Advanced Industrial Synthesis of Methylcobalamin: Eliminating Toxic Reagents for Safer Production
The pharmaceutical and nutraceutical industries are constantly seeking robust, scalable, and environmentally compliant pathways for producing high-value active ingredients. Patent CN1409723A introduces a transformative approach to the production of methylcobalamin, a critical coenzyme form of Vitamin B12 known for its superior neurotrophic properties compared to other cobalamin analogs. This patent addresses long-standing industrial bottlenecks associated with traditional methylation techniques, specifically targeting the hazards of volatile alkylating agents and the inefficiencies of chromatographic purification. By leveraging water-soluble sulfonium derivatives in conjunction with mild reducing agents, this technology enables the direct conversion of cyanocobalamin or hydroxocobalamin into high-purity methylcobalamin. For global procurement leaders and R&D directors, this represents a significant opportunity to optimize supply chains for neurological therapeutics and vitamin supplements, ensuring consistent quality while adhering to stricter environmental, health, and safety (EHS) standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of methylcobalamin has been plagued by significant operational and safety challenges that hinder efficient commercial scale-up of complex pharmaceutical intermediates. Traditional routes often relied on the use of methyl iodide, a highly volatile liquid with a boiling point of merely 41-43°C, which presents severe handling difficulties and toxicity risks, including potential carcinogenicity. Furthermore, alternative methods utilizing metal powders such as zinc introduced heavy metal contamination concerns, necessitating rigorous and costly removal steps to meet pharmacopeial standards. Other approaches involving monomethyl dicarboxylates or mercury-based reagents were deemed unsuitable for large-scale manufacturing due to the instability of reagents or the generation of hazardous pollutants. Perhaps most critically, achieving the requisite purity levels typically demanded multiple rounds of column chromatography, a process that is not only labor-intensive and time-consuming but also generates substantial volumes of organic solvent waste, driving up both production costs and environmental liabilities.
The Novel Approach
The methodology disclosed in CN1409723A fundamentally reengineers the methylation step by substituting hazardous reagents with stable, water-soluble trimethylsulfonium derivatives. This innovative strategy allows the reaction to proceed in aqueous or aqueous-organic media under mild thermal conditions, typically between 15-50°C, thereby eliminating the need for specialized pressure equipment or extreme temperature controls. A distinct advantage of this chemistry is the ability to isolate the final product through simple crystallization or precipitation techniques rather than relying on chromatographic separation. This shift from complex purification to straightforward physical isolation drastically simplifies the downstream processing workflow. By avoiding the use of methyl iodide and heavy metal powders, the process inherently reduces the risk of toxic residue in the final API, aligning perfectly with the increasing demand for cleaner, greener synthesis routes in the fine chemical sector.
Mechanistic Insights into Reductive Methylation of Cobalamins
The core chemical transformation involves the nucleophilic substitution at the cobalt center of the corrin ring, where the axial ligand is exchanged to form the methyl-cobalt bond. In this specific protocol, the starting material, either cyanocobalamin or hydroxocobalamin, undergoes reduction in the presence of a reducing agent such as sodium borohydride. This reduction step is crucial for activating the cobalt center, lowering its oxidation state to facilitate the subsequent attack by the methylating species. The use of water-soluble sulfonium salts, such as trimethylsulfonium iodide or trimethylsulfoxonium bromide, serves as an effective methyl donor that is compatible with aqueous environments. Unlike methyl iodide, which requires anhydrous conditions and generates iodide byproducts that can complicate purification, these sulfonium salts decompose into benign byproducts that do not interfere with the crystal lattice formation of the target molecule.
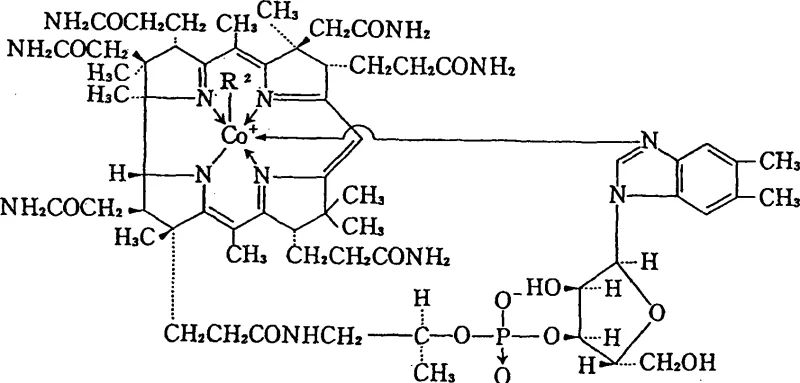
Impurity control is intrinsically built into this mechanism through the solubility characteristics of the reaction components. The patent highlights that the methylated product exhibits distinct solubility behavior in water-miscible organic solvents like acetone or 2-butanone compared to the starting materials and side products. By carefully adjusting parameters such as pH, temperature, and solvent composition, the methylcobalamin can be induced to precipitate selectively. This selective crystallization acts as a powerful purification step, effectively excluding unreacted precursors and inorganic salts without the need for silica gel or resin columns. Furthermore, the optional inclusion of trace amounts of cyanide-trapping agents like ferrous sulfate or cobalt chloride enhances the reaction selectivity, suppressing the formation of unwanted byproducts and ensuring that the final crystalline solid meets stringent purity specifications required for parenteral or oral administration.
How to Synthesize Methylcobalamin Efficiently
Implementing this synthesis route requires precise control over reaction stoichiometry and environmental conditions to maximize yield and crystal quality. The process begins with the dissolution of the cobalamin precursor in ion-exchanged water, followed by the addition of the sulfonium methylating agent and optional trapping agents. The reaction is conducted under an inert nitrogen atmosphere and protected from light to prevent photodegradation of the sensitive corrin ring. Following the addition of the reducing agent, the mixture is maintained at a controlled temperature to allow complete conversion. The workup involves cooling the reaction mass and adding water-miscible organic solvents to induce precipitation. The resulting crude solid is then subjected to a recrystallization cycle involving pH adjustment and solvent washing to achieve the final high-purity specification.
- Dissolve cyanocobalamin or hydroxocobalamin in an aqueous or aqueous-organic solvent system under inert atmosphere.
- Add a water-soluble methylating agent such as trimethylsulfonium iodide and a reducing agent like sodium borohydride at controlled temperatures between 15-50°C.
- Precipitate the high-purity product directly via crystallization or solvent adjustment, bypassing traditional column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process offers tangible strategic benefits that extend beyond mere technical feasibility. The elimination of column chromatography represents a major operational leap, as it removes a significant bottleneck in production throughput. Chromatography is notoriously difficult to scale linearly; by replacing it with crystallization, manufacturers can utilize standard reactor vessels and filtration equipment, significantly enhancing the commercial scale-up of complex vitamin intermediates. This transition leads to substantial cost savings by reducing the consumption of expensive chromatography media and the vast quantities of organic solvents previously required for elution and regeneration. Additionally, the avoidance of methyl iodide mitigates regulatory compliance costs associated with handling hazardous air pollutants and toxic substances, streamlining the permitting process for new manufacturing facilities.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the simplification of downstream processing. By removing the dependency on column chromatography, producers eliminate the recurring costs of stationary phase materials and the energy-intensive processes of solvent recovery associated with large-volume eluents. The use of commercially available, stable sulfonium salts further stabilizes raw material costs, as these reagents do not require the specialized storage or handling infrastructure needed for volatile alkyl halides. Consequently, the overall cost of goods sold (COGS) for high-purity methylcobalamin is significantly optimized, allowing for more competitive pricing in the global vitamin market.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the use of robust, non-volatile reagents that are less susceptible to shipping restrictions and logistical delays compared to controlled substances like methyl iodide. The process operates effectively in aqueous systems, reducing the fire hazard classification of the manufacturing site and minimizing the risk of production stoppages due to safety incidents. Furthermore, the high yield and selectivity of the reaction ensure consistent batch-to-batch output, reducing the variability that often plagues biological extraction or complex semi-synthetic routes. This reliability is critical for maintaining steady inventory levels for downstream formulators of neurological drugs and dietary supplements.
- Scalability and Environmental Compliance: From an EHS perspective, this methodology aligns with modern green chemistry principles by minimizing waste generation and avoiding heavy metal contaminants. The absence of zinc powder and mercury compounds simplifies wastewater treatment protocols, lowering the environmental footprint of the production facility. The ability to run the reaction in water or water-alcohol mixtures reduces the reliance on chlorinated or aromatic solvents, facilitating easier solvent recycling and disposal. This environmental compatibility not only future-proofs the manufacturing asset against tightening regulations but also enhances the brand reputation of the supplier as a responsible partner in the pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear understanding of the process capabilities. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into existing production lines or for sourcing teams negotiating supply agreements.
Q: Why is the new sulfonium salt method superior to traditional methyl iodide processes?
A: Traditional methods rely on methyl iodide, which is volatile, toxic, and carcinogenic, requiring complex handling and extensive column chromatography. The new method uses stable, water-soluble sulfonium salts that allow for direct crystallization, significantly improving operator safety and reducing solvent waste.
Q: Does this process require heavy metal catalysts like zinc powder?
A: No. Unlike older methods that utilized zinc powder which poses heavy metal contamination risks, this process operates efficiently with minimal or no metal ion trapping agents, simplifying downstream purification and ensuring higher product purity.
Q: What represents the key cost saving in this manufacturing route?
A: The primary cost reduction comes from eliminating column chromatography. By achieving high purity through direct precipitation and crystallization, manufacturers save substantially on chromatography media, organic solvent consumption, and waste disposal costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methylcobalamin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the global pharmaceutical market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering high-purity methylcobalamin that adheres to stringent purity specifications, utilizing rigorous QC labs to verify every batch against international pharmacopeial standards. Our infrastructure is designed to support the complex requirements of vitamin B12 derivative synthesis, guaranteeing supply continuity for our partners.
We invite forward-thinking organizations to collaborate with us to leverage these technological advancements for their product portfolios. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
