Revolutionizing Chiral Beta Amino Acid Synthesis: A Deep Dive into Unprotected Asymmetric Hydrogenation
The pharmaceutical industry continuously seeks more efficient pathways to construct chiral building blocks, particularly beta-amino acid derivatives which serve as critical scaffolds for bioactive molecules and drug candidates. Patent CN1972898A introduces a transformative methodology for the preparation of enantiomerically enriched beta-amino acid derivatives wherein the amino group remains unprotected throughout the synthesis. This innovation addresses a longstanding bottleneck in asymmetric synthesis, where the necessity for amine protection groups traditionally inflates process mass intensity and extends production timelines. By leveraging a rhodium metal precursor complexed with specific chiral mono- or bisphosphine ligands, this technology enables the direct enantioselective hydrogenation of prochiral beta-amino acrylic acids. For R&D directors and process chemists, this represents a paradigm shift towards atom-economical and step-efficient manufacturing strategies that align with modern green chemistry principles.
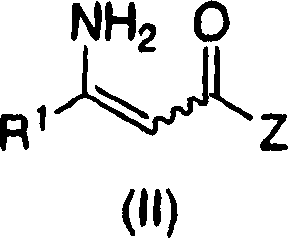
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral beta-amino acids via catalytic hydrogenation has been plagued by the interference of free amine groups with the metal catalyst. Free amines possess lone pair electrons that can strongly coordinate to transition metals like rhodium, effectively poisoning the catalyst and halting the reaction or drastically reducing enantioselectivity. Consequently, conventional protocols mandate a multi-step sequence involving the initial protection of the amine functionality, often using bulky groups like Boc or Cbz, followed by the hydrogenation step, and finally a deprotection stage to reveal the free amine. This three-step sequence not only consumes additional reagents and solvents but also generates significant chemical waste, complicating purification and increasing the overall cost of goods. Furthermore, the introduction and removal of protecting groups can sometimes lead to racemization or side reactions, compromising the optical purity of the final active pharmaceutical ingredient.
The Novel Approach
The methodology disclosed in CN1972898A circumvents these inefficiencies by utilizing a specialized catalyst system capable of tolerating and selectively reducing the carbon-carbon double bond in the presence of a free amine. The core of this innovation lies in the precise tuning of the steric and electronic properties of the chiral phosphine ligands coordinated to the rhodium center. By employing ligands that create a specific chiral environment, the catalyst preferentially binds the olefinic substrate over the amine nitrogen, facilitating highly enantioselective hydrogenation without the need for prior protection. This direct approach collapses the traditional three-step sequence into a single transformation, dramatically reducing the number of unit operations required. The result is a streamlined process that offers superior reactivity and excellent enantiomeric excess, often exceeding 90% ee, thereby providing a robust route for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Rhodium-Catalyzed Asymmetric Hydrogenation
The success of this unprotected hydrogenation relies heavily on the formation of a cationic or neutral rhodium complex that maintains high activity despite the potential for catalyst inhibition by the substrate's amine group. The catalytic cycle typically begins with the coordination of the chiral bisphosphine ligand to a rhodium precursor, such as [Rh(cod)Cl]2 or [Rh(cod)2]OTf, generating the active species. In the presence of hydrogen, this complex undergoes oxidative addition to form a rhodium dihydride species. The prochiral enamine substrate then coordinates to the metal center through its alkene moiety. Crucially, the steric bulk of the ligand prevents the free amine from binding in a manner that would deactivate the catalyst, instead directing the hydride transfer to specific faces of the double bond. This facial selectivity is what dictates the stereochemical outcome, ensuring the formation of the desired (R) or (S) enantiomer with high fidelity.
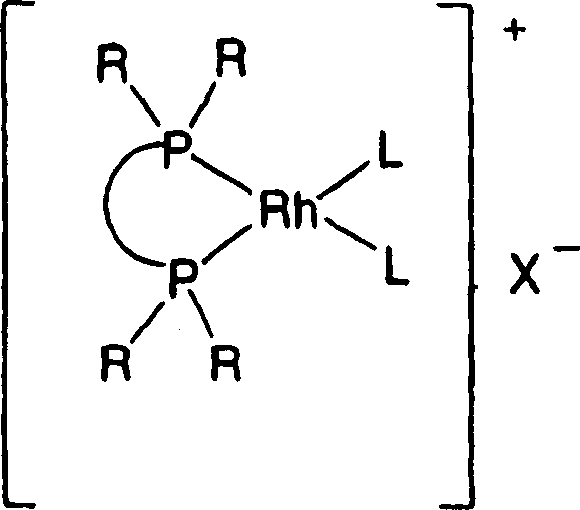
Impurity control in this system is inherently superior due to the reduced number of synthetic steps. In traditional protected routes, impurities can arise from incomplete protection, side reactions during deprotection, or carryover of protecting group fragments. By eliminating these stages, the impurity profile of the final beta-amino acid derivative is significantly cleaner. The patent highlights that the reaction can be conducted in various organic solvents, including fluorinated alcohols like 2,2,2-trifluoroethanol (TFE), which may further enhance reaction rates and selectivity by stabilizing charged intermediates or modifying the solubility of the substrate. The ability to achieve high optical purity, typically greater than 70% ee and often exceeding 90% ee, directly from the hydrogenation reactor minimizes the need for downstream resolution processes, which are often costly and yield-limiting. This mechanistic robustness makes the process highly attractive for the production of high-purity pharmaceutical intermediates required for stringent regulatory filings.
How to Synthesize Enantiomerically Enriched Beta Amino Acid Derivatives Efficiently
The practical implementation of this technology involves dissolving the prochiral beta-amino acrylic acid derivative in a suitable solvent such as methanol, ethanol, or trifluoroethanol. A catalytic amount of the rhodium-ligand complex is introduced, either generated in situ or added as a pre-formed solution. The reaction mixture is then pressurized with hydrogen gas, typically ranging from ambient pressure up to 1500 psig, and heated to temperatures between 10°C and 90°C, with a preferred range of 45°C to 65°C for optimal kinetics. The detailed standardized synthesis steps, including specific molar ratios, workup procedures, and purification techniques described in the patent examples, are outlined below to guide process development teams in replicating these results.
- Prepare the prochiral beta-amino acrylic acid derivative substrate with an unprotected amine group.
- Complex a rhodium metal precursor with a suitable chiral mono- or bisphosphine ligand to form the active catalyst.
- Conduct enantioselective hydrogenation in a suitable organic solvent under controlled temperature and pressure to yield the chiral product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this unprotected hydrogenation technology translates into tangible operational efficiencies and cost optimizations. The most immediate impact is the drastic simplification of the supply chain for raw materials. By removing the requirement for protecting group reagents and the associated bases or acids needed for their installation and removal, the bill of materials is streamlined. This reduction in material complexity lowers the risk of supply disruptions caused by the shortage of niche protecting agents. Furthermore, the elimination of two entire synthetic steps (protection and deprotection) significantly reduces the total processing time per batch. This acceleration allows manufacturing facilities to increase throughput without expanding physical infrastructure, effectively enhancing capacity utilization rates.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the reduction in step count. Each chemical step in a synthesis adds labor, energy, solvent, and waste disposal costs. By collapsing a three-step sequence into one, the overall manufacturing cost is substantially decreased. Additionally, the high enantioselectivity achieved reduces or eliminates the need for chiral chromatography or crystallization resolutions, which are among the most expensive purification techniques in the industry. The use of efficient rhodium catalysts, often at low loadings (e.g., 0.01 to 10 mol%), ensures that precious metal costs remain manageable while delivering high yields. This holistic reduction in operational expenditure supports significant cost reduction in API manufacturing, allowing for more competitive pricing in the global market.
- Enhanced Supply Chain Reliability: Simplifying the synthetic route inherently improves supply chain resilience. Fewer steps mean fewer points of failure where a reaction could stall or produce off-spec material. The robustness of the rhodium-catalyzed hydrogenation, which tolerates various functional groups and solvent systems, ensures consistent production quality. Moreover, the starting materials, such as beta-keto esters and ammonia sources, are commodity chemicals with stable and reliable global supply networks. This reliance on readily available feedstocks mitigates the risk of bottlenecks associated with custom-synthesized protected intermediates. Consequently, manufacturers can offer more reliable lead times and maintain consistent inventory levels, fulfilling the demand for a reliable pharmaceutical intermediate supplier.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this process aligns well with sustainability goals. The reduction in solvent usage and chemical waste generation lowers the environmental footprint of the manufacturing process. Hydrogenation reactions are generally clean, producing water as the only byproduct when reducing simple olefins, although in this specific case, the byproduct profile depends on the substrate. The ability to run the reaction at moderate temperatures and pressures facilitates safe scale-up from laboratory to pilot and commercial scales. The process avoids the use of hazardous reagents often associated with protection chemistry, such as phosgene derivatives or strong alkylating agents. This safer profile simplifies regulatory compliance and waste treatment, making it easier to obtain necessary environmental permits for commercial scale-up of complex chiral intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric hydrogenation technology. These answers are derived directly from the experimental data and scope defined within the patent documentation, providing clarity on the process capabilities and limitations for potential partners and licensees.
Q: What is the primary advantage of this hydrogenation process over conventional methods?
A: The primary advantage is the ability to hydrogenate substrates with unprotected amine groups. Conventional methods typically require protection and deprotection steps, which add complexity, cost, and waste. This method eliminates those steps, significantly streamlining the synthesis.
Q: Which catalyst systems are effective for this transformation?
A: The process utilizes rhodium metal precursors complexed with chiral mono- or bisphosphine ligands. Specific ligand classes such as Biphemp, Josiphos, and various biaryl phosphines have demonstrated high enantioselectivity and yield in this system.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the patent explicitly states suitability for pilot plant or industrial scale. The reaction conditions are relatively mild (temperatures around 20-65°C and pressures up to 1500 psig), making it adaptable for commercial production environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta Amino Acid Derivatives Supplier
The technological advancements detailed in CN1972898A underscore the potential for highly efficient, cost-effective production of chiral beta-amino acids, which are indispensable in modern drug discovery and development. NINGBO INNO PHARMCHEM stands at the forefront of translating such innovative academic and patent literature into robust commercial realities. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop curiosity to market-ready supply. Our facilities are equipped with state-of-the-art high-pressure hydrogenation reactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of beta-amino acid derivatives meets the highest international standards for pharmaceutical applications.
We invite you to leverage our technical expertise to optimize your supply chain and reduce your time-to-market. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific molecule, identifying opportunities to apply this unprotected hydrogenation strategy to your portfolio. We encourage you to contact our technical procurement team today to request specific COA data for similar compounds and discuss route feasibility assessments. Let us help you secure a reliable pharmaceutical intermediate supplier partnership that drives value through innovation and operational excellence.
