Advanced Enzymatic Synthesis of High-Purity L-Heterocyclic Amino Acids for Commercial Scale-Up
Introduction to Breakthrough Biocatalytic Technology
The pharmaceutical industry is currently witnessing a transformative shift towards sustainable and highly selective manufacturing processes, particularly for complex chiral intermediates. Patent CN103276025A introduces a groundbreaking synthetic method for L-heterocyclic amino acids that leverages specific biocatalysts to overcome the longstanding limitations of traditional chemical synthesis. This technology utilizes a specialized phenylalanine dehydrogenase (amino acid sequence SEQ ID No.1) in conjunction with formate dehydrogenase and the NAD+ coenzyme system to drive the reductive amination of heterocyclic keto acids. By replacing harsh chemical reagents with biological precision, this method achieves raw material conversion rates exceeding 80% while maintaining exceptional stereochemical control. For R&D directors and procurement specialists, this represents a critical advancement in securing reliable supply chains for high-value active pharmaceutical ingredients (APIs) that require stringent purity profiles without the burden of heavy metal contamination.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral non-natural heterocyclic amino acids has relied heavily on chemical methodologies that present significant operational and economic drawbacks. Traditional approaches often involve asymmetric catalytic hydrogenation using expensive noble metals such as rhodium or ruthenium, which not only inflate raw material costs but also introduce the risk of toxic heavy metal residues in the final product. Furthermore, the presence of heterocyclic rings in the substrate can interfere with ligand binding, leading to reduced catalytic efficiency and inconsistent batch quality. Alternative strategies like chiral resolution of racemates are inherently inefficient, theoretically wasting 50% of the starting material, while asymmetric synthesis using chiral auxiliaries often requires long, multi-step routes with extensive solvent usage. These legacy methods struggle to meet the modern demands for green chemistry and cost-effective scalability, creating bottlenecks in the supply of critical pharmaceutical intermediates.
The Novel Approach
In stark contrast, the novel approach detailed in the patent employs a highly specific enzymatic cascade that operates under mild, aqueous conditions to deliver superior outcomes. By utilizing a tailored phenylalanine dehydrogenase, the process directly converts heterocyclic keto acids into the desired L-configured amino acids with high atom economy and minimal waste. The reaction system integrates a cofactor regeneration loop using formate dehydrogenase, which continuously recycles NADH from NAD+, thereby reducing the requirement for expensive coenzymes to catalytic levels. This biocatalytic route eliminates the need for organic solvents and precious metal catalysts, significantly simplifying the downstream purification process. The result is a streamlined manufacturing workflow that produces L-heterocyclic amino acids with optical purities often surpassing 99%, effectively removing the need for complex chiral separation steps and ensuring a cleaner, more sustainable production profile.
Mechanistic Insights into Enzymatic Reductive Amination
The core of this technological breakthrough lies in the precise mechanistic action of the phenylalanine dehydrogenase enzyme, which facilitates the stereoselective transfer of a hydride ion to the prochiral keto acid substrate. In the presence of ammonium ions and the reduced cofactor NADH, the enzyme stabilizes the transition state to ensure that hydride addition occurs exclusively from one face of the carbonyl group, locking in the L-configuration. Simultaneously, the coupled formate dehydrogenase oxidizes formate to carbon dioxide, regenerating NADH from the oxidized NAD+ byproduct, thus sustaining the catalytic cycle without the need for stoichiometric amounts of the costly cofactor. This dual-enzyme system operates optimally at a pH range of 8.2 to 8.5 and temperatures between 30°C and 40°C, conditions that are gentle enough to preserve the integrity of sensitive heterocyclic structures while driving the reaction to near-completion.
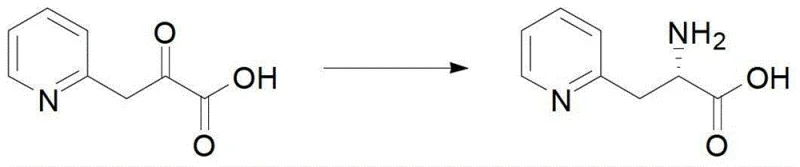
Impurity control is inherently managed through the high substrate specificity of the biocatalyst, which discriminates effectively against side reactions that typically plague chemical synthesis. Unlike chemical hydrogenation, which may lead to over-reduction of the heterocyclic ring or other functional groups, the enzyme targets only the keto-acid moiety, preserving the aromatic or heteroaromatic character of the molecule. The use of water as the primary solvent further mitigates the formation of organic byproducts and simplifies the removal of inorganic salts and residual enzymes through standard filtration and ion exchange techniques. This mechanistic elegance ensures that the final product stream is dominated by the desired L-isomer, with impurity profiles that are significantly cleaner than those obtained via traditional chemical routes, thereby reducing the burden on quality control laboratories and accelerating release times.
How to Synthesize L-Heterocyclic Amino Acid Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing these valuable intermediates, beginning with the preparation of the heterocyclic keto acid precursor followed by the key biotransformation step. The precursor can be synthesized via condensation of heterocyclic ketones with oxalic acid diesters or through Erlenmeyer-Plöchl type reactions, ensuring a steady supply of the substrate for the enzymatic step. Once the keto acid is prepared, it is dissolved in an aqueous buffer along with ammonium formate and the crude enzyme mixture containing both dehydrogenases. The reaction is monitored until the starting material is fully consumed, after which the product is isolated through acidification, filtration, and purification on a strong acidic cation exchange resin.
- Prepare the heterocyclic keto acid precursor by reacting heterocyclic ketones or alkyl compounds with oxalic acid diesters or acetic anhydride derivatives.
- Mix the keto acid with ammonium formate, crude enzyme mixture (containing phenylalanine dehydrogenase and formate dehydrogenase), and NAD+ coenzyme in an aqueous solution.
- Maintain the reaction at pH 8.2-8.5 and 30-40°C until conversion is complete, then purify the resulting L-amino acid via ion exchange resin.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this enzymatic technology offers profound strategic advantages that extend beyond simple technical metrics. The elimination of noble metal catalysts removes a major source of cost volatility and supply risk, as the price of metals like rhodium can fluctuate wildly based on geopolitical factors. Furthermore, the shift to an aqueous-based process drastically reduces the volume of hazardous organic solvents required, leading to substantial savings in solvent procurement, storage, and waste disposal costs. The simplified downstream processing, characterized by the absence of difficult chiral separations and heavy metal scavenging steps, shortens the overall production cycle time, allowing for faster turnaround on customer orders and improved inventory turnover rates.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the replacement of expensive stoichiometric chiral reagents and precious metal catalysts with recyclable biocatalysts. By operating in water and avoiding complex organic synthesis steps, the process significantly lowers the cost of goods sold (COGS) associated with solvent management and hazardous waste treatment. The high conversion efficiency means less raw material is wasted, and the high enantiomeric excess reduces the loss of product during purification, collectively contributing to a more lean and cost-effective manufacturing operation that enhances margin potential for high-volume API production.
- Enhanced Supply Chain Reliability: Reliability is bolstered by the robustness of the enzymatic system, which tolerates a wide range of heterocyclic substrates including pyridines, pyrazoles, and thiophenes, allowing for a flexible production platform. The use of crude enzyme mixtures simplifies the biocatalyst supply chain, as it removes the need for highly purified enzyme preparations that can be fragile and expensive to transport. Additionally, the mild reaction conditions reduce the risk of equipment corrosion and safety incidents associated with high-pressure hydrogenation or reactive chemical reagents, ensuring consistent uptime and uninterrupted supply continuity for critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: From a sustainability perspective, this technology aligns perfectly with modern environmental regulations and corporate ESG goals. The aqueous nature of the reaction minimizes VOC emissions, and the biodegradable nature of the enzyme waste stream simplifies effluent treatment compared to heavy metal-laden chemical waste. The process is inherently scalable, as demonstrated by the patent examples which range from gram to multi-gram scales without loss of efficiency, making it suitable for commercial scale-up of complex pharmaceutical intermediates. This environmental compliance reduces regulatory hurdles and facilitates smoother audits from global pharmaceutical partners who demand green manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enzymatic synthesis route. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, highlighting the practical benefits observed during development. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this technology into their existing manufacturing portfolios.
Q: Does this enzymatic process eliminate heavy metal residues?
A: Yes, unlike traditional noble metal catalytic hydrogenation, this biocatalytic method uses specific enzymes (SEQ ID No.1) and operates in aqueous media, ensuring the final L-heterocyclic amino acid is free from toxic heavy metal contaminants.
Q: What is the chiral purity achievable with this method?
A: The patent data demonstrates that using the specific phenylalanine dehydrogenase described results in chiral purities consistently exceeding 98%, with many examples reaching 99.5% or higher, eliminating the need for difficult chiral resolution steps.
Q: Is this process suitable for large-scale industrial production?
A: Absolutely. The process utilizes mild reaction conditions (30-40°C), water as a solvent, and robust crude enzyme mixtures, making it highly scalable, environmentally friendly, and cost-effective for commercial manufacturing compared to solvent-intensive chemical routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Heterocyclic Amino Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to meet the evolving needs of the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. By leveraging the enzymatic methodologies described in patents like CN103276025A, we can offer our clients a competitive edge through superior product quality and consistent supply reliability.
We invite you to engage with our technical procurement team to discuss how our capabilities can support your specific project requirements. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require specific COA data and route feasibility assessments for new targets, we are ready to provide the expertise you need. Partnering with us means gaining access to a dedicated resource focused on optimizing your API intermediate supply, reducing lead time for high-purity pharmaceutical intermediates, and driving innovation in your drug development pipeline.
