Scaling High-Purity Nevirapine Intermediates: A Technical Breakthrough in Amide Bond Formation and Crystallization
Scaling High-Purity Nevirapine Intermediates: A Technical Breakthrough in Amide Bond Formation and Crystallization
The global demand for effective antiretroviral therapies continues to drive the need for robust, scalable supply chains for key pharmaceutical intermediates. Central to the synthesis of Nevirapine, a widely used Non-nucleoside Reverse Transcriptase Inhibitor (NNRTI), is the critical building block 2-chloro-N-(2-chloro-4-methylpyridin-3-yl) nicotinamide. Recent intellectual property developments, specifically patent CN115028577A, have unveiled a refined purification methodology that addresses long-standing challenges in yield optimization and impurity control. This technical insight report analyzes the mechanistic advantages of this novel approach, offering R&D directors and procurement leaders a clear pathway toward securing a reliable pharmaceutical intermediates supplier capable of delivering ultra-high purity materials. By leveraging a specific aqueous-alcohol recrystallization technique, this process eliminates the need for costly chromatographic separations, thereby significantly enhancing the economic viability of large-scale production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex heterocyclic amides like 2-chloro-N-(2-chloro-4-methylpyridin-3-yl) nicotinamide has been plagued by inefficient purification protocols that rely heavily on organic solvents or silica gel column chromatography. Traditional routes often struggle to remove structurally similar byproducts, such as unreacted amines or hydrolyzed acid chlorides, leading to purity levels that frequently fall short of the rigorous >99.9% specification required for API manufacturing. Furthermore, conventional methods often utilize单一 solvent systems that fail to differentiate effectively between the target molecule and lipophilic impurities, resulting in substantial product loss during the washing stages. These inefficiencies not only inflate the cost of goods sold (COGS) due to low overall yields but also introduce bottlenecks in the supply chain, as chromatographic processes are difficult to scale beyond kilogram quantities without specialized equipment. Consequently, manufacturers face extended lead times and inconsistent batch-to-batch quality, posing significant risks to the continuity of Nevirapine production.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN115028577A introduces a streamlined, two-stage crystallization strategy that leverages the differential solubility of the target compound in mixed aqueous-alcoholic systems. Instead of relying on expensive and hazardous pure organic solvents, this novel approach utilizes a precise mixture of ethanol and water (or methanol and water) to induce selective crystallization. The process begins with the formation of a crude product via standard acylation and amination, followed by a sophisticated purification step where the crude material is dissolved at elevated temperatures (65-78°C) and slowly cooled to 0-10°C. This thermal cycling, combined with the specific polarity of the ethanol-water binary solvent system, forces the target amide to crystallize out while leaving polar and non-polar impurities in the mother liquor. This shift from chromatographic purification to controlled recrystallization represents a paradigm shift in cost reduction in API manufacturing, enabling producers to achieve purities exceeding 99.90% with dramatically improved operational simplicity and throughput.
Mechanistic Insights into Amide Coupling and Selective Crystallization
The core chemical transformation involves a nucleophilic acyl substitution where 2-chloro-3-amino-4-methylpyridine attacks the electrophilic carbonyl carbon of 2-chloronicotinyl chloride. The presence of electron-withdrawing chlorine atoms on both pyridine rings modulates the reactivity, necessitating careful temperature control between 60-70°C to prevent hydrolysis of the acid chloride while ensuring complete conversion. The addition of an acid-binding agent, such as pyridine or triethylamine, is critical to scavenge the generated hydrogen chloride, preventing the protonation of the amino group which would render it non-nucleophilic. Following the reaction, the introduction of water acts as an anti-solvent to precipitate the crude amide. However, the true innovation lies in the subsequent purification mechanism, where the interplay of hydrogen bonding and hydrophobic interactions in the ethanol-water matrix dictates crystal lattice formation.
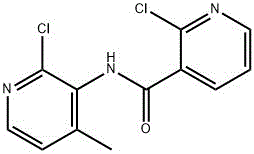
As illustrated in the molecular structure above, the steric environment around the amide bond and the specific positioning of the methyl and chloro substituents create a unique solubility profile that is exploited by this process. The purification step operates on the principle that at higher temperatures (70-75°C), the entropy of dissolution allows both the product and impurities to enter the solution phase. Upon controlled cooling to 0-10°C, the solubility of the target 2-chloro-N-(2-chloro-4-methylpyridin-3-yl) nicotinamide drops precipitously, favoring the formation of a highly ordered crystal lattice that inherently excludes impurity molecules. This phenomenon, known as lattice rejection, is optimized by the 1:1 ethanol-to-water ratio, which provides the ideal dielectric constant to maximize this selectivity. By maintaining the system at low temperatures for 2-4 hours, the process ensures the growth of large, uniform crystals that trap minimal solvent or impurities, thereby achieving the reported 99.98% purity without the need for further refining steps.
How to Synthesize 2-chloro-N-(2-chloro-4-methylpyridin-3-yl) nicotinamide Efficiently
Implementing this synthesis route requires strict adherence to the thermal and stoichiometric parameters defined in the patent to ensure reproducibility and safety. The process initiates with the activation of 2-chloronicotinic acid using thionyl chloride, followed by the coupling reaction in an aromatic solvent like toluene. The critical success factor, however, is the final recrystallization step, where the ratio of alcohol to water must be meticulously controlled to balance yield against purity. Operators must monitor the cooling rate carefully to avoid oiling out, which can trap impurities within the solid mass. For a detailed breakdown of the specific operational parameters, reagent grades, and safety protocols required to execute this synthesis in a GMP environment, please refer to the standardized technical guide below.
- Perform acylation of 2-chloronicotinic acid with thionyl chloride (1: 2.5-3.0 molar ratio) at 65-75°C for 1-4 hours to form 2-chloronicotinyl chloride.
- Conduct amination by reacting the acid chloride with 2-chloro-3-amino-4-methylpyridine (1: 1.05-1.25 ratio) in toluene with pyridine/triethylamine at 60-70°C.
- Purify the crude product by dissolving in ethanol/water (1: 1 ratio), heating to 70-75°C, cooling to 0-10°C for crystallization, and drying under vacuum.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this purification technology translates directly into enhanced supply security and reduced total cost of ownership. By eliminating the dependency on complex chromatographic columns and vast volumes of high-purity organic solvents, the manufacturing process becomes significantly more robust and less prone to operational failures. The use of commodity chemicals such as ethanol, water, and toluene ensures that raw material sourcing is stable and unaffected by the volatility of the specialty solvent market. Furthermore, the simplification of the workflow reduces the number of unit operations, which in turn lowers energy consumption and labor costs associated with batch processing. This streamlined approach facilitates a faster turnaround time from raw material intake to finished goods, allowing suppliers to respond more agilely to fluctuations in market demand for Nevirapine precursors.
- Cost Reduction in Manufacturing: The transition to a crystallization-based purification strategy removes the need for expensive silica gel and the associated disposal costs of hazardous solvent waste streams. By utilizing a recyclable ethanol-water system, the process drastically reduces the environmental footprint and the financial burden of waste treatment. Additionally, the high yield achieved through the optimized solvent ratio means that less raw material is wasted per kilogram of finished product, leading to substantial cost savings in the bill of materials. This efficiency gain allows for a more competitive pricing structure without compromising on the quality margins required for pharmaceutical grade intermediates.
- Enhanced Supply Chain Reliability: The reliance on easily obtainable, bulk-grade solvents mitigates the risk of supply disruptions that often plague specialized chemical supply chains. Since the process does not require exotic catalysts or sensitive reagents that have long lead times, production scheduling becomes more predictable and resilient. The robustness of the crystallization step also ensures consistent batch quality, reducing the likelihood of failed QC tests and the need for re-processing, which can otherwise cause significant delays in delivery schedules. This reliability is crucial for maintaining the continuous flow of materials needed for the uninterrupted production of life-saving antiretroviral medications.
- Scalability and Environmental Compliance: From a scale-up perspective, this method is inherently safer and easier to manage in large reactors compared to exothermic reactions requiring precise dosing or filtration of fine powders. The ability to perform the purification in standard glass-lined or stainless steel reactors without specialized filtration equipment simplifies the technology transfer from pilot plant to commercial scale. Moreover, the reduction in hazardous waste generation aligns with increasingly stringent global environmental regulations, reducing the compliance burden on manufacturing sites. This green chemistry aspect not only future-proofs the supply chain against regulatory changes but also enhances the corporate sustainability profile of the manufacturing partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and quality assurance of this specific Nevirapine intermediate. These insights are derived directly from the experimental data and process descriptions found in the underlying patent literature, providing a transparent view of the technology's capabilities. Understanding these details is essential for technical teams evaluating the feasibility of integrating this intermediate into their existing synthesis workflows or for quality assurance teams establishing incoming inspection criteria.
Q: What is the achieved purity level of the final intermediate using this method?
A: The patented purification method utilizing ethanol-water recrystallization consistently achieves a finished product purity of greater than 99.90%, with specific examples demonstrating up to 99.98% purity, meeting stringent pharmaceutical standards for Nevirapine synthesis.
Q: Why is the ethanol-to-water ratio critical in the purification step?
A: The solvent ratio directly impacts the solubility profile of the impurities versus the target compound. A 1:1 ratio of 95% ethanol to water provides the optimal balance, maximizing yield (approx. 94-97%) while ensuring high purity, whereas pure ethanol or excessive water significantly reduces recovery rates.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process is explicitly designed for industrial scalability. It utilizes common, low-cost solvents like toluene and ethanol, avoids complex chromatographic separation, and employs standard unit operations such as reflux, filtration, and vacuum drying, making it highly adaptable for metric-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-chloro-N-(2-chloro-4-methylpyridin-3-yl) nicotinamide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the consistency of your final API depends entirely on the quality of your starting materials. As a premier CDMO and supplier, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the laboratory-level efficiencies of this patented purification method are faithfully translated to industrial manufacturing. Our facilities are equipped with rigorous QC labs and advanced analytical instrumentation to verify that every batch meets stringent purity specifications, including the >99.9% benchmark established by this novel crystallization technique. We are committed to providing a secure, compliant, and high-quality supply of this critical intermediate to support the global fight against HIV/AIDS.
We invite potential partners to engage with our technical team to discuss how this optimized route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this more efficient purification method for your supply chain. We encourage you to contact our technical procurement team today to索取 specific COA data and route feasibility assessments, ensuring that your next production campaign is built on a foundation of chemical excellence and supply chain resilience.
