Advanced Phosphaphenanthrene Derivatives: Scalable Chiral Ligand Solutions for Industrial Asymmetric Synthesis
Advanced Phosphaphenanthrene Derivatives: Scalable Chiral Ligand Solutions for Industrial Asymmetric Synthesis
Introduction to Patent CN110669073B Technology
The pharmaceutical and fine chemical industries are constantly seeking more efficient and cost-effective pathways to access high-purity chiral intermediates, which are the foundational building blocks for modern drug discovery and development. Patent CN110669073B introduces a groundbreaking methodology for the synthesis of phosphaphenanthrene-type tertiary phosphine derivatives, addressing critical bottlenecks in the production of chiral catalysts. This technology leverages a novel structural design where the chiral center is positioned in close proximity to the reactive phosphorus atom, thereby enhancing catalytic activity and stereoselectivity in asymmetric synthesis reactions. Unlike traditional open-chain tertiary phosphines that suffer from configurational instability at elevated temperatures, the cyclic structure provided by this invention ensures robust thermal stability and superior coordination ability. The strategic integration of a menthyl group not only serves as a chiral source to induce the formation of carbon, phosphorus, and axial chirality but also becomes an integral part of the final molecular architecture. This dual functionality eliminates the need for auxiliary removal steps, representing a significant paradigm shift in ligand design that promises to streamline manufacturing workflows for global supply chains seeking reliable high-purity pharmaceutical intermediate suppliers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of chiral tertiary phosphine compounds has been plagued by excessively tedious and lengthy synthetic routes that drive up costs and limit scalability. Conventional methods often rely on carbon skeleton chiral ligands such as BINAP or DIPAMP, which require no less than five to seven synthetic steps from simple raw materials to reach the final product. These multi-step processes frequently necessitate complex chemical resolution procedures to isolate the desired enantiomer, resulting in substantial material loss and increased waste generation. Furthermore, the chiral phosphine ligands themselves often account for a significant proportion of the total catalyst cost, sometimes exceeding the cost of the precious metals like palladium or rhodium they are paired with. The open-chain structures typical of these traditional compounds are prone to racemization under high-temperature conditions, which compromises the stereochemical integrity of the final active pharmaceutical ingredients. Additionally, the difficulty in obtaining compounds with chiral phosphorus atoms often leads to low stereoselectivity during formation, requiring further purification that further erodes profit margins and extends lead times for high-purity pharmaceutical intermediates.
The Novel Approach
The technology disclosed in patent CN110669073B offers a transformative solution by utilizing CDOP, an industrial flame retardant raw material, as the starting product, which drastically simplifies the supply chain and reduces raw material expenses. This innovative route involves introducing a menthyl group onto the phosphorus atom followed by an intramolecular cyclization reaction to generate a series of cyclic 9-menthyl-9,10-dihydrophosphaphenanthrene compounds with various substituents at the 10-position. By embedding the chiral menthyl group directly into the structure, the process avoids the extra step of removing chiral auxiliaries, which is a common requirement in other synthetic strategies. The resulting cyclic structure provides a rigid framework that stabilizes the configuration of the chiral phosphorus atom, allowing for relaxed reaction conditions during subsequent transformations without the risk of chirality loss. This approach not only enhances the asymmetric induction effect due to the bulky volume of the menthyl group but also enables the use of diastereomer mixtures as starting materials, thereby bypassing the need for tedious splitting processes entirely. 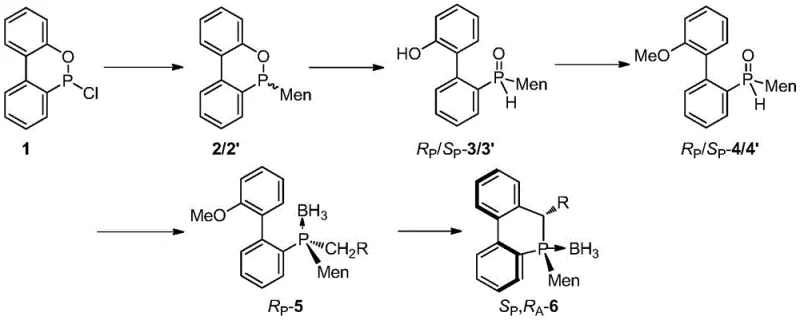
Mechanistic Insights into Menthyl-Induced Cyclization and Chirality
The core mechanistic advantage of this synthesis lies in the rational utilization of the menthyl group to induce the formation of multiple chiral centers, including carbon, phosphorus, and biphenyl axes, through a synergistic effect. The reaction sequence begins with the nucleophilic attack of menthyl magnesium halide or menthyl lithium on the CDOP starting material, creating a mixture of RP and SP configurations at the phosphorus center. Subsequent hydrolysis and methylation steps prepare the intermediate for a critical chlorination-Grignard reaction, where the introduction of various hydrocarbon groups sets the stage for the final ring closure. The intramolecular cyclization is achieved through the addition of butyllithium at low temperatures, which facilitates the formation of the cyclic phosphaphenanthrene backbone with high stereoselectivity. The presence of the menthyl group exerts a strong steric influence that directs the stereochemical outcome of the reaction, ensuring that the resulting product possesses the desired configuration without the need for external chiral catalysts during the synthesis itself. This internal chiral induction mechanism is highly efficient and minimizes the formation of unwanted byproducts, leading to cleaner reaction profiles and simplified downstream processing.
Furthermore, the cyclic nature of the final phosphaphenanthrene derivative plays a crucial role in maintaining the integrity of the chiral phosphorus atom during catalytic applications. In open-chain tertiary phosphines, the lone pair of electrons on the phosphorus atom is more exposed and susceptible to inversion, leading to racemization. However, the confinement imposed by the ring structure in these novel derivatives restricts this inversion, locking the phosphorus atom in a stable chiral configuration. This stability is further reinforced by the bulky menthyl substituent, which creates a protective asymmetric environment around the reactive center. Consequently, these compounds exhibit excellent coordination ability with transition metals, forming robust chiral catalysts that maintain high enantioselectivity over extended reaction periods. The ability to tolerate a wider range of reaction conditions without degradation makes these ligands particularly valuable for the commercial scale-up of complex pharmaceutical intermediates where process robustness is paramount. 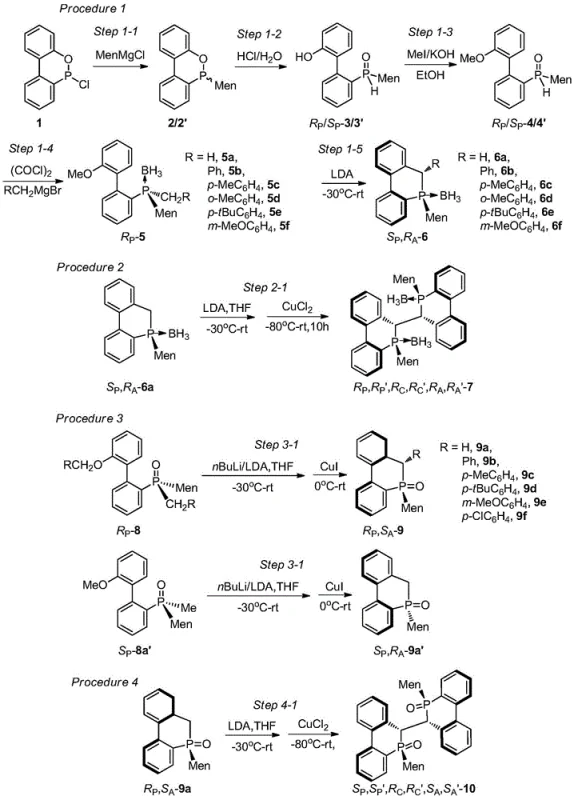
How to Synthesize 9-Menthyl-9,10-dihydrophosphaphenanthrene Efficiently
The synthesis of these high-value chiral ligands follows a well-defined protocol that begins with the preparation of the menthyl Grignard reagent and its reaction with CDOP in an ether solvent under heating conditions. The resulting mixture of isomers is then subjected to hydrolysis with hydrochloric acid to yield the hydroxy-biphenyl menthylphosphine oxide, which is subsequently methylated using iodomethane in an alkaline alcohol solution. The key step involves the conversion of the methoxy intermediate into the final cyclic structure via a chlorination-Grignard sequence followed by low-temperature lithiation and cyclization. For specific derivatives where R1 is hydrogen, the cyclization proceeds directly upon addition of butyllithium, whereas other substituents may require the presence of cuprous iodide to facilitate the coupling. The detailed standardized synthesis steps see the guide below, which outlines the precise stoichiometry, temperature controls, and workup procedures required to achieve high yields and diastereomeric ratios exceeding 99:1 in many cases.
- React CDOP with menthyl magnesium halide or menthyl lithium in ether solvent to form the initial phosphine oxide intermediate.
- Perform hydrolysis and methylation to prepare the methoxy-biphenyl menthylphosphine oxide precursor.
- Execute chlorination-Grignard reaction followed by intramolecular cyclization using butyllithium to finalize the cyclic phosphaphenanthrene structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this technology offers substantial cost savings and risk mitigation by fundamentally altering the raw material landscape for chiral ligand production. The use of CDOP, a commercially available industrial flame retardant raw material, replaces expensive and specialized chiral building blocks, leading to a significant reduction in the overall cost of goods sold for the final catalyst. By eliminating the need for chemical resolution steps and auxiliary removal, the process reduces the number of unit operations, which in turn lowers energy consumption, solvent usage, and labor costs associated with manufacturing. The enhanced stability of the cyclic structure means that the products have a longer shelf life and are less sensitive to storage conditions, reducing the risk of inventory write-offs due to degradation. This robustness also translates to more reliable delivery schedules, as the manufacturing process is less prone to batch failures caused by stereochemical instability. For supply chain heads, the ability to source high-purity pharmaceutical intermediates from a route that avoids precious metal-dependent resolution steps ensures greater supply continuity and reduces exposure to volatile metal markets.
- Cost Reduction in Manufacturing: The integration of the menthyl group as both a chiral source and a structural component eliminates the need for separate chiral auxiliary removal steps, which traditionally add significant cost and time to the synthesis of chiral phosphines. By utilizing CDOP, an industrial commodity chemical, as the starting material, the raw material costs are drastically lower compared to routes relying on specialized chiral pool chemicals. The streamlined process reduces the total number of synthetic steps, which minimizes solvent waste and energy usage, contributing to substantial cost savings in large-scale production. Furthermore, the high stereoselectivity achieved without resolution means that yield losses associated with discarding unwanted enantiomers are avoided, maximizing the efficiency of raw material utilization and driving down the cost per kilogram of the final active ligand.
- Enhanced Supply Chain Reliability: The thermal and configurational stability of the cyclic phosphaphenanthrene structure ensures that the product remains stable during storage and transportation, reducing the need for specialized cold chain logistics. The simplified synthesis route, which avoids complex resolution processes, is more robust and easier to scale, leading to more consistent batch-to-batch quality and reliable lead times for high-purity pharmaceutical intermediates. By reducing dependence on scarce or expensive chiral starting materials, the supply chain becomes more resilient to market fluctuations and raw material shortages. This reliability is critical for pharmaceutical manufacturers who require consistent quality and timely delivery to maintain their own production schedules and regulatory compliance.
- Scalability and Environmental Compliance: The process utilizes common reagents and solvents that are readily available at an industrial scale, facilitating easy technology transfer from laboratory to commercial production facilities. The reduction in synthetic steps and the avoidance of resolution processes significantly decrease the generation of chemical waste, aligning with green chemistry principles and reducing the environmental footprint of the manufacturing process. The high atom economy of the cyclization reaction ensures that most of the starting material ends up in the final product, minimizing waste disposal costs and regulatory burdens associated with hazardous waste management. This scalability and environmental compliance make the technology attractive for long-term partnerships focused on sustainable chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this novel phosphaphenanthrene synthesis technology. These answers are derived directly from the patent data and provide clarity on the operational advantages and chemical properties of the derivatives. Understanding these details is essential for R&D and procurement teams evaluating the feasibility of integrating these ligands into their existing catalytic processes.
Q: What is the primary advantage of using CDOP as a starting material?
A: CDOP is an industrial flame retardant raw material, which significantly lowers the raw material cost compared to specialized chiral building blocks used in conventional ligand synthesis.
Q: How does the menthyl group contribute to the stability of the product?
A: The chiral menthyl group stabilizes the configuration of the chiral phosphorus atom within the cyclic structure, preventing racemization during subsequent transformations and applications.
Q: Is chemical resolution required for this synthesis route?
A: No, the rational utilization of the menthyl group allows for the use of diastereomer mixtures as raw materials, avoiding the tedious and costly chemical resolution processes typical of other chiral phosphines.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phosphaphenanthrene Tertiary Phosphine Supplier
NINGBO INNO PHARMCHEM stands at the forefront of chemical innovation, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production for complex chiral ligands like those described in patent CN110669073B. Our technical team is equipped to adapt this advanced synthesis route to meet your specific purity requirements, ensuring stringent purity specifications are met through our rigorous QC labs and state-of-the-art analytical capabilities. We understand the critical nature of chiral catalysts in drug synthesis and are committed to providing a stable, high-quality supply of phosphaphenanthrene derivatives that can drive your asymmetric synthesis projects forward with confidence and efficiency.
We invite you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your project needs. By partnering with us, you gain access to a Customized Cost-Saving Analysis that demonstrates how switching to this novel ligand class can optimize your overall manufacturing budget. Let us help you overcome synthesis challenges and secure a competitive advantage in the global market with our reliable supply of advanced chiral intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
