Advanced Synthesis of Antiviral Diazacyclospirodiketopiperazine Alkaloid Derivatives for Commercial Scale
The pharmaceutical industry is currently facing an urgent demand for novel antiviral agents capable of addressing resistant strains of Respiratory Syncytial Virus (RSV), Herpes Simplex Virus (HSV-1), and Enterovirus 71 (EV71). Patent CN106795174B introduces a groundbreaking class of diazacyclospirodiketopiperazine alkaloid derivatives that offer a robust structural scaffold for developing next-generation therapeutics. This technology leverages a unique spirocyclic architecture that enhances molecular rigidity and binding affinity, potentially overcoming the limitations of existing linear antiviral compounds. For research and development teams, understanding the synthetic accessibility and structural versatility of these molecules is critical for accelerating drug discovery pipelines. The patent details a comprehensive library of compounds where substituents can be systematically varied to optimize pharmacokinetic profiles without compromising the core bioactivity. As a leading manufacturer, we recognize that translating such complex academic chemistry into reliable commercial supply chains requires deep process expertise and rigorous quality control standards.
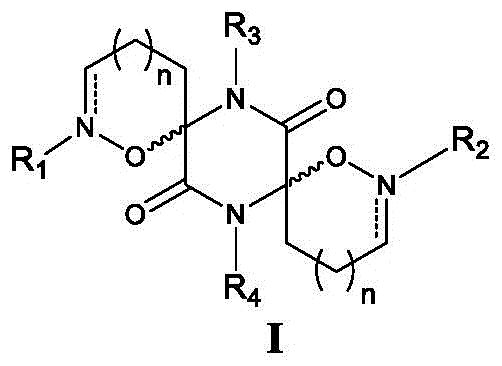
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis pathways for complex heterocyclic antiviral intermediates often suffer from poor atom economy and reliance on hazardous reagents that complicate waste management and increase production costs. Many conventional routes require multiple protection and deprotection steps to manage the reactivity of polyfunctional amino acid precursors, leading to significant yield erosion at each stage of the sequence. Furthermore, establishing the critical spiro-center in earlier methodologies frequently involved harsh acidic conditions or expensive transition metal catalysts that were difficult to remove to pharmaceutical grade standards. These inefficiencies create bottlenecks in the supply chain, resulting in longer lead times and higher volatility in pricing for key pharmaceutical intermediates. Additionally, the lack of modularity in older synthetic designs makes it challenging to rapidly generate analog libraries for structure-activity relationship studies, slowing down the overall drug development timeline. For procurement managers, these technical hurdles translate directly into increased risk of supply disruption and inflated raw material expenses.
The Novel Approach
The methodology outlined in CN106795174B presents a streamlined and modular strategy that significantly simplifies the construction of the diazacyclospirodiketopiperazine core. By utilizing readily available diamino acids such as ornithine or 2,4-diaminobutyric acid as starting materials, the process establishes the foundational diketopiperazine ring through efficient condensation reactions under controlled acidic conditions. The subsequent oxidative transformation of primary amines to oximes allows for a precise and high-yielding cyclization step mediated by silver catalysts, which effectively closes the isoxazoline rings to form the spiro-junction. This approach minimizes the need for extensive protecting group manipulation, thereby reducing the total number of unit operations and improving the overall mass balance of the synthesis. The flexibility to introduce diverse alkyl, acyl, or heteroaryl groups at the nitrogen positions post-cyclization enables rapid customization of the final active pharmaceutical ingredient properties. This technological advancement represents a significant leap forward in process chemistry, offering a more sustainable and cost-effective route for manufacturing high-value antiviral intermediates.
Mechanistic Insights into Silver-Catalyzed Oxidative Cyclization
The core innovation of this technology lies in the mechanistic elegance of the silver-catalyzed oxidative cyclization that constructs the spirocyclic framework. The reaction proceeds through the activation of the oxime functionality by silver species, facilitating an intramolecular nucleophilic attack that forms the N-O bond of the isoxazoline ring system. This transformation is highly sensitive to reaction parameters such as solvent polarity, temperature, and the specific choice of silver salt, with silver carbonate or silver oxide demonstrating superior performance in polar aprotic solvents like DMA or DMF. Understanding these mechanistic nuances is essential for R&D directors aiming to replicate or scale this chemistry, as minor deviations can impact the diastereoselectivity and purity of the final spiro-product. The patent data indicates that the reaction tolerates a wide range of functional groups, suggesting a robust catalytic cycle that is not easily poisoned by common impurities found in bulk chemical feedstocks. Moreover, the ability to control the stereochemistry at the spiro-center through the choice of chiral starting materials ensures that the biological activity can be fine-tuned at the molecular level. This level of mechanistic control provides a solid foundation for developing stable and reproducible manufacturing processes.
Impurity control is another critical aspect where this novel pathway excels, particularly regarding the management of over-oxidation byproducts and unreacted intermediates. The use of stoichiometric oxidants like m-CPBA or hydrogen peroxide is carefully balanced to ensure complete conversion of the amine precursors while minimizing the formation of nitrile oxides or other degradation products. Downstream purification strategies, including silica gel chromatography and recrystallization, are optimized to remove trace metal residues and organic impurities to meet stringent pharmaceutical specifications. The patent highlights that the resulting compounds exhibit high HPLC purity, often exceeding 98%, which reduces the burden on downstream formulation teams. For supply chain stakeholders, high intrinsic purity from the synthesis step means fewer processing delays and lower rejection rates during quality assurance testing. This focus on quality by design ensures that the material delivered for clinical or commercial use maintains consistent potency and safety profiles throughout its shelf life.
How to Synthesize Diazacyclospirodiketopiperazine Efficiently
Executing the synthesis of these complex alkaloid derivatives requires a disciplined approach to reaction monitoring and parameter control to ensure optimal yields and purity. The process begins with the careful selection of chiral or racemic amino acid feedstocks, followed by precise pH control during the initial condensation phase to prevent polymerization. Operators must maintain strict temperature profiles during the oxidative and cyclization stages to manage exotherms and ensure safe operation at scale. Detailed standard operating procedures cover the handling of silver catalysts and the workup protocols necessary to recover valuable metals and minimize environmental impact. The following guide outlines the critical operational milestones for successfully implementing this chemistry in a pilot or production environment.
- Condense 2,4-diaminobutyric acid or ornithine using acidic catalysts like KHSO4 to form the diketopiperazine precursor.
- Oxidize the primary amine groups of the precursor using m-CPBA or hydrogen peroxide to generate the dioxime intermediate.
- Perform silver-catalyzed cyclization in organic solvents like DMA or DMF to close the isoxazoline rings and form the spiro-core.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers substantial advantages in terms of cost structure and supply chain resilience for pharmaceutical manufacturers. The reliance on commodity chemicals such as amino acids and common organic solvents reduces dependency on exotic or single-source reagents that are prone to market volatility. By eliminating complex protection-deprotection sequences, the overall processing time is drastically simplified, allowing for faster batch turnover and improved capacity utilization in manufacturing facilities. This efficiency gain translates into significant cost savings in labor and utility consumption, making the final intermediates more competitive in the global marketplace. Furthermore, the robustness of the chemistry ensures consistent batch-to-batch quality, which is paramount for maintaining regulatory compliance and avoiding costly production stoppages. Procurement teams can leverage these process efficiencies to negotiate better terms and secure long-term supply agreements with greater confidence.
- Cost Reduction in Manufacturing: The streamlined synthesis eliminates the need for expensive protecting groups and reduces the total number of reaction steps, which directly lowers the cost of goods sold. By utilizing abundant raw materials like ornithine and standard oxidants, the process avoids the price fluctuations associated with specialized reagents. The high yields reported in the patent examples indicate efficient material utilization, minimizing waste disposal costs and maximizing output per unit of input. Additionally, the recovery and reuse of silver catalysts further contribute to economic viability by reducing the consumption of precious metals. These factors combine to create a lean manufacturing model that supports competitive pricing strategies without compromising on quality standards.
- Enhanced Supply Chain Reliability: Sourcing stability is significantly improved as the key starting materials are widely available from multiple global suppliers, mitigating the risk of single-point failures. The synthetic route does not rely on cryogenic conditions or highly specialized equipment, allowing for production across a broader network of qualified contract manufacturing organizations. This flexibility ensures that supply can be scaled up rapidly to meet surges in demand during epidemic outbreaks or seasonal peaks. The chemical stability of the intermediates also facilitates easier storage and transportation, reducing the logistical complexities and costs associated with cold chain requirements. Consequently, partners can maintain healthier inventory levels and respond more agilely to market dynamics.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily transferable from laboratory glassware to industrial reactors. The use of aqueous workups and standard organic solvents simplifies waste stream management and aligns with modern green chemistry principles. By avoiding persistent organic pollutants and minimizing heavy metal discharge through effective catalyst recovery, the manufacturing footprint is kept within strict environmental regulations. This commitment to sustainability not only reduces regulatory risk but also enhances the corporate social responsibility profile of the supply chain. Manufacturers can thus achieve commercial scale-up of complex pharmaceutical intermediates with confidence in their environmental stewardship and operational safety.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these antiviral intermediates. Answers are derived from the detailed experimental data and claims within the patent documentation to ensure accuracy and relevance. Understanding these aspects helps stakeholders make informed decisions about integrating this technology into their development programs. We encourage further discussion with our technical team to explore specific customization options for your project needs.
Q: What is the primary antiviral spectrum of these spirodiketopiperazine derivatives?
A: The compounds described in patent CN106795174B exhibit broad-spectrum activity, specifically showing strong inhibitory effects against Respiratory Syncytial Virus (RSV), Herpes Simplex Virus type 1 (HSV-1), and Enterovirus 71 (EV71).
Q: Are the synthesis routes scalable for commercial production?
A: Yes, the synthesis utilizes conventional reagents such as amino acids, standard oxidants, and silver catalysts in common organic solvents, making the process highly adaptable for kilogram-to-ton scale manufacturing.
Q: How is stereochemical purity controlled during the spiro-formation?
A: The process allows for the use of chiral starting materials like L-ornithine or D-ornithine, and the cyclization conditions preserve the stereocenters, enabling the production of specific enantiomers or racemates as required.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diazacyclospirodiketopiperazine Supplier
At NINGBO INNO PHARMCHEM, we possess the technical capability and infrastructure to bring this sophisticated chemistry from the patent literature to commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of antiviral intermediate meets the highest international standards. Our commitment to quality extends beyond mere compliance; we actively engage in process optimization to enhance yield and reduce environmental impact, delivering value at every stage of the partnership. By choosing us as your partner, you gain access to a wealth of chemical expertise dedicated to advancing your antiviral drug development goals.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Our experts are ready to provide specific COA data and route feasibility assessments to demonstrate how we can support your pipeline. Whether you require custom synthesis of specific analogs or large-scale production of the core scaffold, we are equipped to deliver solutions that drive your project forward. Let us collaborate to accelerate the availability of life-saving antiviral therapies to the global market.
