Advanced Manufacturing of 6'-Aminoluteolin Derivatives: A Breakthrough in Anti-Inflammatory Drug Intermediate Synthesis
Advanced Manufacturing of 6'-Aminoluteolin Derivatives: A Breakthrough in Anti-Inflammatory Drug Intermediate Synthesis
The pharmaceutical industry is constantly seeking novel scaffolds to overcome drug resistance and improve therapeutic indices, particularly in the realm of non-steroidal anti-inflammatory drugs (NSAIDs). Patent CN106674179B introduces a transformative approach to functionalizing the flavonoid backbone, specifically targeting the previously underutilized 6' position of the luteolin structure. This innovation moves beyond traditional modification sites, offering a pathway to synthesize stable aminoluteolin o-quinone derivatives with enhanced pharmacological profiles. By leveraging a direct oxidative strategy followed by Michael addition, this technology enables the efficient production of high-value intermediates that possess superior bioavailability and reduced toxicity compared to their parent compounds. For R&D teams and procurement strategists, understanding this methodology is crucial for developing the next generation of anti-inflammatory therapeutics.
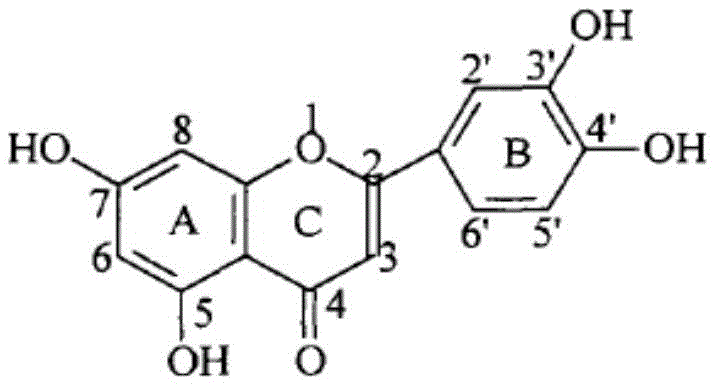
The core of this technology lies in the precise manipulation of the luteolin molecule, a naturally occurring flavonoid known for its antioxidant and antitumor properties. As illustrated in the structure above, luteolin possesses multiple hydroxyl groups that typically dictate its reactivity. However, the patented process ingeniously exploits the electron-rich nature of the B-ring to facilitate selective oxidation. This results in the formation of a reactive luteolin o-quinone intermediate in situ, which then serves as an electrophilic trap for various nucleophilic amines. This strategic shift from passive extraction to active, targeted chemical modification represents a significant leap forward in fine chemical synthesis, providing a robust platform for creating diverse molecular libraries aimed at optimizing the lipid/water distribution coefficients of lead compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the structural modification of flavonoids like luteolin has been predominantly focused on the A and C rings, specifically targeting positions such as C2, C3, C6, C8, C3', and C4'. While these traditional pathways have yielded numerous derivatives, they often suffer from significant drawbacks including harsh reaction conditions, the necessity for protecting group strategies, and limited regioselectivity. Conventional methods frequently rely on strong acids or bases and high temperatures, which can lead to the degradation of the sensitive flavonoid skeleton and the formation of complex impurity profiles that are difficult to separate. Furthermore, the introduction of amino groups at the 6' position was virtually unreported prior to this invention, leaving a gap in the chemical space available for drug discovery. The reliance on multi-step synthesis with low overall yields in older methodologies creates substantial bottlenecks for commercial scale-up, driving up costs and extending lead times for pharmaceutical developers seeking novel API intermediates.
The Novel Approach
In stark contrast to these legacy techniques, the method disclosed in CN106674179B offers a streamlined, one-pot strategy that directly accesses the 6'-amino substituted derivatives. By utilizing a mild ethanol-water mixed solvent system and controlling the pH within a specific alkaline range, the process promotes the spontaneous air oxidation of luteolin to its o-quinone form without the need for external oxidizing agents. This reactive intermediate immediately undergoes a Michael addition with the added amine source, effectively installing the desired functionality in a single operational sequence. The versatility of this approach is demonstrated by its compatibility with a wide array of amines, ranging from simple aliphatic chains to complex aromatic and heterocyclic structures. This not only simplifies the synthetic route but also drastically reduces the environmental burden by eliminating toxic reagents and minimizing waste generation, aligning perfectly with modern green chemistry mandates.
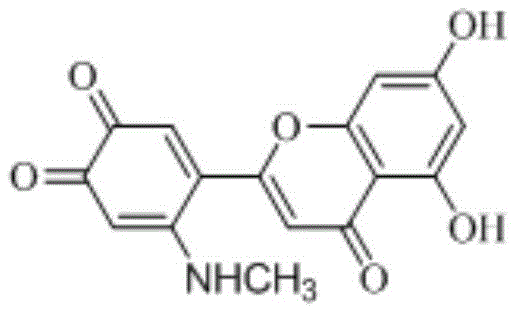
Mechanistic Insights into Oxidative Michael Addition
The mechanistic elegance of this synthesis relies on the delicate balance between oxidation kinetics and nucleophilic attack. Under the optimized alkaline conditions (pH 9.2-9.3), the phenolic hydroxyl groups on the B-ring of luteolin are deprotonated, increasing their susceptibility to single-electron transfer with dissolved oxygen. This generates the luteolin o-quinone, a highly electrophilic species characterized by an α,β-unsaturated carbonyl system. The presence of the amine nucleophile in the reaction mixture ensures that as soon as the quinone forms, it is trapped via a conjugate addition at the 6' position. This tandem oxidation-addition sequence prevents the polymerization or further degradation of the quinone, which is a common side reaction in uncontrolled oxidations. The result is a clean conversion to the 6'-amino derivative, preserving the integrity of the rest of the flavonoid scaffold while introducing the pharmacologically active amino moiety.
Impurity control is inherently built into this mechanism through the strict regulation of reaction parameters. The use of a buffer system maintains the pH within a narrow window, preventing the over-oxidation of the product or the hydrolysis of the newly formed C-N bond. Additionally, the choice of ethanol as the primary solvent ensures that both the polar luteolin starting material and the less polar amine reagents remain in solution, facilitating homogeneous reaction kinetics. Monitoring via Thin Layer Chromatography (TLC) allows for real-time tracking of the disappearance of the starting material and the emergence of the product, ensuring that the reaction is quenched at the optimal point to maximize yield and purity. This level of process control is essential for meeting the stringent quality standards required for pharmaceutical intermediates, where even trace impurities can impact downstream biological testing.
How to Synthesize 6'-Aminoluteolin Derivatives Efficiently
Implementing this synthesis on a laboratory or pilot scale requires adherence to specific procedural guidelines to ensure reproducibility and safety. The process begins with the preparation of a homogeneous solution of luteolin in ethanol, often assisted by ultrasonic dissolution to break up aggregates. Simultaneously, the chosen amine reagent is dissolved in ethanol and added to the luteolin solution. The critical step involves the slow addition of a buffer solution to adjust the pH to the optimal range of 8.4 to 10, with a preference for 9.2 to 9.3, which triggers the color change indicative of quinone formation. The mixture is then heated to a moderate temperature between 30°C and 65°C under reflux conditions. Detailed standard operating procedures for stoichiometry, mixing rates, and workup protocols are essential for consistent results.
- Dissolve luteolin and the chosen amino-active group substance in an ethanol-water mixed solvent system, ensuring complete solubilization often aided by ultrasonic treatment.
- Adjust the pH of the reaction mixture to a precise range of 8.4-10 (optimally 9.2-9.3) using a buffer solution to facilitate the air oxidation of luteolin to luteolin o-quinone.
- Maintain the reaction temperature between 30-65°C under reflux, monitoring progress via TLC, followed by cooling and product isolation through extraction or filtration.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented technology offers compelling advantages that directly address the pain points of procurement managers and supply chain directors. The elimination of expensive transition metal catalysts and hazardous oxidizing agents translates into a significant reduction in raw material costs and waste disposal fees. The use of ethanol and water as solvents not only lowers the cost of goods sold (COGS) but also simplifies the regulatory compliance landscape, as these solvents are generally recognized as safe and easy to recover. Furthermore, the mild reaction temperatures (30-65°C) reduce energy consumption compared to high-temperature processes, contributing to a lower carbon footprint and reduced utility costs for manufacturing facilities. These factors combine to create a highly cost-effective manufacturing route that enhances the overall margin potential for the final API.
- Cost Reduction in Manufacturing: The process achieves cost efficiency by removing the need for precious metal catalysts and complex purification steps associated with metal removal. The simplicity of the workup, which often involves basic extraction or filtration, minimizes labor and equipment time. By avoiding the use of protected intermediates and multi-step sequences, the overall material throughput is improved, leading to substantial savings in precursor costs. The high atom economy of the Michael addition further ensures that the majority of the starting materials end up in the final product, reducing waste and maximizing resource utilization.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as luteolin, ethanol, and common amines (methylamine, ethylamine, benzylamine) ensures a stable and resilient supply chain. These raw materials are widely available from multiple global suppliers, mitigating the risk of shortages or price volatility associated with specialty reagents. The robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, ensuring consistent production output. This reliability allows for better inventory planning and reduces the need for excessive safety stock, optimizing working capital for the organization.
- Scalability and Environmental Compliance: The aqueous-ethanolic solvent system is inherently scalable, posing fewer safety risks than processes using volatile or toxic organic solvents. The absence of heavy metals simplifies the environmental permitting process and reduces the burden on wastewater treatment facilities. The mild thermal profile of the reaction allows for the use of standard glass-lined or stainless steel reactors without the need for specialized high-pressure or cryogenic equipment. This ease of scale-up facilitates a smoother transition from kilogram-scale development to multi-ton commercial production, ensuring that supply can meet market demand without significant capital investment in new infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these novel luteolin derivatives. The answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on the feasibility and benefits of adopting this technology. Understanding these details is vital for stakeholders evaluating the potential integration of 6'-aminoluteolin derivatives into their drug discovery pipelines or manufacturing portfolios.
Q: What is the primary advantage of modifying luteolin at the 6' position?
A: Modifying the 6' position allows for the creation of novel non-steroidal anti-inflammatory structures with adjusted lipophilicity and improved pharmacokinetic parameters, addressing the limitations of traditional substitution patterns at C2, C3, or C7.
Q: Does this synthesis require expensive transition metal catalysts?
A: No, the process described in patent CN106674179B utilizes air oxidation in an alkaline ethanol-water medium, completely eliminating the need for costly transition metal catalysts and the subsequent complex removal steps.
Q: What types of amines can be introduced using this method?
A: The method is highly versatile, accommodating aliphatic amines (methylamine, ethylamine), alcohol amines (ethanolamine), aromatic amines (benzylamine), and heterocyclic amines (furfurylamine), allowing for diverse library generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6'-Aminoluteolin Derivative Supplier
The development of 6'-aminoluteolin derivatives represents a significant opportunity for innovation in the NSAID sector, and NINGBO INNO PHARMCHEM is uniquely positioned to support your journey from bench to bedside. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can grow seamlessly alongside your clinical needs. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of intermediate delivered meets the highest international standards. We understand the critical nature of timeline and quality in pharmaceutical development, and our dedicated technical team is committed to providing the reliability and expertise you require.
We invite you to collaborate with us to explore the full potential of this patented synthesis route. Our experts can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this green chemistry approach can optimize your budget. We encourage you to contact our technical procurement team today to request specific COA data for our reference standards and to discuss route feasibility assessments for your target molecules. Let us be your partner in transforming this promising chemical technology into a commercial reality.
