Advanced Synthesis of Linezolid Base Cationic Amphiphilic Compounds for Resistant Bacteria
The pharmaceutical industry faces an escalating crisis regarding multidrug-resistant bacterial infections, necessitating urgent innovation in antibiotic development as detailed in patent CN107382893B. Traditional oxazolidinone antibiotics like linezolid have served as critical therapeutics for Gram-positive infections, yet the emergence of resistant strains such as MRSA and VRE demands structural evolution beyond simple analogs. This patent discloses a novel series of linezolid base cationic amphiphilic compounds designed to overcome these resistance mechanisms through a dual-action pharmacological profile. By integrating a cationic hydrophilic head with a lipophilic tail onto the linezolid scaffold, these derivatives achieve enhanced membrane permeability and ribosomal binding affinity. The strategic modification preserves the essential oxazolidinone core responsible for protein synthesis inhibition while introducing physicochemical properties that disrupt bacterial cell integrity. This comprehensive approach represents a significant leap forward in medicinal chemistry, offering a robust pipeline for next-generation antibacterial agents capable of addressing the limitations of current standard-of-care treatments in clinical settings.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, efforts to modify linezolid have focused on replacing the morpholine moiety or altering the acetamide side chain, often resulting in complex synthetic routes with poor atom economy. Many prior art methods rely on multi-step heterocyclic constructions that require harsh reaction conditions, expensive catalysts, and rigorous purification protocols to remove toxic metal residues. These conventional pathways frequently suffer from low overall yields due to the accumulation of byproducts at each transformation stage, making commercial scale-up economically unviable for widespread distribution. Furthermore, the structural rigidity of many traditional analogs limits their ability to adapt to evolving bacterial defense mechanisms, leading to rapid cross-resistance development in clinical environments. The reliance on protecting group strategies that are difficult to remove cleanly also introduces significant impurity profiles, complicating regulatory approval processes for new drug candidates. Consequently, the industry has struggled to identify modification strategies that balance synthetic feasibility with enhanced biological potency against resistant pathogens.
The Novel Approach
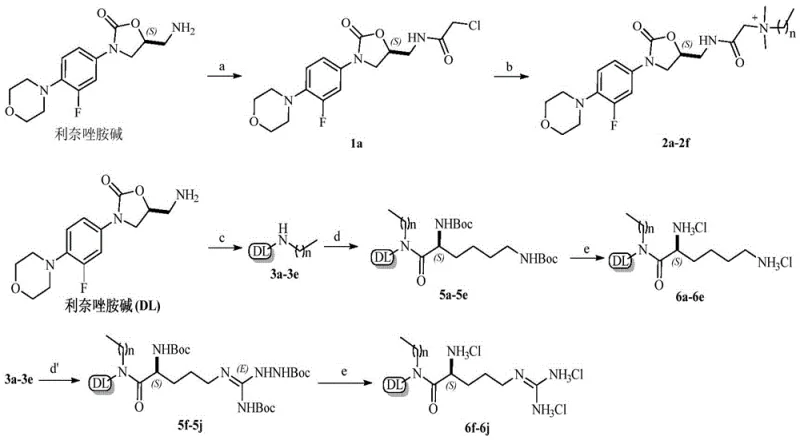 The methodology presented in CN107382893B utilizes a streamlined two-step or three-step reaction sequence that drastically simplifies the manufacturing workflow compared to legacy techniques. Starting from readily available linezolid base, the process employs mild acylation with chloroacetyl chloride followed by nucleophilic substitution with various amines to install the cationic functionality. This route avoids the use of transition metal catalysts, thereby eliminating the need for costly and time-consuming heavy metal scavenging steps during downstream processing. The reaction conditions are notably benign, often proceeding at room temperature or moderate heating in common solvents like acetonitrile or acetone, which facilitates easy solvent recovery and recycling. The versatility of this approach allows for the rapid generation of diverse libraries by simply varying the alkyl chain length or the amine component, accelerating the structure-activity relationship studies. Ultimately, this novel synthetic strategy provides a scalable and cost-effective platform for producing high-purity antibacterial intermediates suitable for industrial pharmaceutical applications.
The methodology presented in CN107382893B utilizes a streamlined two-step or three-step reaction sequence that drastically simplifies the manufacturing workflow compared to legacy techniques. Starting from readily available linezolid base, the process employs mild acylation with chloroacetyl chloride followed by nucleophilic substitution with various amines to install the cationic functionality. This route avoids the use of transition metal catalysts, thereby eliminating the need for costly and time-consuming heavy metal scavenging steps during downstream processing. The reaction conditions are notably benign, often proceeding at room temperature or moderate heating in common solvents like acetonitrile or acetone, which facilitates easy solvent recovery and recycling. The versatility of this approach allows for the rapid generation of diverse libraries by simply varying the alkyl chain length or the amine component, accelerating the structure-activity relationship studies. Ultimately, this novel synthetic strategy provides a scalable and cost-effective platform for producing high-purity antibacterial intermediates suitable for industrial pharmaceutical applications.
Mechanistic Insights into Cationic Amphiphilic Modification
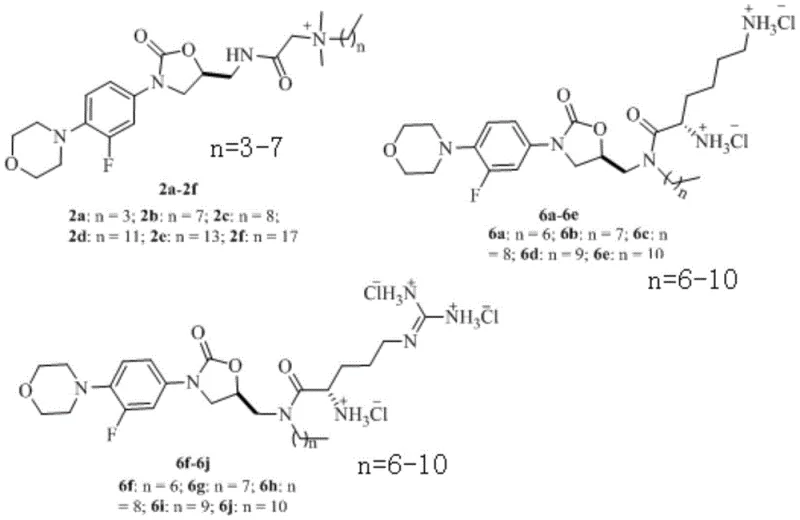 The therapeutic efficacy of these novel compounds stems from their unique amphiphilic architecture, which combines the specific ribosomal binding of the oxazolidinone core with the non-specific membrane disruption of the cationic tail. The oxazolidinone ring system binds to the 50S subunit of the bacterial ribosome, inhibiting the initiation complex formation of protein synthesis, a mechanism that remains effective even in some resistant strains. Simultaneously, the positively charged quaternary ammonium or amidine groups interact electrostatically with the negatively charged phospholipids of the bacterial cell membrane, increasing permeability and facilitating intracellular accumulation. This dual mechanism of action significantly raises the barrier for resistance development, as bacteria would need to mutate both ribosomal binding sites and membrane composition simultaneously to survive. The variation in alkyl chain length, denoted by n in the general structure, allows for fine-tuning of the lipophilicity to optimize the balance between potency and solubility. Such precise molecular engineering ensures that the compounds maintain high bioavailability while exerting potent bactericidal effects against a broad spectrum of Gram-positive and Gram-negative pathogens.
The therapeutic efficacy of these novel compounds stems from their unique amphiphilic architecture, which combines the specific ribosomal binding of the oxazolidinone core with the non-specific membrane disruption of the cationic tail. The oxazolidinone ring system binds to the 50S subunit of the bacterial ribosome, inhibiting the initiation complex formation of protein synthesis, a mechanism that remains effective even in some resistant strains. Simultaneously, the positively charged quaternary ammonium or amidine groups interact electrostatically with the negatively charged phospholipids of the bacterial cell membrane, increasing permeability and facilitating intracellular accumulation. This dual mechanism of action significantly raises the barrier for resistance development, as bacteria would need to mutate both ribosomal binding sites and membrane composition simultaneously to survive. The variation in alkyl chain length, denoted by n in the general structure, allows for fine-tuning of the lipophilicity to optimize the balance between potency and solubility. Such precise molecular engineering ensures that the compounds maintain high bioavailability while exerting potent bactericidal effects against a broad spectrum of Gram-positive and Gram-negative pathogens.
Controlling impurity profiles in the synthesis of these complex molecules is critical for ensuring patient safety and meeting stringent regulatory standards for pharmaceutical intermediates. The use of Boc-protected amino acids in the synthesis of series 6a-6j serves as a crucial strategy to prevent unwanted side reactions at the alpha-amino group during the coupling process. By masking the reactive amine functionality, the reaction specificity is directed exclusively towards the carboxylic acid condensation, minimizing the formation of oligomeric byproducts or regioisomers. Subsequent deprotection under mild acidic conditions using in-situ generated hydrogen chloride ensures the removal of the protecting group without degrading the sensitive oxazolidinone ring. This careful orchestration of protection and deprotection steps results in a final product with a clean impurity spectrum, reducing the burden on purification units and enhancing the overall yield of the active pharmaceutical ingredient. Rigorous analytical characterization using NMR and mass spectrometry confirms the structural integrity and purity of each batch, validating the robustness of the proposed synthetic methodology.
How to Synthesize Linezolid Base Cationic Compounds Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for reproducing these high-value antibacterial intermediates with consistent quality and yield. The process begins with the activation of the linezolid base followed by sequential functionalization to introduce the desired cationic properties. Operators should pay close attention to stoichiometry and temperature control during the acylation step to minimize hydrolysis of the acid chloride. Detailed standardized synthesis steps see the guide below for specific molar ratios and workup procedures that ensure optimal outcomes. Adhering to these validated parameters allows manufacturing teams to replicate the laboratory success on a larger production scale with minimal deviation. This structured approach minimizes trial-and-error experimentation, saving valuable time and resources during the process development phase.
- Prepare linezolid base amino chloroacetylated intermediate by reacting linezolid base with chloroacetyl chloride in acetone.
- Synthesize quaternary ammonium cationic compounds via nucleophilic substitution with N,N-dimethyl-N-alkylamine in acetonitrile.
- Condense with Boc-protected amino acids using HBTU/DIPEA catalysts followed by deprotection with in-situ hydrogen chloride.
Commercial Advantages for Procurement and Supply Chain Teams
The economic and logistical benefits of adopting this synthetic route extend far beyond the laboratory, offering tangible value to procurement managers and supply chain directors seeking optimization. By leveraging a process that relies on commodity chemicals and standard equipment, organizations can significantly reduce their exposure to volatile raw material markets and specialized infrastructure costs. The simplified purification requirements mean less solvent consumption and lower waste disposal fees, contributing to a more sustainable and cost-efficient operation. Furthermore, the robustness of the reaction conditions ensures high batch-to-batch consistency, reducing the risk of production delays caused by failed runs or out-of-specification results. These factors combine to create a resilient supply chain capable of meeting the demanding timelines of modern drug development programs without compromising on quality or compliance standards.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts from the synthetic route directly translates to substantial cost savings by removing the requirement for expensive palladium or nickel reagents and their associated removal resins. The utilization of commodity solvents such as acetone and acetonitrile, which are readily available in bulk quantities at stable market prices, further stabilizes the production budget against raw material volatility. Simple workup procedures involving filtration or rotary evaporation reduce the energy consumption and equipment time required for isolation, lowering the operational expenditure per kilogram of produced material. Additionally, the high yields reported in the patent examples indicate efficient atom economy, meaning less raw material is wasted as byproduct, which optimizes the overall material cost structure. These factors collectively contribute to a more lean manufacturing process that can offer competitive pricing for high-quality antibacterial intermediates in the global supply chain.
- Enhanced Supply Chain Reliability: The starting material, linezolid base, is a well-established intermediate with a mature and robust global supply network, ensuring consistent availability for large-scale production campaigns. The reagents required for the modification steps, including chloroacetyl chloride and various alkyl amines, are standard industrial chemicals produced by multiple vendors, mitigating the risk of single-source supply disruptions. The mild reaction conditions reduce the dependency on specialized high-pressure or cryogenic equipment, allowing for manufacturing flexibility across different facilities with standard glass-lined or stainless-steel reactors. This adaptability ensures that production schedules can be maintained even during periods of high demand or logistical constraints, providing partners with reliable lead times for their drug development programs. Consequently, procurement teams can secure long-term supply agreements with greater confidence in the continuity of material flow.
- Scalability and Environmental Compliance: The synthetic pathway is inherently scalable, as demonstrated by the successful execution of reactions in standard laboratory glassware which can be directly translated to pilot and commercial plant scales without fundamental process changes. The absence of hazardous heavy metals simplifies the waste treatment process, aligning with increasingly strict environmental regulations regarding effluent discharge and solid waste disposal in chemical manufacturing. Solvent recovery systems can be easily integrated to recycle acetonitrile and acetone, minimizing the environmental footprint and reducing the volume of hazardous waste requiring incineration. The use of aqueous workups where possible further reduces the reliance on chlorinated solvents, promoting greener chemistry principles throughout the production lifecycle. This commitment to sustainable manufacturing practices not only ensures regulatory compliance but also enhances the corporate social responsibility profile of the supply chain partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these linezolid derivatives. Answers are derived directly from the experimental data and technical disclosures within the patent documentation to ensure accuracy. Understanding these details helps stakeholders make informed decisions about integrating this technology into their existing portfolios. The responses cover aspects of mechanism, synthesis, and safety to provide a holistic view of the compound's potential.
Q: How do these derivatives overcome linezolid resistance?
A: The cationic amphiphilic structure disrupts bacterial cell membranes while the oxazolidinone core inhibits ribosomal protein synthesis, creating a dual mechanism that prevents resistance.
Q: What are the synthetic advantages of this method?
A: The process uses mild conditions, avoids transition metal catalysts, and employs simple workup procedures like filtration, making it highly scalable and cost-effective.
Q: Is the toxicity profile suitable for drug development?
A: In vitro erythrocyte toxicity tests demonstrate low hemolytic activity, indicating a favorable safety margin for further preclinical and clinical investigation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Linezolid Base Derivative Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production for global pharmaceutical clients. Our technical team is uniquely qualified to adapt the patented route for linezolid base cationic compounds to meet your specific stringent purity specifications and project timelines. We operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch meets the highest international standards for identity and purity. By partnering with us, you gain access to a wealth of process knowledge that ensures smooth technology transfer and reliable commercial supply. Our commitment to excellence makes us the preferred choice for companies seeking a dependable source of complex antibacterial intermediates.
We invite you to contact our technical procurement team to discuss your specific requirements and explore how we can support your drug development goals. Request a Customized Cost-Saving Analysis to understand the economic benefits of sourcing these intermediates through our optimized manufacturing channels. We are prepared to provide specific COA data and route feasibility assessments to demonstrate our capability to deliver on your promises. Let us help you accelerate your timeline to market with a supply partner dedicated to quality and innovation.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
