Advanced Biocatalytic Production of High-Purity Moxifloxacin Side Chain Intermediates
The pharmaceutical industry is constantly seeking more efficient and sustainable pathways for the production of critical antibiotic intermediates, and the technology disclosed in patent CN106399418B represents a significant leap forward in this domain. This patent details a novel biocatalytic method for preparing the moxifloxacin side chain, specifically the (S,S)-2,8-diazabicyclo[4.3.0]nonane derivatives, which are essential building blocks for third-generation fluoroquinolone antibiotics. Unlike traditional chemical synthesis routes that often rely on harsh conditions and stoichiometric chiral reagents, this invention leverages the specificity of omega-transaminases to achieve exceptional stereocontrol. By utilizing engineered enzymes derived from sources such as Arthrobacter or Vibrio fluvialis, the process converts readily available ketone precursors into the desired chiral amines with unprecedented efficiency. This shift from chemocatalysis to biocatalysis not only addresses the growing demand for green chemistry but also provides a robust solution for manufacturing high-purity pharmaceutical intermediates at a commercial scale. The implications for supply chain stability and cost reduction are profound, as the method simplifies the synthetic route while drastically improving overall yield and optical purity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the moxifloxacin side chain has been plagued by inefficiencies inherent in classical chemical methodologies, particularly the resolution method which has been the dominant industrial approach. As illustrated in the conventional pathway below, the process typically begins with pyridine dicarboxylate and involves high-pressure hydrogenation followed by a resolution step using agents like tartaric acid. 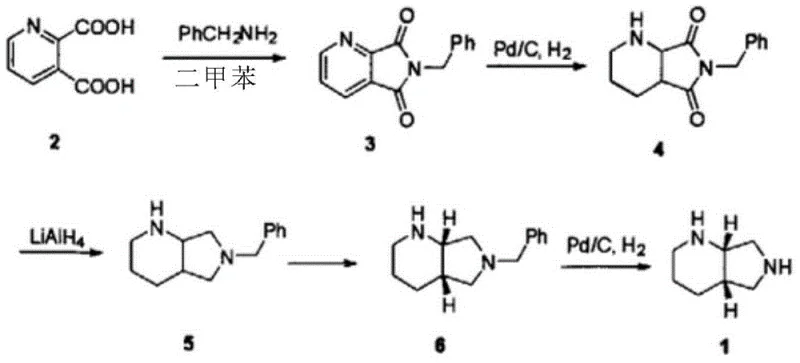
The fundamental flaw in this traditional resolution strategy is the theoretical yield ceiling of 50%, as the unwanted enantiomer is typically discarded or requires complex recycling processes that add significant cost and waste. Furthermore, these chemical routes often necessitate the use of hazardous reagents such as lithium aluminum hydride for reduction steps, posing serious safety risks and environmental burdens during large-scale production. The multi-step nature of these legacy processes, often exceeding nine distinct synthetic operations, compounds the loss of material at each stage, resulting in overall yields that can drop as low as 27%. Additionally, the reliance on column chromatography for purification in some asymmetric synthesis variants further hinders scalability, making it difficult to meet the rigorous demands of global API supply chains without incurring prohibitive manufacturing expenses.
The Novel Approach
In stark contrast to these cumbersome chemical protocols, the biocatalytic approach introduced in the patent offers a streamlined and highly efficient alternative that fundamentally reimagines the construction of the bicyclic core. This new methodology utilizes a specific transaminase to catalyze the asymmetric amination of a ketone substrate, followed by a spontaneous intramolecular cyclization to form the target diazabicyclo structure. The elegance of this route lies in its ability to bypass the need for external chiral auxiliaries or expensive metal catalysts, relying instead on the innate chirality of the enzyme active site to dictate stereochemistry. By operating under mild aqueous conditions, the process eliminates the safety hazards associated with high-pressure hydrogenation and reactive hydride reagents. Moreover, the integration of a dynamic kinetic resolution mechanism allows for the recycling of the unwanted isomer back into the reaction cycle, theoretically breaking the 50% yield barrier and enabling near-quantitative conversion of starting materials into the valuable chiral intermediate. This represents a paradigm shift towards more sustainable and economically viable manufacturing practices for complex pharmaceutical intermediates.
Mechanistic Insights into Transaminase-Catalyzed Cyclization
The core of this technological breakthrough resides in the precise enzymatic transformation of the linear ketone precursor into the cyclic amine product through a coupled transamination and cyclization sequence. As depicted in the reaction scheme, the omega-transaminase facilitates the transfer of an amino group from a donor, such as isopropylamine, to the carbonyl carbon of compound 4, generating the chiral amine intermediate 3. 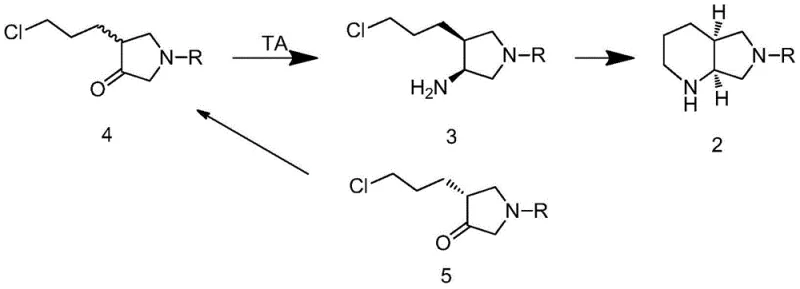
What makes this mechanism particularly powerful for industrial application is the subsequent spontaneous ring closure that occurs under the reaction conditions, converting the unstable intermediate 3 directly into the stable bicyclic product 2 without the need for additional reagents or isolation steps. Crucially, the system is designed to handle the stereochemical byproduct; the unreacted R-isomer of the ketone substrate (compound 5) undergoes spontaneous racemization in the alkaline reaction environment, reverting back to the racemic substrate 4 which can then re-enter the enzymatic cycle. This dynamic kinetic resolution ensures that essentially all starting material is funneled towards the desired S,S-configured product, maximizing atom economy. The enzyme variants specified, such as those corresponding to SEQ ID NO: 18-20, exhibit high tolerance to organic co-solvents like DMSO, which is essential for solubilizing the hydrophobic substrate while maintaining enzymatic activity. This delicate balance of chemical spontaneity and biological specificity allows for the production of the moxifloxacin side chain with greater than 99% enantiomeric excess and diastereomeric excess, meeting the stringent purity requirements of modern regulatory standards.
Controlling impurities in such a complex bicyclic system is critical, and the enzymatic route offers superior selectivity compared to chemical catalysts which might promote side reactions like over-reduction or non-specific alkylation. The high fidelity of the transaminase ensures that the chiral centers are established with precision, minimizing the formation of diastereomers that are notoriously difficult to separate by crystallization or chromatography. By maintaining the reaction pH between 9.0 and 10.0 using triethanolamine buffers, the process optimizes the equilibrium of the transamination reaction while facilitating the necessary base-catalyzed racemization of the unwanted ketone isomer. This dual function of the reaction medium simplifies the process control parameters, reducing the need for complex feedback loops or intermediate adjustments. Furthermore, the use of isopropylamine as the amino donor generates acetone as the only byproduct, which is easily removed due to its volatility, thereby driving the reaction equilibrium forward and preventing product inhibition. This clean reaction profile significantly reduces the burden on downstream purification units, resulting in a final product with a much cleaner impurity profile compared to chemically synthesized equivalents.
How to Synthesize Moxifloxacin Side Chain Efficiently
Implementing this biocatalytic route requires careful attention to the reaction parameters to ensure optimal enzyme performance and substrate conversion. The process begins with the preparation of a reaction mixture containing the ketone substrate, an organic co-solvent to enhance solubility, and a buffered aqueous phase to maintain the required alkalinity. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction system with substrate compound 4, organic solvent like DMSO, amino donor such as isopropylamine, and buffer solution maintained at pH 9.0-10.0.
- Introduce the specific omega-transaminase enzyme (e.g., SEQ ID NO: 18-20) and cofactor PLP, maintaining the temperature at 35°C for 8 to 24 hours.
- Allow the intermediate amine compound 3 to undergo spontaneous ring closure to form the final bicyclic product 2, followed by extraction and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this biocatalytic technology translates into tangible strategic advantages that go beyond mere technical specifications. The elimination of precious metal catalysts and stoichiometric chiral resolving agents removes significant cost drivers from the bill of materials, leading to substantial cost savings in pharmaceutical intermediates manufacturing. By simplifying the synthetic sequence from over nine steps down to a concise enzymatic cascade, the process reduces the consumption of raw materials, solvents, and energy, thereby lowering the overall cost of goods sold. The high yield and selectivity minimize waste generation, which not only aligns with corporate sustainability goals but also reduces the costs associated with waste disposal and environmental compliance. This efficiency gain allows for more competitive pricing structures without compromising on margin, providing a buffer against raw material price volatility in the global chemical market.
- Cost Reduction in Manufacturing: The transition to an enzymatic process eliminates the need for expensive heavy metal catalysts and complex chiral separation technologies, which are traditionally major cost centers in fine chemical production. By achieving near-quantitative yields through dynamic kinetic resolution, the process maximizes the utility of every kilogram of starting material, effectively halving the raw material cost per unit of product compared to resolution-based methods. The mild reaction conditions also reduce energy consumption associated with heating, cooling, and high-pressure equipment, contributing to a leaner operational expenditure model. Furthermore, the simplified workup procedure reduces the demand for chromatographic resins and extensive solvent exchanges, streamlining the production timeline and reducing labor costs.
- Enhanced Supply Chain Reliability: Relying on biocatalysis diversifies the supply risk by reducing dependence on scarce or geopolitically sensitive chemical reagents such as specialized chiral ligands or noble metals. The enzymes used in this process can be produced via fermentation using renewable feedstocks, ensuring a stable and scalable supply of the biocatalyst itself. The robustness of the process under mild conditions means that production is less susceptible to disruptions caused by utility failures or equipment maintenance issues common in high-pressure chemical plants. This reliability ensures consistent lead times for high-purity pharmaceutical intermediates, allowing downstream API manufacturers to plan their production schedules with greater confidence and reduce safety stock levels.
- Scalability and Environmental Compliance: The aqueous nature of the biocatalytic reaction significantly reduces the volume of organic solvents required, simplifying solvent recovery and reducing the facility's volatile organic compound (VOC) emissions. This aligns perfectly with increasingly stringent environmental regulations, minimizing the risk of compliance-related shutdowns or fines. The process is inherently safer, removing hazards associated with pyrophoric reagents and high-pressure hydrogen, which facilitates easier permitting and expansion of production capacity. The ability to scale from laboratory to commercial tonnage without significant re-engineering of the process parameters ensures that supply can grow in lockstep with market demand for moxifloxacin and related antibiotics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biocatalytic platform for antibiotic intermediate production. These answers are derived directly from the experimental data and claims within the patent documentation to ensure accuracy and relevance for technical decision-makers.
Q: How does the biocatalytic route improve yield compared to traditional resolution methods?
A: Traditional resolution methods are theoretically limited to a maximum 50% yield due to the discarding of the unwanted enantiomer. The patented biocatalytic process utilizes a dynamic kinetic resolution mechanism where the unwanted R-isomer spontaneously racemizes back to the substrate, allowing for theoretical yields approaching 100% and demonstrated yields greater than 95%.
Q: What are the specific reaction conditions required for this transaminase process?
A: The process operates under mild conditions, specifically at a temperature of 35°C and a pH range of 9.0 to 10.0 using triethanolamine buffer. It requires an amino donor like isopropylamine and a cofactor such as pyridoxal phosphate (PLP), eliminating the need for high-pressure hydrogenation or cryogenic temperatures.
Q: Is this process suitable for large-scale commercial manufacturing of API intermediates?
A: Yes, the process is highly scalable. It eliminates the need for expensive chiral reagents and heavy metal catalysts, simplifying downstream processing. The high conversion rate (>99%) and excellent stereoselectivity (>99% ee/de) reduce waste and purification steps, making it economically viable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Moxifloxacin Side Chain Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced biocatalytic manufacturing requires a partner with deep technical expertise and proven industrial capability. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent literature can be reliably translated into bulk supply. We maintain stringent purity specifications and operate rigorous QC labs equipped to verify the stereochemical integrity of every batch, guaranteeing that the moxifloxacin side chain intermediates we deliver meet the exacting standards required for GMP API synthesis. Our commitment to quality assurance means that every shipment is accompanied by comprehensive analytical data, providing full transparency and traceability for your regulatory filings.
We invite you to collaborate with us to optimize your supply chain for next-generation antibiotics. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our biocatalytic capabilities can enhance your production efficiency and reduce overall project costs. Let us help you secure a sustainable and competitive source for your critical pharmaceutical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
