Advanced Synthesis of Site-Specific Deuterated 1,2,3-Triazoles for Drug Discovery
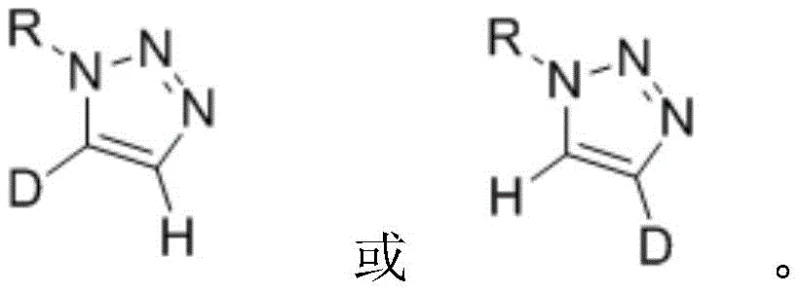
The pharmaceutical industry is increasingly recognizing the strategic value of deuterated compounds in optimizing drug metabolic profiles and extending half-lives through the kinetic isotope effect. A pivotal advancement in this domain is disclosed in patent CN113461624A, which details a highly efficient preparation method for selective mono-deuterated markers of mono-1-substituted 1,2,3-triazoles. This technology addresses a critical gap in synthetic chemistry by enabling the precise installation of a single deuterium atom at either the C4 or C5 position of the triazole ring, a feat that was previously challenging with conventional cycloaddition strategies. The ability to generate these specific isotopologues is essential for rigorous reaction kinetics studies, detailed NMR structural elucidation, and the development of next-generation deuterated drug entities where metabolic stability at specific sites is paramount.
For R&D directors and process chemists, the significance of this patent lies in its versatility and operational simplicity. Unlike earlier methods that often yielded complex mixtures of di-deuterated products or required harsh conditions, this approach utilizes a straightforward base-catalyzed hydrogen-deuterium exchange mechanism. By leveraging common organic bases and controlling solvent systems, manufacturers can now access high-purity deuterated triazoles that serve as critical building blocks for bioactive molecules such as tazobactam and rufinamide analogs. This level of control over isotopic positioning transforms the triazole scaffold from a static structural element into a tunable component for advanced medicinal chemistry programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of deuterated 1,2,3-triazoles has relied heavily on copper-catalyzed azide-alkyne cycloaddition reactions using deuterated acetylene sources. While effective for generating the triazole core, these traditional pathways frequently suffer from a lack of regioselectivity regarding the isotopic label. For instance, reacting deuterated acetylene with organic azides typically results in the formation of 4,5-dideutero-1,2,3-triazoles, where both carbon atoms on the ring carry a deuterium atom. Separating these di-deuterated species from mono-deuterated impurities is notoriously difficult and often requires extensive chromatographic purification, leading to significant material loss and increased production costs. Furthermore, the generation of deuterated acetylene often involves the use of calcium carbide and heavy water, which introduces safety hazards and handling complexities associated with gas evolution and moisture sensitivity.
Another significant drawback of conventional click chemistry approaches is the reliance on transition metal catalysts, specifically copper. Residual copper in the final product is a major concern for pharmaceutical applications due to strict regulatory limits on heavy metal impurities. Removing trace copper to parts-per-million levels adds additional processing steps, such as scavenging treatments or specialized filtration, which complicates the manufacturing workflow. Additionally, the harsh conditions sometimes required to drive these cycloadditions can be incompatible with sensitive functional groups present on complex drug intermediates. Consequently, there has been a persistent demand in the fine chemical sector for a milder, more selective, and metal-free alternative that can deliver site-specific deuteration without compromising yield or purity.
The Novel Approach
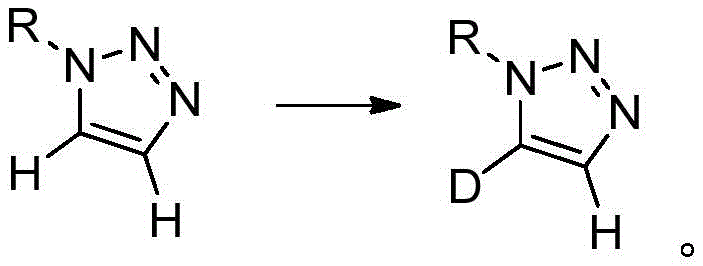
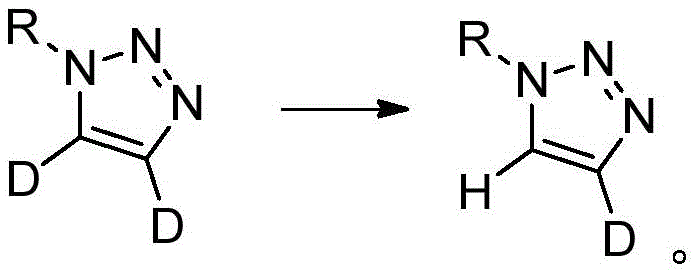
The methodology outlined in CN113461624A represents a paradigm shift by decoupling the formation of the triazole ring from the deuteration event. Instead of building the ring with deuterium already in place, this innovative strategy starts with a pre-formed mono-1-substituted 1,2,3-triazole and performs a selective post-synthetic modification. By employing strong organic bases such as potassium tert-butoxide, sodium tert-butoxide, or cesium carbonate, the protocol facilitates a reversible proton abstraction at the acidic C5 or C4 positions. When conducted in a deuterated solvent environment, this exchange effectively replaces the hydrogen atom with deuterium, yielding the C5-deuterated product with exceptional selectivity. Conversely, starting with a dideutero precursor and reacting in a protic solvent allows for the selective removal of one deuterium atom to generate the C4-deuterated isomer.
This direct exchange approach offers profound advantages in terms of process efficiency and scalability. The reaction conditions are remarkably mild, operating effectively at temperatures ranging from 0°C to 80°C, which minimizes thermal degradation of sensitive substrates. The absence of transition metal catalysts eliminates the need for costly metal removal steps, thereby streamlining the downstream processing and ensuring a cleaner impurity profile. Moreover, the use of readily available bases and common organic solvents makes this method economically attractive for large-scale manufacturing. The ability to toggle between C4 and C5 labeling simply by adjusting the solvent system and starting material provides chemists with unprecedented flexibility in designing isotopic tracers for ADME (Absorption, Distribution, Metabolism, and Excretion) studies.
Mechanistic Insights into Base-Catalyzed H/D Exchange
The core mechanism driving this transformation relies on the inherent acidity of the protons attached to the triazole ring carbons. In a mono-1-substituted 1,2,3-triazole, the C5-proton is generally more acidic than the C4-proton due to the electron-withdrawing influence of the adjacent nitrogen atoms. Upon addition of a base additive like cesium carbonate or lithium tert-butoxide, the base abstracts the C5-proton to form a stabilized triazolyl anion intermediate. In the presence of a deuterium source, such as deuterated dimethyl sulfoxide (DMSO-d6) or deuterium oxide (D2O), this anion is rapidly quenched by a deuteron, resulting in the incorporation of deuterium at the C5 position. The equilibrium favors the deuterated product when a large excess of deuterated solvent is used, driving the reaction to completion with high isotopic enrichment.
For the synthesis of C4-deuterated analogs, the mechanism involves a reverse logic known as deuterium-hydrogen exchange. Starting with a 4,5-dideutero-1,2,3-triazole, the base abstracts the remaining deuterium at the C5 position more readily than the one at C4, or through thermodynamic control, allows for the selective reprotonation at C5 using a protic solvent like water or methanol. This leaves the deuterium atom intact at the C4 position. The precision of this exchange is governed by the pKa differences and the specific solvation effects of the reaction medium. Understanding these mechanistic nuances allows process chemists to fine-tune reaction parameters, such as base strength and temperature, to maximize the ratio of the desired mono-deuterated isomer while suppressing the formation of non-deuterated or di-deuterated byproducts.
How to Synthesize Selective Mono-Deuterated 1,2,3-Triazoles Efficiently
The practical implementation of this synthesis route is designed for robustness and ease of execution in standard laboratory or pilot plant settings. The general procedure involves dissolving the triazole substrate in the chosen solvent system, adding the base catalyst under an inert atmosphere to prevent moisture interference, and stirring for a defined period. Detailed operational parameters, including specific molar ratios of additives and precise temperature ramps for different substrates, are critical for achieving the reported high yields and deuteration rates. For a comprehensive, step-by-step standardized operating procedure that ensures reproducibility and safety compliance, please refer to the technical guide below.
- Dissolve the mono-1-substituted-1,2,3-triazole substrate in a suitable deuterated solvent (for C5 labeling) or non-deuterated solvent (for C4 labeling).
- Add a catalytic amount of base additive such as potassium tert-butoxide or cesium carbonate under a nitrogen atmosphere.
- Stir the reaction mixture at temperatures between 0°C and 80°C for 1 to 8 hours, followed by aqueous quenching, extraction, and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this base-catalyzed deuteration technology offers significant strategic benefits over legacy synthetic routes. The elimination of expensive transition metal catalysts and the reliance on commodity chemicals like potassium tert-butoxide and common solvents drastically reduces the raw material cost base. Furthermore, the simplified workup procedure, which typically involves standard aqueous extraction and filtration rather than complex metal scavenging, shortens the overall production cycle time. This efficiency translates directly into improved throughput and the ability to respond more agilely to fluctuating market demands for deuterated standards and intermediates.
- Cost Reduction in Manufacturing: The economic viability of producing deuterated compounds is often hindered by the high cost of deuterated reagents and low overall yields due to difficult purifications. This novel method mitigates these issues by achieving high conversion rates and excellent selectivity, which minimizes the loss of valuable deuterated solvents and starting materials. By avoiding the use of precious metal catalysts, the process removes a major cost driver and reduces the burden on waste management systems associated with heavy metal disposal. The streamlined purification process further lowers operational expenditures by reducing solvent consumption and labor hours required for chromatography.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available and stable reagents. Unlike specialized deuterated gases or unstable organometallic complexes, the bases and solvents used in this protocol are standard inventory items for most chemical manufacturers. This reduces the risk of supply disruptions caused by vendor shortages or logistical bottlenecks. Additionally, the mild reaction conditions reduce the need for specialized high-pressure or cryogenic equipment, allowing production to be distributed across a broader range of manufacturing facilities, thereby diversifying supply sources and enhancing continuity.
- Scalability and Environmental Compliance: The environmental footprint of this synthesis is notably lower than traditional methods. The absence of copper catalysts aligns with green chemistry principles by preventing heavy metal contamination of waste streams. The reaction operates at near-ambient pressures and moderate temperatures, resulting in lower energy consumption for heating and cooling. These factors facilitate easier regulatory approval for commercial scale-up, as the process generates less hazardous waste and poses fewer safety risks. The scalability from milligram to multi-kilogram batches is supported by the homogeneous nature of the reaction mixture, ensuring consistent quality and performance regardless of batch size.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these selective mono-deuterated triazoles. The answers are derived directly from the experimental data and technical specifications provided in the underlying patent literature, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for integrating these materials into your drug discovery pipelines or analytical workflows effectively.
Q: What is the primary advantage of this base-catalyzed method over traditional click chemistry for deuteration?
A: Traditional copper-catalyzed azide-alkyne cycloaddition often results in 4,5-dideutero mixtures that are difficult to separate. This novel method allows for precise regioselective labeling at either the C4 or C5 position exclusively, significantly simplifying purification and improving isotopic purity.
Q: Can this method be scaled for commercial production of deuterated drug intermediates?
A: Yes, the process utilizes mild reaction conditions (0-80°C) and commercially available bases without requiring expensive transition metal catalysts. This makes the protocol highly amenable to scale-up from gram to multi-kilogram quantities while maintaining cost efficiency.
Q: What types of substituents are compatible with this deuteration protocol?
A: The method demonstrates broad substrate scope, tolerating various groups at the N1-position including alkyl, aryl, substituted phenyl, and benzyl groups. Functional groups such as methoxy, methyl, bromo, chloro, and cyano remain intact during the exchange process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Deuterated 1,2,3-Triazole Supplier
As the demand for deuterated pharmaceuticals continues to surge, partnering with a technically proficient CDMO is essential for navigating the complexities of isotope labeling. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition seamlessly from benchtop discovery to full-scale manufacturing. Our facility is equipped with rigorous QC labs capable of verifying isotopic purity and stringent purity specifications, guaranteeing that every batch of deuterated triazole meets the exacting standards required for clinical and preclinical applications.
We invite you to leverage our expertise to optimize your supply chain for deuterated intermediates. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecular targets. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that your project timelines and budget constraints are met with precision and reliability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
