Advanced Synthesis of Pyrazole-Benzothiazole Bisamides for Commercial Agrochemical Applications
The agricultural and pharmaceutical sectors are constantly seeking novel chemical entities that offer dual-functionality and robust biological profiles. Patent CN102285979B introduces a significant advancement in this domain by disclosing a class of N-(2-(substituted benzothiazole-2-carbamoyl)-phenyl)-substituted pyrazole carboxamide compounds. These molecules represent a sophisticated fusion of pyrazole and benzothiazole pharmacophores linked via a bisamide scaffold, designed to target both plant viral pathogens and mammalian cancer cells. For R&D directors and procurement specialists, understanding the structural versatility and synthetic accessibility of these compounds is crucial for integrating them into new product pipelines. The general formula (I) described in the patent allows for extensive substitution at the R1, R2, and R3 positions, enabling the fine-tuning of physicochemical properties to meet specific regulatory and efficacy requirements.
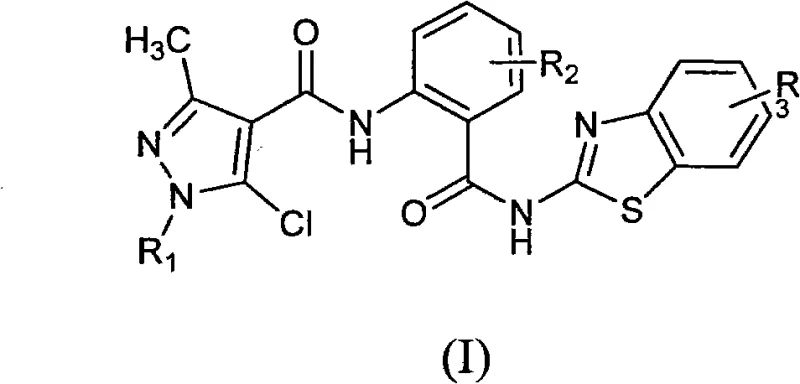
This technological breakthrough is particularly relevant for the development of next-generation agrochemical intermediates and pharmaceutical leads. The structural integrity of the bisamide linkage, combined with the electron-rich heterocyclic systems, provides a unique mechanism of action that differs from traditional single-target agents. As a reliable agrochemical intermediate supplier, recognizing the potential of such multi-heterocyclic systems allows for strategic positioning in the market. The patent details not only the biological potential but also a reproducible synthetic pathway, making it a viable candidate for commercial scale-up of complex bisamides. The ability to modulate substituents such as halogens, alkyl, and alkoxy groups offers a broad chemical space for optimization, ensuring that supply chains can adapt to evolving resistance patterns in agricultural pests or specific therapeutic needs in oncology.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing bisamide derivatives often suffer from inefficiencies related to step economy and purification challenges. Conventional methods for linking heterocyclic amines to benzoic acid derivatives frequently require harsh coupling reagents or transition metal catalysts that introduce heavy metal impurities, necessitating expensive downstream purification processes. Furthermore, older synthetic routes may lack the modularity required to easily swap out heterocyclic components, limiting the speed at which structure-activity relationship (SAR) studies can be conducted. In many historical contexts, the formation of the amide bond adjacent to sterically hindered heterocycles results in low yields and significant formation of side products, which complicates the isolation of high-purity intermediates. These limitations directly impact cost reduction in agrochemical manufacturing, as low overall yields and complex workups drive up the cost of goods sold (COGS). Additionally, the reliance on moisture-sensitive reagents in traditional protocols can pose safety risks and scalability issues, hindering the transition from laboratory benchtop to industrial reactor vessels.
The Novel Approach
The methodology outlined in CN102285979B overcomes these hurdles through a streamlined four-step sequence that prioritizes atom economy and operational simplicity. By utilizing a benzoxazinone intermediate, the process avoids the need for direct coupling of sterically hindered amines to carboxylic acids, instead leveraging a ring-opening strategy that proceeds under milder conditions. This approach significantly enhances the yield of the final coupling step, as demonstrated by the consistent formation of target compounds across various substituents. The use of common solvents like dichloromethane and acetonitrile, along with inexpensive bases such as triethylamine and potassium carbonate, ensures that the process remains economically viable for large-scale production. This novel route facilitates the commercial scale-up of complex bisamides by minimizing the number of isolation steps and reducing the generation of hazardous waste. For supply chain heads, this translates to a more robust and predictable manufacturing timeline, reducing lead time for high-purity agrochemical intermediates and ensuring a steady flow of material for formulation development.
Mechanistic Insights into Benzoxazinone-Mediated Amidation
The core of this synthetic innovation lies in the formation and subsequent reactivity of the benzoxazinone intermediate. In the third step of the synthesis, 2-substituted benzamidobenzoic acid undergoes cyclodehydration in the presence of acetic anhydride under reflux conditions. This transformation generates a highly electrophilic cyclic imide structure, specifically a benzo[d][1,3]oxazin-4-one derivative. The mechanistic advantage of this intermediate is its susceptibility to nucleophilic attack at the carbonyl carbon adjacent to the oxygen atom. When exposed to substituted benzothiazole amines in the presence of a base like potassium carbonate, the amine nucleophile attacks the carbonyl group, leading to the cleavage of the C-O bond in the oxazinone ring. This ring-opening reaction effectively installs the second amide linkage while simultaneously regenerating the carboxylic acid functionality on the central phenyl ring, although in the final product, it remains as part of the bisamide structure due to the specific connectivity.
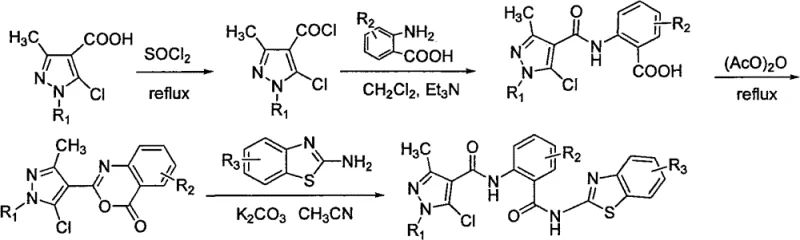
This mechanism is particularly advantageous for controlling impurity profiles, a key concern for R&D directors focused on purity and impurity spectra. The cyclization step tends to be clean, producing a solid intermediate that can be easily purified by recrystallization or washing, thereby removing unreacted starting materials before the final coupling. The subsequent ring-opening is driven by the stability of the resulting bisamide product and the release of ring strain. By avoiding direct activation of the carboxylic acid with reagents like thionyl chloride in the final step, the process minimizes the formation of symmetrical anhydrides or other acylation byproducts. The use of acetonitrile as a solvent in the final step provides a polar environment that solubilizes the ionic intermediates formed during the base-mediated reaction, ensuring homogeneous reaction conditions that promote consistent conversion. This level of mechanistic control is essential for maintaining batch-to-batch consistency, which is a critical metric for qualifying suppliers in the regulated agrochemical and pharmaceutical industries.
How to Synthesize N-(2-(substituted benzothiazole-2-carbamoyl)-phenyl)-substituted pyrazole carboxamides Efficiently
The synthesis of these high-value intermediates follows a logical progression that balances reactivity with safety. The process begins with the activation of the pyrazole acid, followed by sequential assembly of the molecular framework. Each step has been optimized in the patent examples to provide reliable yields and manageable physical forms, such as solids that are easy to filter and dry. The detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating these results.
- Preparation of 1-substituent-3-methyl-5-chloro-1H-pyrazole-4-carbonyl chloride via reflux with thionyl chloride.
- Acylation of substituted anthranilic acid with the prepared pyrazole acid chloride in dichloromethane using triethylamine.
- Cyclization of the resulting benzamide benzoic acid with acetic anhydride under reflux to form the benzoxazinone intermediate.
- Ring-opening reaction of the benzoxazinone with substituted benzothiazole amine in acetonitrile using potassium carbonate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers tangible strategic benefits beyond mere chemical novelty. The process is designed with scalability in mind, utilizing unit operations that are standard in fine chemical manufacturing facilities. This alignment with existing infrastructure reduces the capital expenditure required for technology transfer, allowing for faster time-to-market for new formulations containing these active ingredients. The reliance on commodity chemicals for reagents and solvents mitigates the risk of supply disruptions associated with specialized or proprietary catalysts.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of stoichiometric reagents like thionyl chloride and acetic anhydride significantly lowers the raw material costs. Furthermore, the ability to isolate stable solid intermediates allows for flexible scheduling of production batches, optimizing plant utilization rates. The high purity achieved through simple recrystallization steps reduces the need for costly chromatographic purification, directly contributing to substantial cost savings in the overall manufacturing budget. By streamlining the synthesis to four distinct steps, the process minimizes labor hours and energy consumption associated with prolonged reaction times or complex workups.
- Enhanced Supply Chain Reliability: The starting materials, including various substituted anthranilic acids and benzothiazole amines, are widely available from multiple global vendors, reducing dependency on single-source suppliers. This diversity in the supply base enhances resilience against market volatility and geopolitical disruptions. The robust nature of the reaction conditions, which tolerate minor variations in temperature and stoichiometry without significant yield loss, ensures consistent output even when scaling from pilot plants to full commercial production. This reliability is crucial for maintaining long-term contracts with downstream formulators who require guaranteed volumes of active intermediates.
- Scalability and Environmental Compliance: The synthetic pathway generates waste streams that are relatively straightforward to treat, primarily consisting of aqueous salts and organic solvents that can be recovered and recycled. The avoidance of heavy metals simplifies the environmental permitting process and reduces the burden of hazardous waste disposal. The exothermic nature of the acylation and cyclization steps can be managed with standard cooling systems, ensuring safe operation at larger scales. This environmental profile aligns with increasingly stringent global regulations on chemical manufacturing, positioning the production of these intermediates as a sustainable choice for eco-conscious corporate partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these bisamide compounds. The answers are derived directly from the experimental data and specifications provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: What are the primary biological activities of these pyrazole-benzothiazole bisamides?
A: According to patent CN102285979B, these compounds exhibit significant inhibitory activity against plant viruses such as Tobacco Mosaic Virus (TMV) and Cucumber Mosaic Virus (CMV), as well as anti-tumor activity against human breast cancer cells (Bcap-37).
Q: What is the key intermediate in the synthesis of these bisamides?
A: The synthesis relies on a critical benzoxazinone intermediate, formed by the cyclization of 2-substituted benzamidobenzoic acid with acetic anhydride, which subsequently undergoes ring-opening with benzothiazole amines.
Q: Are the raw materials for this synthesis commercially available?
A: Yes, the process utilizes readily available starting materials including substituted pyrazole acids, anthranilic acids, and benzothiazole amines, facilitating scalable production for industrial applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-(2-(substituted benzothiazole-2-carbamoyl)-phenyl)-substituted pyrazole carboxamides Supplier
The development of advanced agrochemical and pharmaceutical intermediates requires a partner with deep technical expertise and a commitment to quality. NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications for complex heterocyclic compounds. We understand the critical nature of impurity control in bisamide synthesis and employ advanced analytical techniques to ensure every batch meets the highest standards required for regulatory submission and commercial use.
We invite you to collaborate with us to leverage this patented technology for your product pipeline. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing capabilities can support your goals for cost reduction in agrochemical manufacturing and reliable supply chain management.
