5 percent after drying indicates a robust crystal lattice that is stable for storage and transport, further validating the commercial viability of this synthetic route for global supply chains.
How to Synthesize Beta-Tricyclic Amine Hydrochloride Efficiently
Executing this synthesis requires strict adherence to the optimized parameters defined in the patent to ensure maximum yield and safety. The process begins with the preparation of the reaction mixture in a pressure-rated autoclave, combining the tricyclic ketoxime starting material with the 5-10% Pd/C catalyst in a solvent blend of dehydrated ethanol and concentrated ammonia water. It is critical to maintain the ratio of substrate to catalyst within the specified range to balance reaction rate with economic efficiency. Once the reactor is sealed and purged with inert gas, high-purity hydrogen is introduced to reach the target pressure, and the mixture is heated to the designated temperature range with vigorous stirring to ensure efficient mass transfer of hydrogen gas into the liquid phase. Monitoring the hydrogen consumption allows operators to determine the reaction endpoint, after which the vessel is cooled and depressurized safely. The detailed standardized synthetic steps, including specific workup procedures and quality control checkpoints, are outlined in the guide below.
- Charge the autoclave with tricyclic ketoxime (Compound II), 5-10% Pd/C catalyst, ethanol, and strong ammonia water.
- Pressurize the reactor with high-purity hydrogen to 25-35 atm and maintain temperature between 40-50°C for 15-24 hours.
- Filter the catalyst, concentrate the filtrate, and treat the residue with ethanol and concentrated hydrochloric acid to crystallize the pure hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Pd/C catalyzed route translates into tangible strategic advantages regarding cost stability and operational reliability. The shift away from pyrophoric Raney-Nickel to stable Pd/C significantly reduces the safety overhead and insurance costs associated with handling hazardous materials, while the ability to reuse the catalyst multiple times drastically lowers the recurring cost of consumables per kilogram of product. This process intensification, characterized by the elimination of the second reduction step and the removal of molecular distillation, shortens the overall cycle time and increases the throughput capacity of existing manufacturing assets. Consequently, this leads to substantial cost savings in API manufacturing by reducing energy consumption, solvent usage, and labor hours required for purification. The robustness of the crystallization-based purification also minimizes the risk of batch failures due to distillation column fouling or thermal degradation, ensuring a more consistent supply of material for downstream drug formulation.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the high atom economy of the one-pot reduction and the recyclability of the precious metal catalyst. Unlike single-use nickel catalysts, the palladium-on-carbon system retains its activity over several batches, allowing for the recovery of valuable palladium and spreading the initial capital cost of the catalyst over a larger production volume. Furthermore, the avoidance of high-vacuum molecular distillation eliminates a major bottleneck in production, reducing utility costs associated with heating and vacuum generation. The simplified workflow means fewer unit operations, which directly correlates to reduced equipment maintenance and lower operational expenditure, providing a competitive edge in pricing for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: From a logistics perspective, the use of stable, non-pyrophoric catalysts simplifies the storage and transportation requirements for raw materials, mitigating the risk of supply disruptions caused by hazardous material shipping regulations. The high yield and purity achieved in the crude reaction reduce the dependency on complex external purification services or specialized toll manufacturers, allowing for more vertical integration within the supply chain. This self-sufficiency enhances the agility of the supply network, enabling faster response times to market demand fluctuations for analgesic drugs. Additionally, the use of common solvents like ethanol and ammonia ensures that raw material sourcing remains resilient against regional shortages of exotic reagents.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing standard hydrogenation equipment found in most fine chemical facilities, which facilitates rapid technology transfer from pilot plant to commercial scale. The reduction in waste generation, particularly the absence of heavy metal sludge from disposable nickel catalysts and the lower solvent volumes required due to higher concentrations, aligns with increasingly stringent environmental regulations. This green chemistry approach not only reduces waste disposal costs but also improves the sustainability profile of the final pharmaceutical product, a factor that is becoming increasingly important for corporate social responsibility reporting and regulatory approvals in major markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this hydrogenation technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, aiming to clarify the operational benefits for potential partners. Understanding these specifics helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios for analgesic intermediates.
Q: Why is Pd/C preferred over Raney-Ni for this specific tricyclic reduction?
A: Pd/C offers superior selectivity for the beta-isomer and can be reused multiple times, whereas Raney-Ni often requires a two-step process with lower overall yields and poses higher safety risks due to pyrophoric nature.
Q: What is the role of ammonia water in the reaction solvent system?
A: Strong ammonia water acts as a basic modifier that suppresses the formation of secondary amine byproducts and stabilizes the intermediate imine species, ensuring high conversion to the primary amine target.
Q: Can this process be scaled for industrial production without molecular distillation?
A: Yes, the patented method eliminates the need for complex molecular distillation by achieving high crude purity directly through crystallization, making it highly suitable for large-scale industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Tricyclic Amine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in the competitive landscape of pain management therapeutics. Our team of expert process chemists has thoroughly evaluated the Pd/C catalyzed hydrogenation pathway described in Patent CN102060720A and confirmed its potential for robust commercial execution. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and compliant with cGMP standards. Our state-of-the-art facilities are equipped with high-pressure hydrogenation reactors and rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of beta-tricyclic amine hydrochloride meets the exacting requirements of global pharmaceutical clients.
We invite you to collaborate with us to optimize your supply chain for this key analgesic intermediate. By leveraging our technical expertise, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our implementation of this advanced catalytic method can lower your total cost of ownership. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your drug development pipeline.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
