Advanced Enzymatic Synthesis of C3-Aromatic Pyrroloindole Alkaloids for Pharmaceutical Applications
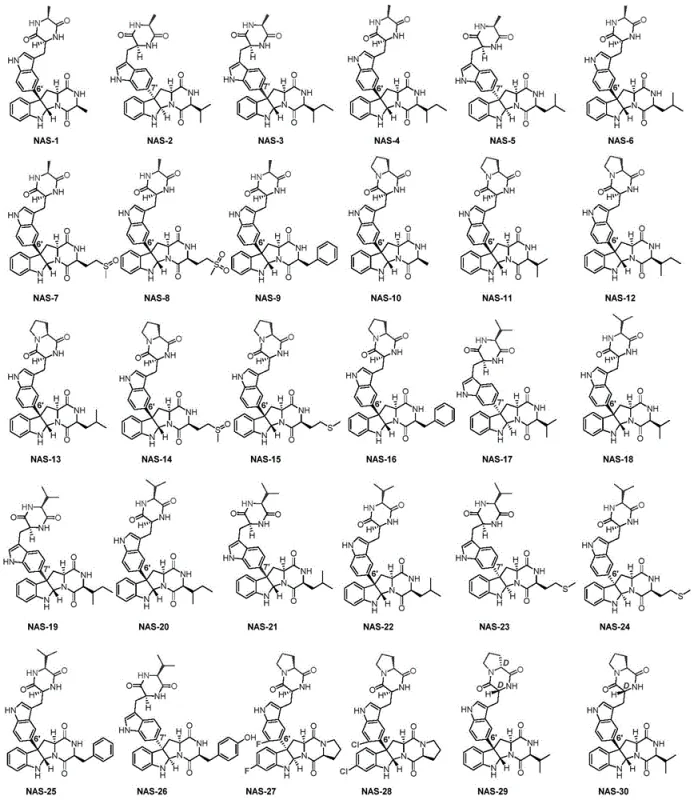
The pharmaceutical industry continuously seeks robust methodologies for constructing complex molecular scaffolds, particularly those featuring challenging stereochemical architectures. Patent CN109180691B introduces a groundbreaking biocatalytic approach for the synthesis of C3-aromatic pyrroloindole alkaloids, a class of natural products renowned for their potent physiological activities including antibacterial, antitumor, and neuroprotective properties. This technology leverages a sophisticated whole-cell catalytic system involving the nascB-P450 enzyme and a spinach-derived electron transport system to achieve high-efficiency dimerization of tryptophan-containing cyclic dipeptides. Unlike traditional chemical routes that struggle with the steric hindrance of C3 quaternary carbons, this enzymatic pathway delivers exceptional regio- and stereoselectivity, generating a diverse library of compounds designated as NAS-1 through NAS-30. For R&D teams focused on neurodegenerative disease targets, access to such high-purity intermediates is critical for accelerating lead optimization campaigns.
The structural complexity of pyrroloindole alkaloids presents a formidable barrier to conventional organic synthesis, primarily due to the presence of an ortho-positioned C3 quaternary carbon and a C2 tertiary carbon chiral center. Traditional chemical strategies often rely on intricate multi-step sequences employing expensive chiral catalysts or auxiliaries to induce stereospecificity, yet these methods frequently suffer from moderate enantiomeric excess (e.e. values around 90%) and low overall yields. Furthermore, the reliance on heavy metal catalysts necessitates rigorous downstream purification to meet stringent pharmaceutical impurity specifications, significantly driving up manufacturing costs and extending lead times. In contrast, the novel approach detailed in the patent utilizes a biosynthetic strategy that mimics nature's efficiency, bypassing the need for harsh reagents and complex protecting group manipulations. By employing a genetically engineered Escherichia coli strain expressing the nascB-P450 gene, the process achieves direct oxidative coupling of cyclic dipeptide substrates, resulting in products with superior optical purity and simplified post-treatment workflows.
Mechanistic Insights into nascB-P450 Catalyzed Oxidative Dimerization
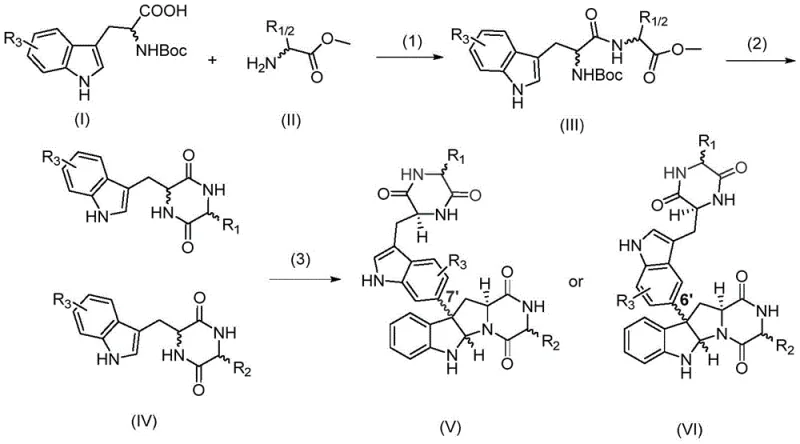
The core of this technological advancement lies in the precise mechanistic action of the nascB-P450 cytochrome P450 monooxygenase, which functions in concert with a ferredoxin (Fd) and ferredoxin reductase (FdR) electron transport chain derived from spinach. This enzymatic machinery facilitates the activation of molecular oxygen to generate highly reactive iron-oxo species capable of abstracting hydrogen atoms from the unactivated C-H bonds of the indole ring. The subsequent radical recombination occurs with exquisite control over the regioselectivity, favoring the formation of C3-C3', C3-C6', or C3-N linkages depending on the specific substrate geometry and enzyme active site constraints. This level of control is virtually impossible to replicate with small-molecule catalysts, which often produce mixtures of regioisomers requiring difficult chromatographic separation. The integration of the spinach-derived electron transfer system ensures a continuous supply of reducing equivalents (NADPH regeneration), sustaining the catalytic cycle efficiently within the whole-cell environment.
Furthermore, the impurity profile generated by this biocatalytic route is significantly cleaner compared to chemical alternatives. Because the enzyme exhibits high substrate specificity, side reactions such as over-oxidation or non-selective polymerization are minimized. The use of a whole-cell system also provides a protective intracellular environment that stabilizes the reactive intermediates, preventing degradation pathways common in organic solvents. For quality control laboratories, this translates to simpler analytical validation and higher confidence in batch-to-batch consistency. The ability to tune the regioselectivity by simply varying the amino acid component of the cyclic dipeptide precursor allows for the rapid generation of structural analogs, providing medicinal chemists with a powerful tool for exploring structure-activity relationships (SAR) without the burden of developing new synthetic routes for each derivative.
How to Synthesize C3-Aromatic Pyrroloindole Alkaloids Efficiently
The implementation of this synthesis protocol involves a streamlined sequence beginning with the preparation of cyclic dipeptide substrates followed by the biotransformation step. The substrate synthesis utilizes standard peptide coupling chemistry to link N-Boc-tryptophan analogs with various amino acid methyl esters, followed by deprotection and cyclization to form the diketopiperazine core. Once the substrate is prepared, the biocatalytic phase employs the engineered E. coli GB05dir-T7 strain harboring the necessary plasmids for enzyme expression and electron transport. The reaction proceeds in an aqueous buffer system under mild temperatures, eliminating the safety hazards associated with high-pressure or high-temperature chemical reactors. Detailed standardized synthesis steps for replicating this high-efficiency pathway are provided in the guide below.
- Synthesize tryptophan-containing cyclic dipeptide substrates (IV) via condensation of N-Boc-tryptophan analogs and amino acid methyl esters, followed by deprotection and cyclization.
- Construct the biocatalytic system by co-transforming nascB-P450 gene and spinach-derived electron transport system (Fd/FdR) plasmids into modified E. coli GB05dir-T7.
- Perform whole-cell catalysis by incubating the engineered bacteria with the cyclic dipeptide substrate in M9 medium at 18°C for 24 hours, followed by extraction and HPLC purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the shift from chemical synthesis to this enzymatic platform offers substantial strategic benefits regarding cost stability and operational resilience. The elimination of precious metal catalysts and chiral ligands removes a significant variable cost component that is often subject to volatile market pricing and geopolitical supply risks. Additionally, the aqueous nature of the biocatalytic process drastically reduces the consumption of hazardous organic solvents, leading to lower waste disposal costs and simplified environmental compliance reporting. This green chemistry alignment not only enhances the sustainability profile of the supply chain but also mitigates regulatory risks associated with solvent residues in final drug products.
- Cost Reduction in Manufacturing: The enzymatic route fundamentally alters the cost structure by replacing expensive stoichiometric chiral reagents with catalytic amounts of biocatalysts that can be produced via fermentation. This transition eliminates the need for costly chiral resolution steps, which typically result in a maximum theoretical yield of 50% for racemic mixtures. By achieving high stereoselectivity directly, the process maximizes atom economy and reduces the raw material input required per kilogram of active pharmaceutical ingredient (API) intermediate. Furthermore, the simplified downstream processing, which avoids complex metal scavenging operations, contributes to significant operational expenditure savings.
- Enhanced Supply Chain Reliability: Reliance on fermentation-based production enhances supply security by decoupling manufacturing from the supply chains of rare earth metals or specialized chemical reagents. The engineered bacterial strains can be stored long-term and revived rapidly, ensuring business continuity even during disruptions in chemical feedstock availability. The scalability of fermentation technology is well-established in the industry, allowing for seamless capacity expansion from pilot scale to multi-ton commercial production without the need for extensive re-engineering of the process infrastructure. This flexibility ensures that procurement managers can secure reliable long-term contracts with consistent quality specifications.
- Scalability and Environmental Compliance: The process operates under mild physiological conditions (near neutral pH and moderate temperatures), which reduces energy consumption for heating and cooling compared to traditional high-energy chemical syntheses. The use of water as the primary reaction medium minimizes the generation of volatile organic compound (VOC) emissions, aligning with increasingly strict global environmental regulations. This eco-friendly profile facilitates easier permitting for new manufacturing facilities and reduces the liability associated with hazardous waste management. The robustness of the whole-cell system also allows for high substrate loading, improving the volumetric productivity of the bioreactors and further driving down the unit cost of production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biocatalytic technology for producing neuroprotective intermediates. These insights are derived directly from the experimental data and process descriptions found in the patent literature, ensuring accuracy for technical decision-makers. Understanding these details is essential for evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: What is the primary advantage of using nascB-P450 for synthesizing pyrroloindole alkaloids compared to chemical methods?
A: The nascB-P450 enzymatic method offers superior regio- and stereoselectivity, specifically enabling the construction of difficult C3 quaternary carbon centers with high optical purity without the need for complex chiral metal catalysts or extensive purification steps required in traditional organic synthesis.
Q: Can this enzymatic process be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the patent describes a whole-cell catalytic system using modified E. coli, which is inherently scalable through fermentation technology. The process avoids expensive transition metals and operates under mild aqueous conditions, facilitating large-scale manufacturing compliance with environmental standards.
Q: What specific biological activity do the synthesized NAS-1 to NAS-30 compounds exhibit?
A: The synthesized C3-aromatic pyrroloindole alkaloids demonstrate significant in vitro anti-nerve damage activity, particularly showing protective effects against glutamate-induced PC-12 cell apoptosis, indicating potential utility in treating neurodegenerative conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable C3-Aromatic Pyrroloindole Alkaloids Supplier
As the demand for complex natural product analogs grows in the neuropharmacology sector, having a manufacturing partner with deep expertise in biocatalysis is essential. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from laboratory discovery to market supply. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including advanced chiral HPLC analysis to guarantee the optical integrity of every batch. We understand the critical nature of supply continuity for clinical trials and commercial launches, and our robust quality management systems are designed to meet the exacting standards of global regulatory agencies.
We invite you to discuss how this innovative enzymatic technology can optimize your development pipeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Our experts are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how we can support your goal of delivering high-quality neuroprotective therapies to patients efficiently and sustainably.
