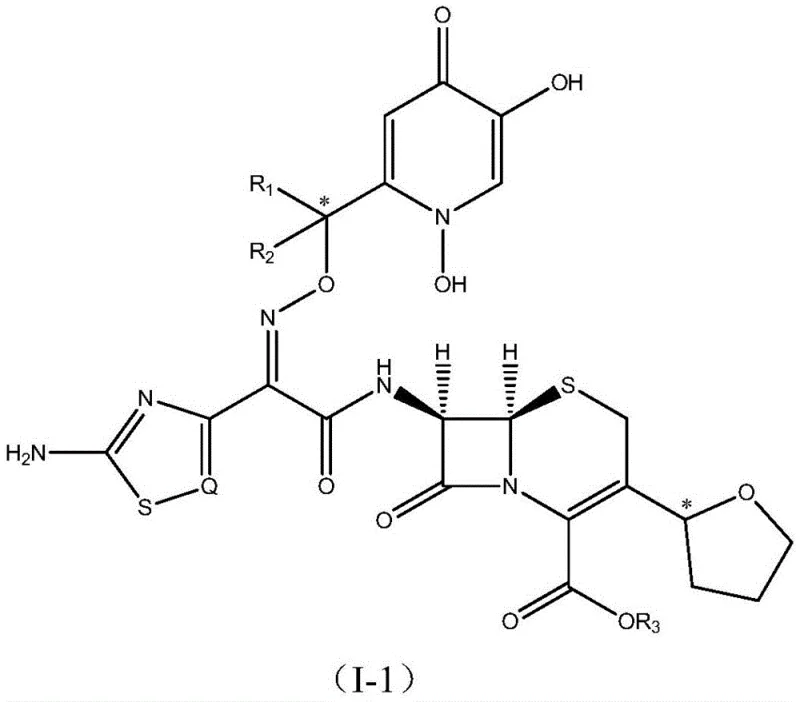
Advanced Cephalosporin-Siderophore Conjugates: Overcoming Resistance via Trojan Horse Strategy
The escalating crisis of antimicrobial resistance, particularly among Gram-negative pathogens, necessitates the development of innovative therapeutic strategies that bypass traditional resistance mechanisms. Patent CN111620893A introduces a groundbreaking class of C-3 tetrahydrofuran substituted cephalosporin-siderophore conjugates designed to address this urgent medical challenge through a sophisticated "Trojan Horse" approach. These novel compounds integrate a cefovecin-derived active fragment with specific siderophore moieties, such as dihydroxypyridone or catechol derivatives, to exploit the iron-uptake systems of bacteria. By mimicking essential nutrients, these conjugates facilitate active transport across the impermeable outer membranes of resistant strains, effectively delivering the antibiotic payload directly into the periplasmic space where it can inhibit cell wall synthesis. This dual-functionality not only restores susceptibility in multidrug-resistant organisms but also leverages the inherent beta-lactamase stability provided by the chiral tetrahydrofuran ring at the C-3 position.
The significance of this technology extends beyond mere structural modification; it represents a paradigm shift in how we approach the treatment of infections caused by formidable pathogens like Pseudomonas aeruginosa and Klebsiella pneumoniae. Traditional cephalosporins often fail against these organisms due to reduced outer membrane permeability and the presence of efflux pumps, but the siderophore conjugation strategy effectively circumvents these barriers. The patent details a comprehensive library of compounds, including various stereoisomers and side-chain variations, demonstrating robust in vitro activity against clinically isolated resistant strains. For pharmaceutical developers and procurement specialists, understanding the synthesis and potential of these intermediates is crucial for securing the next generation of veterinary and human antibiotics that can withstand the evolving landscape of bacterial defense mechanisms.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Conventional cephalosporin antibiotics, while historically effective, face diminishing returns against modern Gram-negative bacteria due to sophisticated resistance mechanisms that limit drug accumulation within the cell. The primary obstacle is the outer membrane of Gram-negative bacteria, which acts as a formidable permeability barrier, preventing hydrophilic antibiotics from reaching their penicillin-binding protein targets in sufficient concentrations. Furthermore, the overexpression of efflux pumps actively expels drugs that manage to enter the cell, while the production of beta-lactamases enzymatically degrades the antibiotic before it can exert its effect. Traditional methods of modifying cephalosporins often focus solely on enhancing beta-lactamase stability or affinity for binding proteins, neglecting the critical issue of cellular uptake. Consequently, even potent molecules fail clinically because they cannot penetrate the bacterial fortress, leading to treatment failures in cases involving ESBL-producing E. coli or carbapenem-resistant Enterobacteriaceae. This limitation underscores the need for a delivery system that can actively breach these defenses rather than passively relying on diffusion.
The Novel Approach
The novel approach detailed in the patent utilizes a siderophore-drug conjugate strategy that fundamentally alters the uptake mechanism of the antibiotic by hijacking the bacteria's own iron acquisition systems. Bacteria require iron for survival and have evolved high-affinity siderophore receptors to scavenge this scarce nutrient from the host environment; by covalently linking a siderophore to the cephalosporin core, the resulting conjugate is recognized as an iron source and actively transported into the cell. This active transport mechanism bypasses the porin channels that are often downregulated in resistant strains, ensuring high intracellular concentrations of the drug. Additionally, the specific incorporation of a C-3 tetrahydrofuran ring via a carbon-carbon bond provides exceptional stability against beta-lactamases, addressing the enzymatic degradation pathway simultaneously. This dual-pronged attack—enhancing uptake while protecting the pharmacophore—results in compounds that exhibit potent bactericidal activity against strains that are completely impervious to standard third-generation cephalosporins, offering a viable solution for treating complex multidrug-resistant infections.
Mechanistic Insights into Siderophore-Mediated Active Transport
The mechanistic efficacy of these conjugates relies on the precise molecular recognition between the siderophore moiety and the specific outer membrane receptors of Gram-negative bacteria, such as FhuA or FepA. Upon exposure to the extracellular environment, the dihydroxypyridone or catechol group on the conjugate chelates ferric iron (Fe3+) to form a stable complex that mimics natural siderophore-iron complexes. This biomimetic complex is then bound by the TonB-dependent transporters on the bacterial surface, triggering a conformational change that allows the entire conjugate to traverse the outer membrane into the periplasm. Once inside, the acidic environment or specific periplasmic esterases may facilitate the release of the active cephalosporin species, although the conjugate itself often retains activity. The presence of the tetrahydrofuran ring at the C-3 position is critical here, as it sterically hinders the access of beta-lactamases to the beta-lactam ring, thereby prolonging the half-life of the drug within the periplasmic space. This intricate interplay between transport kinetics and enzymatic stability ensures that the drug reaches its target site in an active form, effectively neutralizing the bacteria's primary defense strategies.
Furthermore, the structural diversity allowed by the variable R groups and the stereochemistry at the tetrahydrofuran linkage enables fine-tuning of the pharmacokinetic and pharmacodynamic properties to match specific pathogen profiles. The patent highlights that different siderophore types (catechol vs. hydroxypyridone) interact with distinct receptor sets, potentially broadening the spectrum of activity across different genera of bacteria. For instance, catechol-based siderophores might target Pseudomonas species more effectively, while hydroxypyridone derivatives could show superior uptake in Enterobacteriaceae. This modularity allows for the rational design of second-line agents tailored to specific resistance patterns observed in clinical settings. The stability of the oxime linkage connecting the side chain to the beta-lactam nucleus is also optimized to prevent premature hydrolysis in plasma, ensuring that the conjugate remains intact until it reaches the infection site. Such detailed mechanistic control is essential for developing robust therapeutics that can withstand the harsh physiological conditions of the host while maintaining potency against resilient pathogens.
How to Synthesize Cefovecin-Siderophore Conjugates Efficiently
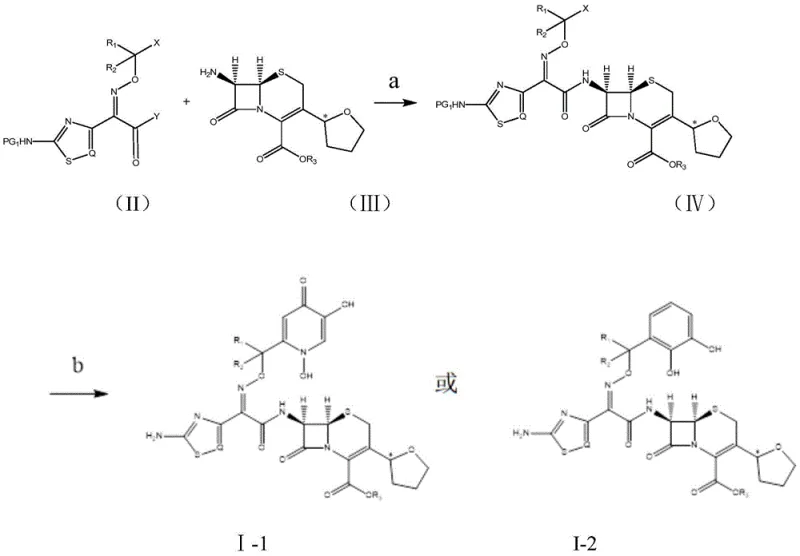
The synthesis of these advanced conjugates involves a convergent strategy that couples a protected siderophore acid intermediate with a C-3 tetrahydrofuran substituted cephalosporin core, followed by global deprotection to reveal the active functional groups. The process begins with the preparation of the siderophore acid, where hydroxyl groups are protected as benzhydryl ethers to prevent side reactions during subsequent coupling steps, ensuring high regioselectivity. The activation of the carboxylic acid on the siderophore fragment is typically achieved using peptide coupling reagents like HATU in the presence of a mild base, facilitating amide bond formation with the 7-amino group of the cephalosporin nucleus under controlled temperatures to preserve the stereochemical integrity of the beta-lactam ring. The final step employs trifluoroacetic acid and a scavenger like triethylsilane to remove the acid-labile protecting groups, yielding the free hydroxyl and amino functionalities required for biological activity. This route is designed to be scalable and robust, minimizing the formation of difficult-to-remove impurities that could compromise the safety profile of the final pharmaceutical product.
- Preparation of protected siderophore acids (dihydroxypyridone or catechol derivatives) via protection, Michael addition, and condensation reactions.
- Coupling of the protected siderophore acid with the C-3 tetrahydrofuran substituted cephalosporin core using HATU and base in DMSO.
- Final deprotection of benzhydryl and trityl groups using trifluoroacetic acid and triethylsilane to obtain the target active compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this siderophore-conjugate technology offers substantial strategic advantages by diversifying the portfolio of available antibiotic intermediates with high-value, differentiation-driven products. The synthesis route described in the patent utilizes readily available starting materials and standard organic transformations, which mitigates the risk of supply chain disruptions associated with exotic reagents or highly specialized catalytic processes. The modular nature of the synthesis allows for the parallel production of various analogues, enabling manufacturers to respond rapidly to emerging resistance patterns without requiring entirely new manufacturing lines. This flexibility translates into enhanced supply chain reliability, as producers can switch between different siderophore variants or cephalosporin cores based on raw material availability and market demand. Moreover, the high yields reported in the experimental examples for key coupling and deprotection steps suggest a cost-effective manufacturing process that minimizes waste and maximizes throughput, contributing to overall cost reduction in antibiotic intermediate manufacturing without compromising on quality standards.
- Cost Reduction in Manufacturing: The synthetic pathway leverages efficient coupling reactions and high-yielding deprotection steps that significantly reduce the consumption of expensive reagents and solvents per kilogram of product. By avoiding the use of transition metal catalysts that require rigorous removal and validation, the process simplifies downstream purification, thereby lowering operational expenditures associated with waste treatment and quality control testing. The robustness of the protection group strategy ensures minimal formation of by-products, which further reduces the need for complex chromatographic separations that often drive up production costs. Consequently, manufacturers can achieve a more favorable cost structure, making these advanced intermediates commercially viable for both veterinary and human pharmaceutical applications while maintaining healthy margins.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals for the construction of the siderophore and cephalosporin fragments ensures a stable supply of raw materials, reducing vulnerability to geopolitical or logistical shocks that often affect specialized fine chemical supply chains. The synthetic steps are performed under mild conditions that are easily transferable between different manufacturing sites, facilitating geographic diversification of production capacity to mitigate regional risks. Additionally, the stability of the intermediates allows for stockpiling of key precursors, enabling just-in-time manufacturing of the final conjugates to meet fluctuating demand without long lead times. This resilience is critical for maintaining continuity of supply for essential antibiotics, ensuring that healthcare providers have consistent access to effective treatments even during periods of global instability.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are safe and manageable on a multi-kilogram to ton scale without requiring specialized high-pressure or cryogenic equipment. The use of aqueous workups and crystallization for purification minimizes the generation of hazardous organic waste, aligning with increasingly stringent environmental regulations and sustainability goals. The elimination of heavy metal catalysts further simplifies waste disposal and reduces the environmental footprint of the manufacturing process. This compliance with green chemistry principles not only avoids regulatory penalties but also enhances the brand reputation of suppliers as responsible partners in the pharmaceutical value chain, appealing to clients who prioritize sustainable sourcing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these cephalosporin-siderophore conjugates, providing clarity for stakeholders evaluating this technology for integration into their supply chains. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these details is essential for assessing the feasibility of scaling these compounds for commercial production and their potential impact on current treatment protocols for resistant bacterial infections.
Q: What is the primary mechanism of action for these cephalosporin-siderophore conjugates?
A: These conjugates utilize a 'Trojan Horse' strategy where the siderophore moiety binds to bacterial iron transport receptors, actively transporting the antibiotic across the outer membrane of Gram-negative bacteria to overcome permeability-based resistance.
Q: Why is the C-3 tetrahydrofuran substitution significant in this structure?
A: The C-3 tetrahydrofuran ring, linked via a C-C bond, enhances stability against beta-lactamases and contributes to broad-spectrum antibacterial activity, distinguishing it from traditional cephalosporin structures.
Q: Which resistant bacterial strains are targeted by this technology?
A: The compounds demonstrate potent activity against multidrug-resistant Gram-negative strains including ESBL-producing Escherichia coli, KPC2 Klebsiella pneumoniae, and IMP4 Pseudomonas aeruginosa.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cephalosporin-Siderophore Conjugate Supplier
As the demand for next-generation antibiotics capable of overcoming multidrug resistance intensifies, partnering with a technically proficient manufacturer is essential for securing a stable supply of high-quality intermediates. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex molecules like these siderophore-conjugated cephalosporins can be manufactured reliably and consistently. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch meets the exacting standards required for pharmaceutical and veterinary applications. We understand the critical nature of antibiotic supply chains and are committed to delivering products that support the development of life-saving therapies against resistant pathogens.
We invite you to engage with our technical procurement team to discuss your specific requirements and explore how our capabilities align with your project goals. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized synthetic routes can reduce your overall production costs while maintaining superior quality. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions about integrating these advanced cephalosporin-siderophore conjugates into your pipeline. Together, we can accelerate the availability of effective treatments and contribute to the global fight against antimicrobial resistance.
