Advanced Synthesis of Hydrogenated Ring-Band [8] Arene Compounds for Supramolecular Applications
The chemical landscape of supramolecular chemistry is continually evolving with the discovery of novel macrocyclic hosts that offer precise molecular recognition capabilities. Patent CN110372468B introduces a significant advancement in this field by disclosing a robust preparation method for hydrogenated ring-band [8] aromatic hydrocarbon compounds. These unique structures, characterized by their barrel-shaped cavity architecture, variable cavity volume, and tunable inner wall polarity, represent a new class of artificially synthesized macrocyclic molecules. Unlike traditional crown ethers or calixarenes, these hydrogenated ring-band [8] arenes provide distinct electronic properties that enable the selective recognition of organic molecules from mixed solutions. This technological breakthrough is particularly relevant for industries requiring high-performance separation materials and advanced sensing platforms, positioning these compounds as critical functional macrocyclic intermediates for next-generation applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex macrocyclic architectures often suffered from rigorous reaction conditions, low overall yields, and limited control over stereochemistry. Traditional routes to similar cage-like or belt-shaped molecules frequently relied on high-temperature thermodynamic control or multiple steps involving expensive transition metal catalysts that were difficult to remove completely. Furthermore, achieving the specific barrel-shaped conformation required for effective guest inclusion was often unpredictable, leading to mixtures of oligomers or polymers rather than the discrete macrocycle. The lack of mild, direct cyclization methods meant that scaling these processes for industrial applications was fraught with challenges regarding reproducibility and cost efficiency. Consequently, the supply of high-purity supramolecular hosts remained constrained, limiting their adoption in commercial separation technologies.
The Novel Approach
The methodology outlined in patent CN110372468B overcomes these historical barriers by utilizing a direct intramolecular Friedel-Crafts alkylation strategy. This novel approach starts from readily available calix[4]resorcinarene derivatives, specifically compounds of formula (II) or (III), and subjects them to acid-catalyzed cyclization. As illustrated in the reaction scheme below, this process builds the ring structure efficiently under relatively mild conditions.
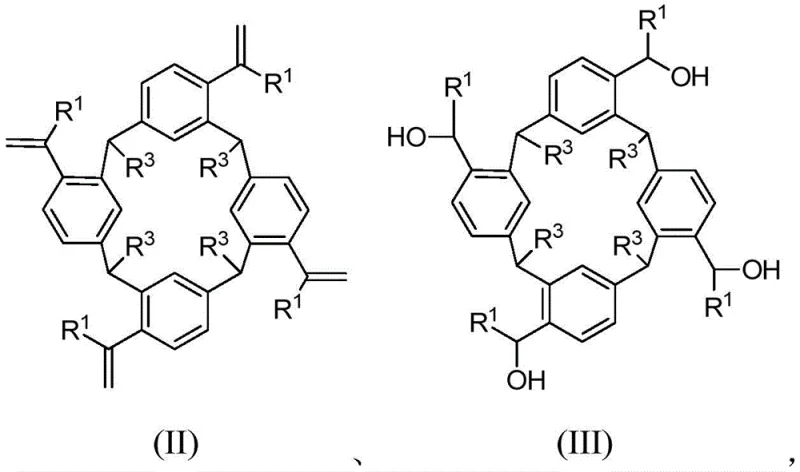
By employing acids such as trifluoromethanesulfonic acid or methanesulfonic acid in solvents like 1,2-dichloroethane or dichloromethane, the reaction proceeds smoothly at temperatures ranging from 0°C to 40°C. This represents a drastic improvement over harsher conventional methods, significantly reducing energy consumption and equipment stress. The ability to generate the target hydrogenated ring-band [8] aromatic hydrocarbon directly from accessible precursors ensures a more streamlined workflow, facilitating cost reduction in specialty chemical manufacturing while maintaining high structural integrity and purity essential for downstream applications.
Mechanistic Insights into Intramolecular Friedel-Crafts Alkylation
The core of this synthetic innovation lies in the acid-catalyzed intramolecular Friedel-Crafts alkylation mechanism, which effectively closes the macrocyclic ring. The process begins with the activation of the alkene or alcohol functionalities on the calix[4]resorcinarene backbone by a strong Brønsted acid. For instance, when using trifluoromethanesulfonic anhydride or trifluoromethanesulfonic acid, the electrophilic species generated attacks the electron-rich aromatic rings positioned strategically within the precursor molecule. This electrophilic aromatic substitution creates new carbon-carbon bonds that bridge the gaps between the phenolic units, locking the molecule into the desired belt-like conformation. The choice of solvent plays a critical role here; 1,2-dichloroethane is preferred for formula (II) precursors to ensure adequate solubility and reaction kinetics at 25°C to 50°C, whereas dichloromethane is optimal for formula (III) precursors at 0°C.
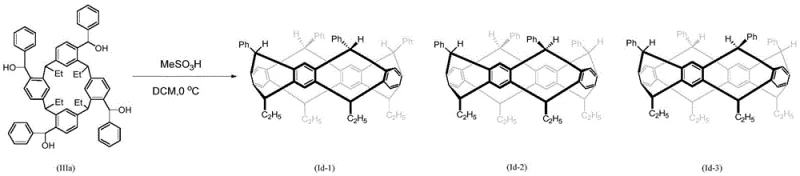
Furthermore, the reaction conditions allow for the generation of specific stereoisomers, which is crucial for tuning the host-guest chemistry properties. As demonstrated in the preparation of compounds Id-1, Id-2, and Id-3, the cyclization of formula (IIIa) with methanesulfonic acid at 0°C yields a mixture of stereoisomers that can be separated via preparative thin-layer chromatography. This level of control indicates that the transition states during the ring-closing step are sensitive to the steric environment created by the R groups, such as phenyl or substituted phenyl moieties. Understanding this mechanistic nuance allows chemists to predict and optimize the ratio of isomers, ensuring that the final product possesses the exact cavity dimensions and electronic characteristics required for recognizing specific organic guests like 1,2-dichloroethane or ethanol.
How to Synthesize Hydrogenated Ring-Band [8] Arene Efficiently
Implementing this synthesis requires careful attention to reagent quality and temperature control to maximize the yield of the cyclized product. The process generally involves dissolving the precursor in the specified solvent, cooling the mixture to the recommended temperature, and slowly adding the acid catalyst to manage the exotherm. Following the reaction, standard aqueous workup procedures involving extraction with dichloromethane and washing with saturated brine are sufficient to isolate the crude product. Purification is typically achieved through column chromatography using silica gel and petroleum ether/dichloromethane mixtures. For detailed operational parameters and specific stoichiometric ratios derived from the patent examples, please refer to the standardized guide below.
- Prepare the precursor compound of formula (II) or (III) via cross-coupling or Grignard addition.
- Dissolve the precursor in an organic solvent such as 1,2-dichloroethane or dichloromethane.
- Add an acid catalyst like trifluoromethanesulfonic acid or methanesulfonic acid at controlled temperatures between 0°C and 40°C to induce cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this synthetic route offers substantial strategic benefits beyond mere technical feasibility. The reliance on cheap and easily available raw materials, specifically auxiliary cup[4]resorcinol arene derivatives, mitigates the risk of raw material shortages that often plague specialized chemical supply chains. Since the key cyclization step does not require expensive transition metal catalysts like palladium or platinum in the final stage, the overall cost of goods sold is significantly optimized. This elimination of precious metals also simplifies the purification process, as there is no need for costly and time-consuming heavy metal scavenging steps, thereby enhancing the throughput of the manufacturing facility.
- Cost Reduction in Manufacturing: The process utilizes common mineral acids and organic solvents that are commodity chemicals, ensuring stable pricing and easy sourcing globally. By avoiding complex multi-step sequences involving rare earth catalysts in the final ring-closing step, the production cost is drastically simplified. The mild reaction temperatures further contribute to energy savings, as there is no need for high-pressure reactors or extreme heating systems. This economic efficiency translates directly into a more competitive pricing structure for the final high-purity supramolecular hosts, making them viable for large-scale industrial separation applications.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate a range of temperatures from -40°C to 150°C depending on the specific derivative, ensures consistent production outcomes even with minor variations in processing. The stability of the obtained products in air means that storage and transportation logistics are less stringent compared to air-sensitive organometallic intermediates. This stability reduces the lead time for high-purity macrocyclic intermediates, as special inert atmosphere packaging is not required for the final product, allowing for faster delivery to end-users in the pharmaceutical and materials sectors.
- Scalability and Environmental Compliance: The use of standard organic solvents like dichloromethane and 1,2-dichloroethane allows for straightforward scale-up using existing infrastructure in fine chemical plants. The waste streams generated are primarily acidic aqueous layers and organic solvents, which can be managed through established neutralization and distillation protocols. This aligns well with modern environmental compliance standards, reducing the regulatory burden associated with introducing new chemical entities. The ability to scale from gram quantities in the lab to commercial production without changing the fundamental chemistry ensures a seamless transition from R&D to full-scale manufacturing.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of integrating these compounds into their workflows, we have compiled answers to common inquiries based on the patent specifications. These questions address critical aspects such as reaction optimization, isomer separation, and application potential. Understanding these details is essential for R&D directors planning pilot runs and procurement managers assessing long-term supply viability. The following responses are grounded in the experimental data provided in the patent documentation.
Q: What are the optimal reaction conditions for the cyclization step?
A: The patent specifies using acids like trifluoromethanesulfonic acid or methanesulfonic acid in solvents like 1,2-dichloroethane at temperatures ranging from 0°C to 40°C for optimal yield and selectivity.
Q: Can this method produce specific stereoisomers?
A: Yes, by adjusting reaction conditions and purification methods such as preparative TLC, specific stereoisomers like Id-1, Id-2, and Id-3 can be separated and isolated.
Q: What are the primary applications of these macrocycles?
A: These compounds feature barrel-shaped cavities suitable for selective recognition and inclusion of organic small molecules, making them ideal for separation materials and supramolecular sensing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hydrogenated Ring-Band [8] Arene Supplier
As the demand for advanced functional materials grows, having a partner capable of delivering complex macrocyclic structures with precision is vital. NINGBO INNO PHARMCHEM leverages extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring these innovative hydrogenated ring-band [8] arenes to the market. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that ensure every batch meets the exacting standards required for supramolecular research and industrial separation processes. We understand the nuances of handling sensitive macrocycles and have the infrastructure to support both custom synthesis and bulk supply requirements.
We invite you to collaborate with us to explore how these unique host molecules can enhance your product portfolio. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating how our optimized synthesis route can lower your overall material costs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a reliable supply of these cutting-edge specialty chemicals for your next breakthrough project.
