Advanced Synthesis of Antibacterial Quinoline Derivatives for Global Pharmaceutical Supply Chains
Introduction to Patent CN101547907A and Therapeutic Potential
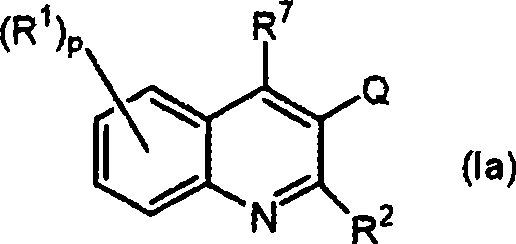
The pharmaceutical landscape is constantly evolving to combat the rising threat of antimicrobial resistance, particularly in the realm of mycobacterial infections. Patent CN101547907A introduces a novel class of substituted quinoline derivatives, defined by general formulas (Ia) and (Ib), which represent a significant advancement in the treatment of bacterial diseases. These compounds are specifically engineered to address the critical need for effective therapies against pathogenic mycobacteria, including Mycobacterium tuberculosis, Mycobacterium bovis, and various resistant strains that pose a severe burden on global health systems. The core innovation lies in the unique structural configuration featuring an unsaturated carbon chain connected at the 3-position of the quinoline nucleus, a distinct modification that differentiates these molecules from prior art quinoline derivatives.
This structural novelty is not merely academic; it translates directly into enhanced biological activity. The compounds demonstrate potent sterilization or bacteriostatic activity against a broad spectrum of pathogens, including Gram-positive microorganisms like Staphylococcus aureus (including MRSA) and Streptococcus pneumoniae. By targeting the F1F0 ATP synthase, specifically the c subunit of the F0 mixture, these derivatives exhaust bacterial cell ATP, leading to cell death. This mechanism of action is crucial for overcoming existing drug resistance, offering a viable pathway for developing next-generation antibiotics. For procurement and R&D teams, understanding the chemical backbone of these active pharmaceutical ingredients (APIs) is the first step toward securing a reliable supply chain for critical anti-infective medications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of functionalized quinoline derivatives has been plagued by several synthetic challenges that hinder efficient commercial manufacturing. Traditional methods often rely on harsh reaction conditions that can degrade sensitive functional groups or lead to poor regioselectivity. A common issue in prior art is the difficulty in controlling the stereochemistry of side chains attached to the heterocyclic core. Many conventional routes result in complex mixtures of enantiomers or diastereomers, necessitating expensive and yield-reducing purification steps such as chiral chromatography. Furthermore, older methodologies frequently employ transition metal catalysts that are not only costly but also introduce the risk of heavy metal contamination, requiring stringent and expensive removal processes to meet pharmaceutical purity standards.
Another significant bottleneck in conventional synthesis is the formation of the carbon-carbon double bond in the side chain. Standard elimination reactions often lack specificity, producing unpredictable ratios of E and Z isomers. This lack of stereochemical control complicates the biological evaluation process, as different isomers may exhibit vastly different pharmacokinetic profiles and potency levels. Consequently, manufacturers face increased lead times and higher production costs when attempting to isolate the therapeutically active isomer from a crude reaction mixture. These inefficiencies create substantial vulnerabilities in the supply chain, making it difficult to scale production to meet the demands of global health initiatives targeting tuberculosis and other resistant infections.
The Novel Approach
The methodology disclosed in CN101547907A offers a transformative solution to these longstanding synthetic hurdles. The novel approach centers on a highly efficient dehydration strategy that converts hydroxy-intermediates directly into the desired unsaturated quinoline derivatives. By utilizing specific dehydrating agents such as polyphosphoric acid (PPA) or thionyl chloride (SOCl2) in the presence of suitable bases like pyridine or triethylamine, the process achieves superior control over the reaction outcome. This method significantly reduces the formation of unwanted byproducts and allows for the selective preparation of specific stereoisomers, thereby minimizing the need for complex downstream purification.
Moreover, this synthetic route is designed with scalability in mind. It leverages readily available starting materials and reagents that are compatible with large-scale industrial reactors. The ability to synthesize the core quinoline structure and attach the functionalized side chain through a streamlined sequence of lithiation and nucleophilic addition ensures high overall yields. This robustness is essential for cost reduction in pharmaceutical intermediate manufacturing, as it lowers the cost of goods sold (COGS) and enhances the economic viability of producing these life-saving drugs. For supply chain managers, this translates to a more predictable and reliable sourcing strategy, reducing the risk of production delays caused by synthetic failures or purification bottlenecks.
Mechanistic Insights into Acid-Catalyzed Dehydration and Cyclization
To fully appreciate the technical superiority of this process, one must examine the mechanistic details of the key transformation steps. The synthesis begins with the preparation of quinoline intermediates, often achieved through Vilsmeier-Haack formylation followed by cyclization. However, the critical step that defines the novelty of this patent is the dehydration of the beta-hydroxy ketone intermediates (Formula II-a, II-b, etc.) to form the alpha,beta-unsaturated system found in the final products (Formula Ia, Ib). When polyphosphoric acid is used, the mechanism likely involves the protonation of the hydroxyl group, facilitating its departure as water and generating a carbocation intermediate. Subsequent elimination of a proton from the adjacent carbon yields the double bond. The choice of acid and reaction temperature allows chemists to influence the thermodynamic stability of the resulting isomer, favoring the formation of the more stable E-isomer in many cases.
Alternatively, the use of thionyl chloride (SOCl2) proceeds through a different mechanistic pathway involving the formation of a chlorosulfite ester intermediate. This activation of the hydroxyl group makes it an excellent leaving group, promoting elimination under milder conditions compared to strong mineral acids. This versatility is a major advantage for R&D directors, as it provides options to optimize the process based on the specific substitution pattern of the molecule. For instance, acid-sensitive groups on the quinoline ring might tolerate the SOCl2 conditions better than the harsh environment of refluxing PPA. Understanding these nuances is vital for process chemists aiming to replicate or scale this chemistry, ensuring that the integrity of the complex molecular architecture is maintained throughout the synthesis.
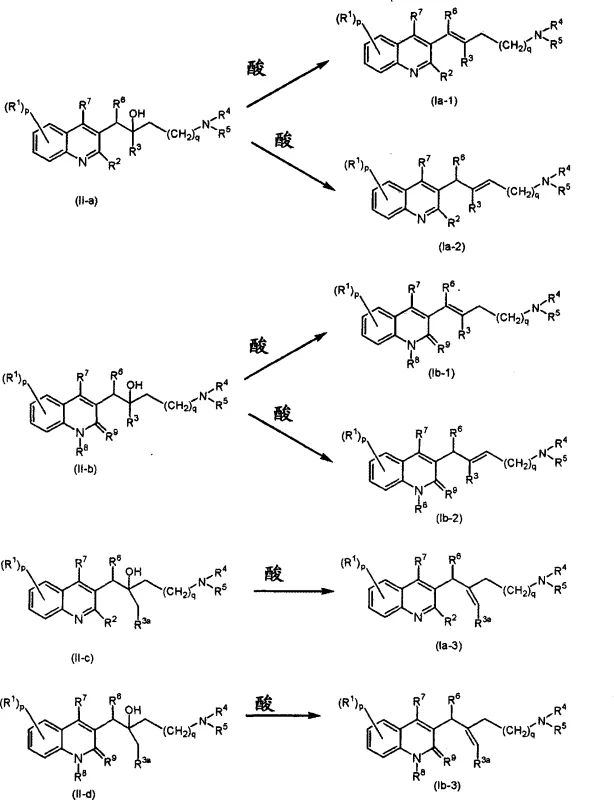
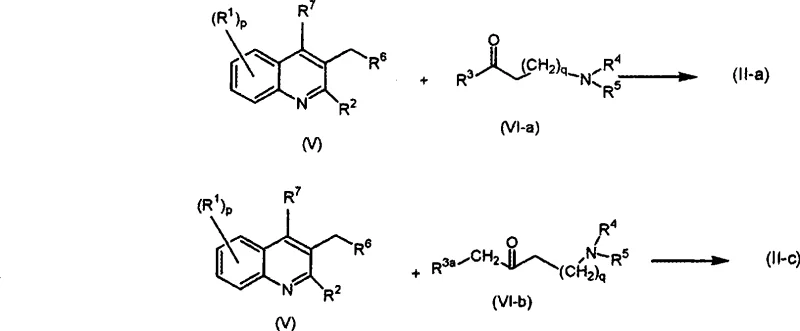
Furthermore, the construction of the side chain itself involves sophisticated organometallic chemistry. The patent describes the use of n-butyllithium (nBuLi) to deprotonate the quinoline core at the 3-position, generating a highly reactive nucleophile. This species then attacks a functionalized ketone, forging the critical carbon-carbon bond that links the heterocyclic core to the amino-alkyl side chain. This lithiation strategy is highly effective but requires precise temperature control, typically ranging from -20°C to -70°C, to prevent side reactions and ensure high regioselectivity. The ability to execute this step reliably on a large scale demonstrates a high level of process maturity. By mastering these low-temperature organometallic transformations, manufacturers can access a wide library of derivatives by simply varying the ketone component, thus accelerating the discovery of new drug candidates with improved efficacy profiles.
How to Synthesize Antibacterial Quinoline Derivatives Efficiently
The synthesis of these high-value antibacterial intermediates requires a disciplined approach to reaction engineering and purification. The process generally follows a convergent strategy where the quinoline core and the side chain are prepared separately before being coupled. Key to success is the strict control of stoichiometry and temperature during the lithiation step, as well as the careful selection of the dehydration agent to maximize the yield of the desired stereoisomer. Following the coupling reaction, the crude product typically undergoes aqueous workup and extraction, followed by purification via silica gel chromatography or crystallization. The detailed standardized synthesis steps outlined below provide a roadmap for replicating this chemistry in a GMP-compliant environment, ensuring consistency and quality in every batch produced.
- Preparation of quinoline intermediates via Vilsmeier-Haack formylation and cyclization of substituted anilines.
- Lithiation of the quinoline core using n-butyllithium followed by nucleophilic addition to functionalized ketones.
- Dehydration of the resulting hydroxy-intermediates using polyphosphoric acid or thionyl chloride to form the final unsaturated side chain.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the significant simplification of the manufacturing process. By eliminating the need for expensive and scarce transition metal catalysts often used in cross-coupling reactions, the process drastically reduces raw material costs. This shift to more commodity-grade reagents like thionyl chloride and polyphosphoric acid not only lowers the direct cost of production but also mitigates supply risk associated with specialized catalysts that may have long lead times or volatile pricing. Consequently, this leads to substantial cost savings in the overall manufacturing budget, allowing for more competitive pricing of the final API.
Enhanced supply chain reliability is another critical benefit derived from this technology. The robustness of the synthetic route means that production campaigns are less likely to suffer from batch failures due to sensitive reaction conditions. The intermediates generated are stable and can be isolated and stored if necessary, providing flexibility in production scheduling. This stability is crucial for maintaining continuous supply lines, especially for essential medicines where interruptions can have severe public health consequences. Furthermore, the high selectivity of the dehydration step reduces the burden on purification units. Less solvent and fewer chromatography cycles are required to achieve the necessary purity specifications, which in turn reduces waste generation and environmental impact. This aligns with modern green chemistry principles and helps manufacturers meet increasingly stringent environmental compliance regulations without compromising on output volume.
Scalability is inherently built into this process design. The reaction conditions are compatible with standard stainless steel reactors found in most fine chemical manufacturing facilities. There is no requirement for exotic high-pressure equipment or specialized containment systems beyond standard safety protocols for handling corrosive acids and reactive organolithiums. This ease of scale-up ensures that production can be ramped up quickly from pilot plant quantities to multi-ton commercial volumes to meet surging demand. For partners looking to secure a long-term supply of these critical intermediates, this scalability guarantees that the supplier can grow alongside their needs, ensuring continuity of supply from clinical trial phases through to full commercial launch.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these quinoline derivatives. These answers are derived directly from the technical disclosures within the patent documentation, providing clarity on the capabilities and limitations of the described technology. Understanding these details helps stakeholders make informed decisions regarding partnership and procurement strategies.
Q: What is the primary therapeutic application of these quinoline derivatives?
A: These compounds are designed primarily for treating bacterial infections, with specific efficacy against Mycobacterium tuberculosis, including multiple drug-resistant (MDR-TB) strains, as well as Staphylococcus and Streptococcus species.
Q: How does the novel synthesis route improve upon conventional methods?
A: The patented process utilizes specific dehydration conditions (PPA or SOCl2) that allow for better control over stereochemistry (E/Z isomers) and avoid the formation of complex enantiomeric mixtures often seen in traditional quinoline functionalization.
Q: Are these intermediates scalable for commercial production?
A: Yes, the synthesis relies on standard industrial reagents such as thionyl chloride, polyphosphoric acid, and n-butyllithium, which facilitates straightforward scale-up from laboratory to multi-ton commercial manufacturing without requiring exotic catalysts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Antibacterial Quinoline Derivatives Supplier
As the global demand for effective treatments against resistant bacterial infections continues to rise, securing a dependable source of high-quality intermediates is paramount. NINGBO INNO PHARMCHEM stands ready to support your development and commercialization goals with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of this synthesis, including low-temperature lithiation and corrosive acid handling, ensuring that every batch meets stringent purity specifications. Our rigorous QC labs employ advanced analytical techniques to verify the identity and stereochemical purity of each lot, guaranteeing that the material you receive is fit for purpose in your drug development pipeline.
We invite you to engage with our technical procurement team to discuss how we can tailor our manufacturing capabilities to your specific project needs. Whether you require a Customized Cost-Saving Analysis for your current supply chain or need specific COA data and route feasibility assessments for new analogues, our experts are available to provide the insights you need. By partnering with us, you gain access to a supply chain that is not only cost-effective but also resilient and compliant with international quality standards. Let us help you accelerate your journey from discovery to market with our proven expertise in complex heterocyclic chemistry.
