Scalable Synthesis of Functionalized Chiral Monomers for Advanced Covalent Organic Frameworks
Scalable Synthesis of Functionalized Chiral Monomers for Advanced Covalent Organic Frameworks
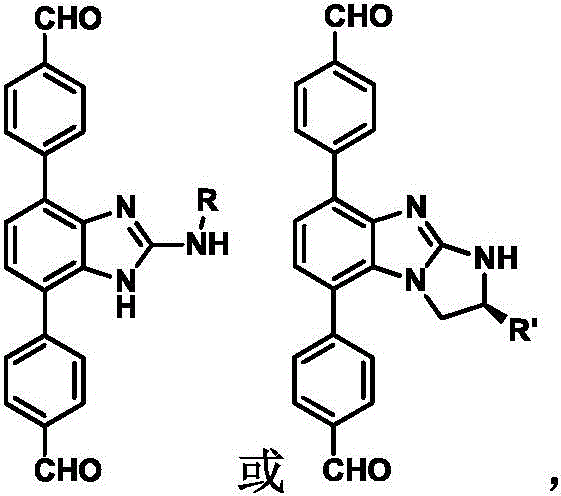
The development of chiral porous materials has long been a bottleneck in the advancement of enantioselective technologies, particularly in the pharmaceutical and fine chemical sectors. Patent CN109293575B introduces a groundbreaking methodology for the synthesis of novel chiral monomers that serve as the fundamental building blocks for Chiral Covalent Organic Frameworks (CCOFs). Unlike traditional approaches that often struggle with structural irregularity or poor stability, this invention leverages a robust Suzuki-Miyaura coupling strategy to attach formyl-functionalized phenyl groups to a chiral benzimidazole core. This technical breakthrough addresses the critical scarcity of diverse, high-purity chiral precursors required to construct stable and highly ordered porous networks. By establishing a reliable pathway to these complex intermediates, the technology opens new avenues for creating materials with tunable pore sizes and specific chiral environments, essential for next-generation separation and catalysis processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral porous materials has been plagued by significant technical hurdles that limit their industrial viability. Conventional methods often rely on the direct polymerization of ill-defined chiral polymers or the modification of existing achiral frameworks, which frequently results in materials with inconsistent pore distributions and low surface areas. Furthermore, many existing synthetic routes for chiral monomers require harsh reaction conditions, such as extreme temperatures or the use of unstable intermediates that degrade rapidly, leading to poor yields and difficult purification processes. The lack of modular synthetic strategies means that altering the chiral environment often requires redesigning the entire synthetic pathway from scratch, making the exploration of structure-activity relationships prohibitively expensive and time-consuming for research and development teams. Additionally, the difficulty in removing residual metal catalysts or organic impurities from these complex matrices often compromises the performance of the final material in sensitive applications like drug purification.
The Novel Approach
The methodology disclosed in the patent represents a paradigm shift by decoupling the construction of the chiral core from the installation of the framework-forming functional groups. By utilizing a dibrominated benzimidazole scaffold as a versatile platform, the process allows for the late-stage introduction of aldehyde moieties via palladium-catalyzed cross-coupling. This approach ensures that the delicate chiral centers, introduced in the earlier nucleophilic substitution step, are not exposed to the potentially racemizing conditions of the final coupling reaction. The use of p-formylphenylboronic acid as a coupling partner is particularly strategic, as it provides the necessary reactive handles for subsequent condensation reactions to form the COF network without requiring additional protection-deprotection steps. This streamlined workflow not only enhances the overall yield but also significantly simplifies the downstream processing, making the production of these high-value intermediates far more accessible for commercial scale-up.
Mechanistic Insights into Suzuki-Miyaura Cross-Coupling for Monomer Functionalization
The core of this synthetic innovation lies in the efficient execution of the Suzuki-Miyaura coupling reaction under optimized conditions that preserve stereochemical integrity. The mechanism involves the oxidative addition of the palladium(0) catalyst to the carbon-bromine bond of the chiral benzimidazole precursor, forming an organopalladium intermediate. This step is critical and is facilitated by the electron-deficient nature of the benzimidazole ring, which activates the halide for insertion. Subsequently, the transmetallation with the p-formylphenylboronic acid, activated by the base (typically potassium carbonate), transfers the aryl group to the palladium center. The final reductive elimination releases the desired chiral monomer and regenerates the active palladium catalyst. Crucially, the reaction conditions—specifically the use of a 1,4-dioxane and water mixture at controlled temperatures around 100°C—are mild enough to prevent the epimerization of the chiral alpha-carbon adjacent to the nitrogen atom, ensuring high enantiomeric excess in the final product.
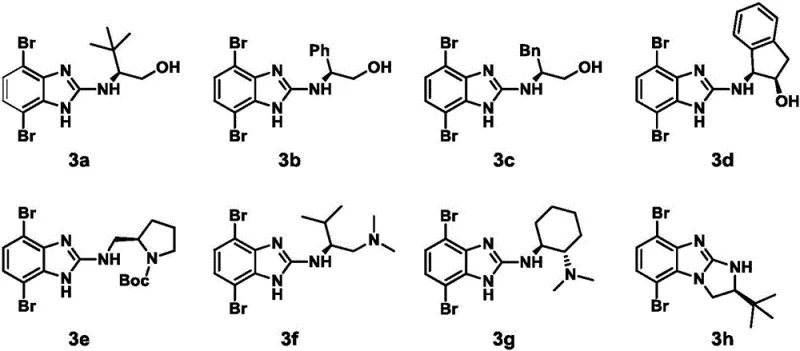
Impurity control is another vital aspect of this mechanistic design, particularly concerning the removal of palladium residues which can be detrimental to downstream applications. The choice of ligands and the specific solvent system plays a dual role in driving the reaction to completion while keeping metal species soluble for easier extraction. The patent data indicates that the reaction tolerates a wide range of steric and electronic environments on the chiral amine component, as evidenced by the successful synthesis of various derivatives ranging from simple alkyl chains to bulky cyclic structures. This tolerance suggests that the catalytic cycle is robust against steric hindrance, likely due to the small bite angle of the triphenylphosphine ligands used in the tetrakis(triphenylphosphine)palladium catalyst. Furthermore, the preservation of the aldehyde functionality during the coupling is paramount, as these groups are the reactive sites for the eventual formation of the imine linkages in the COF; the mild basicity of potassium carbonate ensures that aldol condensation side reactions are minimized during the monomer synthesis phase.
How to Synthesize Chiral Benzimidazole Monomers Efficiently
The synthesis of these high-value chiral monomers follows a logical two-step sequence that balances reactivity with selectivity. First, the chiral amine is coupled to the dibromobenzimidazole core under basic conditions to establish the stereocenter. Following purification, this intermediate undergoes the key Suzuki coupling to install the framework-building aldehyde groups. This modular approach allows manufacturers to stockpile the common brominated intermediate and diversify the product line by simply changing the chiral amine input. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below.
- Prepare the chiral precursor by reacting 2-chloro-4,7-dibromobenzimidazole with a chiral amine under basic conditions at elevated temperatures.
- Purify the intermediate brominated benzimidazole derivative using column chromatography to ensure high optical purity.
- Execute the Suzuki coupling reaction between the purified precursor and p-formylphenylboronic acid using a palladium catalyst in a dioxane-water solvent system.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain and procurement perspective, the adoption of this synthetic route offers substantial strategic benefits over legacy methods for producing chiral building blocks. The reliance on commodity chemicals such as p-formylphenylboronic acid and standard palladium catalysts reduces dependency on exotic or single-source reagents, thereby mitigating supply risk. Moreover, the robustness of the Suzuki coupling reaction translates to higher process reliability, reducing the frequency of batch failures and the associated costs of reprocessing. The ability to produce a library of structurally diverse monomers from a common intermediate allows for economies of scale in the production of the precursor, driving down the unit cost for specialized derivatives. This flexibility is crucial for meeting the dynamic demands of the pharmaceutical and agrochemical industries, where rapid iteration of material properties is often required.
- Cost Reduction in Manufacturing: The elimination of complex protection-deprotection sequences significantly lowers the consumption of auxiliary reagents and solvents. By avoiding harsh conditions that require specialized pressure-rated equipment, the process can be executed in standard glass-lined or stainless steel reactors, reducing capital expenditure requirements. The high yields reported for the coupling step minimize raw material waste, directly improving the cost-of-goods-sold (COGS) profile for these advanced intermediates.
- Enhanced Supply Chain Reliability: The use of widely available starting materials ensures a stable supply base, insulating production schedules from the volatility often seen with niche chiral reagents. The simplified purification protocols, primarily involving standard extraction and chromatography techniques, reduce the lead time for releasing batches for quality control. This efficiency enables a more responsive supply chain capable of adapting quickly to fluctuations in demand for chiral separation media.
- Scalability and Environmental Compliance: The reaction solvent system, consisting of dioxane and water, is amenable to recycling and recovery, aligning with modern green chemistry principles and reducing waste disposal costs. The absence of toxic heavy metals other than the catalytic amount of palladium, which can be scavenged, simplifies environmental compliance and wastewater treatment. The thermal stability of the intermediates allows for safe handling and storage, facilitating logistics and distribution to global customers without stringent cold-chain requirements.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral monomer technology. These insights are derived directly from the experimental data and structural analysis provided in the patent documentation, offering clarity on performance and applicability.
Q: What is the primary advantage of this chiral monomer synthesis route?
A: The primary advantage is the modularity of the Suzuki coupling reaction, which allows for the precise introduction of aldehyde functional groups necessary for COF formation while preserving the stereochemical integrity of the chiral center.
Q: Can these monomers be scaled for industrial production?
A: Yes, the reaction conditions utilize standard reagents like potassium carbonate and tetrakis(triphenylphosphine)palladium, which are compatible with large-scale batch reactors and established purification workflows.
Q: What applications do these chiral covalent organic frameworks support?
A: These materials are specifically designed for chiral separation, enantioselective catalysis, and chiral sensing applications due to their regular pore structures and inherent chirality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Monomer Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route described in CN109293575B for the production of high-performance chiral materials. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to industrial application is seamless. Our facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch of chiral monomer delivered. We understand that the success of your final Covalent Organic Framework depends entirely on the quality of the monomeric building blocks, and we are committed to providing materials that meet the highest standards of enantiomeric purity and chemical consistency.
We invite you to collaborate with our technical procurement team to explore how this technology can optimize your material development pipeline. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of how our manufacturing efficiencies can translate into tangible value for your organization. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your unique project requirements, ensuring a partnership built on transparency and technical excellence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
