Advanced Manufacturing of 5-(2-Amino-1-hydroxyethyl)quinolin-2(1H)-one Derivatives for Global Pharma Supply Chains
Advanced Manufacturing of 5-(2-Amino-1-hydroxyethyl)quinolin-2(1H)-one Derivatives for Global Pharma Supply Chains
The pharmaceutical industry constantly seeks robust, scalable, and high-purity synthetic routes for complex active pharmaceutical ingredients (APIs) and their intermediates. A significant breakthrough in this domain is detailed in patent CN102348691A, which outlines an improved process for manufacturing 5-(2-{[6-(2,2-difluoro-2-phenylethoxy)hexyl]amino}-1-hydroxyethyl)-8-hydroxyquinolin-2(1H)-one and its pharmaceutically acceptable salts. This compound represents a critical structural motif often found in potent beta-adrenergic agonists used for respiratory conditions. The innovation lies not merely in the creation of the molecule but in the strategic re-engineering of the solvent system and purification protocols. By shifting from polar aprotic solvents like DMSO to aromatic hydrocarbons such as xylene, the inventors have unlocked a pathway that dramatically enhances both chemical purity and overall process efficiency. For R&D directors and procurement specialists alike, this patent offers a compelling case study in how subtle changes in reaction conditions can translate into massive commercial advantages, ensuring a reliable supply of high-quality pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, specifically those disclosed in WO 2006/122788 A1 and WO 2008/095720 A1, relied heavily on dimethyl sulfoxide (DMSO) as the primary solvent for the key coupling reaction between the quinolinone precursor and the fluoro-amine side chain. While chemically feasible, these conventional routes suffered from severe drawbacks when scaled to industrial levels. The use of DMSO often led to the formation of difficult-to-remove impurities, necessitating rigorous and costly purification steps such as column chromatography after every single reaction stage. Consequently, the overall yield for the final napadisilate salt was abysmally low, calculated at merely 9.5%. Furthermore, the HPLC analysis of the final product revealed impurity levels ranging between 5% and 6%, which is unacceptable for modern regulatory standards requiring high-purity API intermediates. The reliance on chromatographic purification not only inflated manufacturing costs but also created significant bottlenecks in production throughput, making the supply chain fragile and prone to delays.
The Novel Approach
The novel approach described in CN102348691A fundamentally disrupts this inefficient paradigm by introducing xylene as the solvent for the initial nucleophilic substitution step. This seemingly simple solvent swap has profound implications for the reaction profile. The unexpected discovery was that conducting the reaction between compound (V) and compound (IV) in refluxing xylene significantly suppresses the formation of by-products, resulting in a much cleaner crude intermediate (III). This improvement in crude quality allows manufacturers to bypass intermediate chromatographic purification entirely. Instead, the process utilizes crystallization techniques, specifically using naphthalene-1,5-disulfonic acid, to purify the intermediates. The result is a staggering increase in overall yield to approximately 60.7% for the final salt, with HPLC impurities reduced to just 1.5%. This shift from a linear, purification-heavy process to a streamlined, crystallization-based workflow exemplifies how process chemistry optimization can drive substantial cost reduction in pharmaceutical intermediate manufacturing.
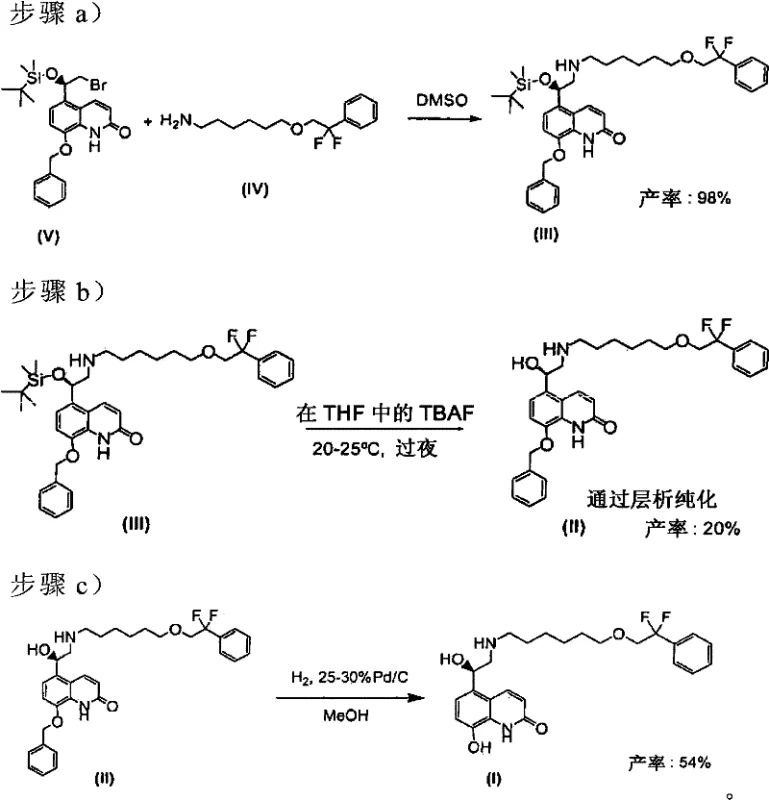
The visual representation of the synthesis highlights the critical transition points where the new methodology diverges from the old. In the initial step (Step a), the reaction of the bromo-quinolinone derivative with the amino-ether side chain is shown proceeding in high yield (98%) under the new conditions. Subsequent deprotection steps (Steps b and c) are optimized to maintain this purity, avoiding the degradation often seen in harsher solvent systems. The final conversion to the napadisilate salt (Step d) is depicted as a high-yielding precipitation, underscoring the efficiency of the salt formation strategy which locks in the purity achieved in the earlier stages.
Mechanistic Insights into Solvent Effects and Deprotection Strategies
The mechanistic superiority of the xylene-based system can be attributed to the differential solubility of reactants and by-products, as well as the thermal properties of the solvent. Xylene, having a higher boiling point than many ethers but lower polarity than DMSO, facilitates the nucleophilic attack of the amine on the alkyl bromide while keeping inorganic salts and polar impurities insoluble, allowing them to be filtered off hot. This "hot filtration" capability is a game-changer for scale-up, as it removes the need for aqueous workups that often lead to emulsion formation and product loss. Furthermore, the deprotection strategy employs orthogonal protecting groups (P1 as benzyl and P2 as tert-butyldimethylsilyl) which allows for selective removal. The P2 deprotection using TBAF in THF is carefully controlled at 40-50°C to prevent racemization, ensuring the chiral integrity of the benzylic alcohol center is maintained throughout the synthesis.
Impurity control is further enhanced during the P1 deprotection step, which involves catalytic hydrogenation. The patent specifies the use of palladium on carbon (Pd/C) with careful control of catalyst loading (less than 10 wt%). This specific condition is crucial for minimizing the formation of defluorinated impurities, such as 5-(2-{[6-(2-phenoxy)hexyl]amino}...), which can arise from over-reduction. By optimizing the hydrogenation pressure (1-2 crust) and time, the process ensures that only the benzyl ether is cleaved while the sensitive difluoro-ethyl moiety remains intact. The final salt formation with naphthalene-1,5-disulfonic acid acts as a final "chemical filter," co-crystallizing the desired enantiomer while leaving residual organic impurities in the mother liquor. This multi-layered approach to impurity management ensures that the final product consistently meets the stringent e.e. > 98% specification required for clinical applications.
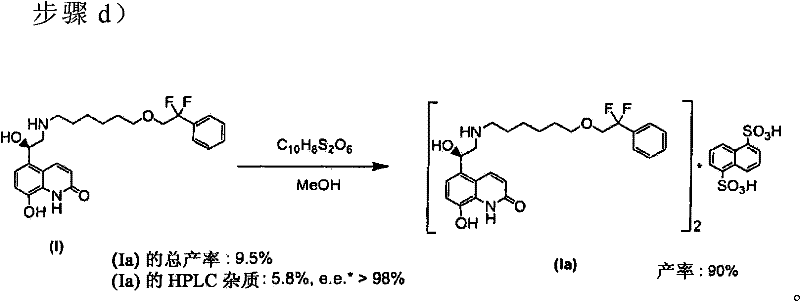
How to Synthesize 5-(2-Amino-1-hydroxyethyl)quinolin-2(1H)-one Derivatives Efficiently
The synthesis of this complex quinolinone derivative requires precise adherence to the optimized conditions outlined in the patent to replicate the high yields and purity profiles. The process begins with the preparation of the protected intermediate in xylene, followed by a sequential deprotection sequence that leverages crystallization rather than chromatography. This approach is specifically designed for pilot and commercial scale operations where throughput and solvent recovery are paramount. Operators must pay close attention to the temperature controls during the TBAF deprotection and the hydrogenation steps to avoid side reactions. The following guide summarizes the critical operational parameters derived from the experimental examples, providing a roadmap for technical teams looking to implement this superior manufacturing route.
- React compound (V) with amine (IV) in xylene solvent at reflux to form intermediate (III), replacing traditional DMSO conditions to minimize impurities.
- Perform P2 deprotection using TBAF in THF at 40-50°C, followed by crystallization with naphthalene-1,5-disulfonic acid to purify intermediate (II).
- Execute P1 deprotection via hydrogenation with Pd/C in acetate/alcohol mixture, followed by direct salt formation to yield the final napadisilate product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the process described in CN102348691A offers transformative benefits that go beyond simple chemistry. The most immediate impact is the drastic simplification of the manufacturing workflow. By eliminating multiple chromatographic purification steps, the process reduces the consumption of expensive silica gel and large volumes of elution solvents. This directly translates to significant cost reduction in API intermediate manufacturing, as the operational expenditure associated with waste disposal and solvent recovery is markedly lowered. Furthermore, the reduction in unit operations shortens the overall cycle time, allowing facilities to produce more batches per year with the same equipment footprint. This increased asset utilization is a key driver for improving the return on investment for production lines dedicated to respiratory therapeutics.
- Cost Reduction in Manufacturing: The shift from a 9.5% overall yield to over 60% represents a six-fold increase in material efficiency. This means that for every kilogram of starting material purchased, the output of saleable product is exponentially higher. Additionally, the replacement of DMSO with xylene allows for easier solvent recycling, as xylene can be distilled and reused more efficiently than high-boiling polar solvents. The elimination of column chromatography also removes a major cost center, as it requires specialized equipment, skilled labor, and generates substantial solid waste. These factors combine to create a leaner, more cost-effective production model that enhances margin potential for the final drug product.
- Enhanced Supply Chain Reliability: Supply chain resilience is often compromised by complex processes with low yields and high failure rates. The robust nature of the xylene-based synthesis, with its built-in purification via crystallization, significantly reduces the risk of batch failures. Crystallization is a well-understood, scalable unit operation that is less prone to variability than chromatography. This reliability ensures consistent delivery schedules to downstream API manufacturers. Moreover, the reagents used, such as xylene, TBAF, and Pd/C, are commodity chemicals with stable global supply chains, reducing the risk of raw material shortages that could halt production. This stability is crucial for maintaining the continuity of supply for life-saving medications.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, the new process is far superior. DMSO is known to penetrate skin rapidly and carry dissolved contaminants with it, posing occupational health risks. Xylene, while requiring standard handling precautions, is a more manageable solvent in large-scale reactors. The reduction in solvent usage and the elimination of silica waste align with green chemistry principles, helping companies meet increasingly strict environmental regulations. The ability to scale this process from 100 kgs to 100 MT annual commercial production without fundamental changes to the chemistry makes it an ideal candidate for long-term commercial supply agreements, ensuring that capacity can be ramped up seamlessly as market demand grows.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this patented process is essential for stakeholders evaluating its implementation. The following questions address common inquiries regarding the scalability, purity, and regulatory implications of this synthetic route. These answers are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Whether you are a quality assurance manager concerned about impurity profiles or a process engineer looking at reactor compatibility, these insights clarify the operational realities of adopting this advanced manufacturing technology.
Q: How does the xylene solvent system improve yield compared to conventional DMSO methods?
A: The use of xylene instead of DMSO in the initial coupling step significantly reduces the formation of side products, allowing the overall yield of the napadisilate salt to increase from approximately 9.5% in prior art to over 60% in this optimized process.
Q: What purification techniques are eliminated in this new manufacturing route?
A: This process eliminates the need for column chromatography for intermediate purification. Instead, it utilizes crystallization techniques using naphthalene-1,5-disulfonic acid, which is more scalable and cost-effective for industrial production.
Q: What is the enantiomeric excess (e.e.) achieved with this synthetic route?
A: The optimized process maintains high stereochemical integrity, achieving an enantiomeric excess (e.e.) of greater than 98%, which is critical for the biological activity of the final pharmaceutical ingredient.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-(2-Amino-1-hydroxyethyl)quinolin-2(1H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust infrastructure. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields demonstrated in patent CN102348691A can be realized on an industrial scale. Our state-of-the-art facilities are equipped to handle the specific solvent systems and reaction conditions required for this synthesis, including high-pressure hydrogenation and precise temperature-controlled crystallizations. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of intermediate meets the highest international standards, providing our partners with the confidence they need to advance their drug development pipelines.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this optimized synthetic route. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this process can improve your bottom line. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples for your evaluation. Let us help you secure a sustainable, high-quality supply of this critical pharmaceutical intermediate, driving your projects forward with speed and efficiency.
