Advanced Regioselective Acylation of 2-Methylpiperazine for Commercial API Intermediate Production
The pharmaceutical industry constantly seeks robust synthetic routes for complex heterocyclic intermediates, particularly those serving as key scaffolds for antiviral therapeutics. Patent CN100448849C introduces a transformative methodology for the preparation of 4-acyl substituted-2-methylpiperazine compounds, a critical structural motif found in potent HIV-1 inhibitors such as BMS-378806. This innovation addresses a longstanding challenge in medicinal chemistry: achieving high regioselectivity during the monoacylation of asymmetric piperazine rings without resorting to hazardous reagents. By leveraging N,N'-carbonyldiimidazole (CDI) as an activating agent, the disclosed process effectively replaces dangerous organo-aluminum catalysts, thereby streamlining the production workflow for high-purity pharmaceutical intermediates. The technical breakthrough lies not only in the safety profile but also in the operational simplicity, allowing for precise control over the acylation site on the piperazine ring. As a reliable pharmaceutical intermediate supplier, understanding these mechanistic nuances is vital for ensuring consistent quality in downstream API synthesis. The following analysis dissects the chemical ingenuity of this patent and its profound implications for cost reduction in API manufacturing.
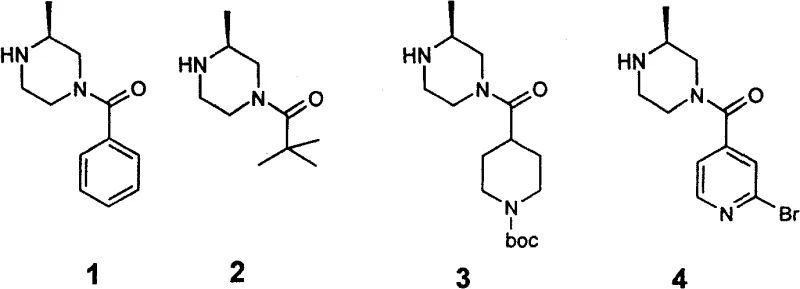
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the selective monoacylation of 2-methylpiperazine has been plagued by significant technical and safety hurdles that impede efficient commercial scale-up of complex heterocyclic intermediates. Traditional protocols often rely on strong Lewis acids or organometallic reagents, most notably diethylaluminum chloride, to direct the acylation to the desired nitrogen atom. However, diethylaluminum chloride is notoriously pyrophoric, igniting spontaneously upon contact with air, which necessitates specialized inert atmosphere equipment and rigorous safety protocols that drastically inflate operational expenditures. Furthermore, conventional acylation methods lacking specific activation strategies frequently result in poor regioselectivity, yielding inseparable mixtures of 1-acyl and 4-acyl isomers alongside unwanted diacylated byproducts. This lack of selectivity forces manufacturers to employ resource-intensive purification techniques, such as preparative HPLC or repeated recrystallization, which severely diminish overall process yields and extend production timelines. The inherent instability and high cost of these traditional reagents create a bottleneck for reducing lead time for high-purity pharmaceutical intermediates, making the supply chain vulnerable to disruptions and regulatory scrutiny regarding hazardous material handling.
The Novel Approach
In stark contrast, the methodology outlined in patent CN100448849C utilizes N,N'-carbonyldiimidazole (CDI) to activate carboxylic acids prior to coupling with the piperazine substrate, representing a paradigm shift towards greener and safer chemistry. This novel approach capitalizes on the formation of a reactive acyl-imidazole intermediate, which reacts selectively with the less sterically hindered nitrogen atom of the 2-methylpiperazine ring. By avoiding the use of pyrophoric diethylaluminum chloride, the process eliminates the need for extreme safety measures, allowing for operation in standard glass-lined reactors with simplified workup procedures. The use of CDI, a stable and commercially abundant solid, ensures consistent reagent quality and simplifies logistics, directly contributing to cost reduction in pharmaceutical intermediate manufacturing. Moreover, the reaction conditions are mild, typically employing common solvents like tetrahydrofuran at moderate reflux temperatures, which enhances the energy efficiency of the process. This strategic substitution of reagents not only mitigates safety risks but also improves the purity profile of the final product by minimizing side reactions associated with harsh Lewis acids, thereby delivering a superior intermediate for subsequent drug synthesis steps.
Mechanistic Insights into CDI-Mediated Regioselective Acylation
The core of this technological advancement lies in the exploitation of steric hindrance effects to drive regioselectivity, a concept that is critical for R&D teams aiming to optimize impurity profiles. In the 2-methylpiperazine scaffold, the presence of the methyl group at the 2-position creates a distinct spatial environment around the two nitrogen atoms. The nitrogen at the 1-position is adjacent to the methyl group, experiencing significant steric repulsion that hinders the approach of bulky electrophiles. Conversely, the nitrogen at the 4-position is distal to the methyl substituent and remains relatively unhindered. When the carboxylic acid is activated by CDI to form an acyl-imidazole, the resulting electrophile possesses sufficient steric bulk—especially when using aromatic or branched aliphatic acids like benzoic acid or pivalic acid—to be excluded from the crowded 1-position. Consequently, the nucleophilic attack occurs almost exclusively at the 4-position nitrogen. This steric differentiation is the key mechanism that allows for the isolation of single isomers without the need for complex protecting group strategies, ensuring that the high-purity 4-acyl-2-methylpiperazine is obtained directly from the reaction mixture.
Furthermore, the choice of the acylating agent plays a pivotal role in determining the success of this selective transformation. The patent data indicates that while small acyl groups like acetyl might lead to mixtures due to insufficient steric differentiation, bulky groups such as benzoyl or pivaloyl enforce strict selectivity for the 4-position. This mechanistic understanding allows chemists to predict the outcome of acylations with various carboxylic acids, facilitating the rapid development of diverse analog libraries. The reaction proceeds through a well-defined pathway where the CDI first reacts with the carboxylic acid to release carbon dioxide and imidazole, generating the active acylating species. This species then undergoes nucleophilic substitution with the piperazine, releasing a second equivalent of imidazole as a benign byproduct. The absence of metal residues in the final product is a significant advantage, as it removes the necessity for expensive and time-consuming metal scavenging steps, which are often required when transition metal catalysts are employed in similar transformations.
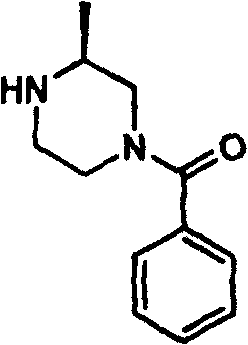
How to Synthesize 4-Benzoyl-2-Methylpiperazine Efficiently
The practical implementation of this synthesis route is straightforward and amenable to standard laboratory and pilot plant operations, making it an ideal candidate for technology transfer. The process begins with the activation of the carboxylic acid, such as benzoic acid, using a stoichiometric amount of CDI in a dry aprotic solvent like tetrahydrofuran. This activation step is crucial and typically requires heating under reflux to ensure complete conversion to the acyl-imidazole intermediate. Once activation is confirmed, the 2-methylpiperazine is introduced to the reaction mixture, where the temperature is maintained to facilitate the coupling reaction. The detailed standardized synthesis steps below outline the precise molar ratios and conditions required to achieve optimal yields and purity, serving as a foundational protocol for process chemists.
- Activate the carboxylic acid by reacting it with N,N'-carbonyldiimidazole (CDI) in a dry organic solvent such as tetrahydrofuran under reflux conditions.
- Introduce the 2-methylpiperazine substrate to the activated acyl-imidazole intermediate solution to initiate the nucleophilic attack.
- Maintain the reaction mixture at elevated temperatures for several hours to ensure complete conversion, followed by purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this CDI-mediated synthesis route offers tangible strategic benefits that extend beyond mere chemical elegance. The primary advantage is the substantial reduction in raw material costs and hazard management expenses. By replacing diethylaluminum chloride with CDI, companies can eliminate the high costs associated with the storage, handling, and disposal of pyrophoric materials. This shift significantly lowers the barrier to entry for manufacturing these intermediates, as it does not require specialized containment facilities or highly trained personnel for hazardous material handling. Additionally, the reagents involved, specifically CDI and common carboxylic acids, are commodity chemicals with stable global supply chains, ensuring consistent availability and shielding production schedules from the volatility often seen with specialty organometallic reagents.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous organo-aluminum reagents directly translates to lower raw material expenditures and reduced waste disposal costs. The process operates under mild conditions with simple workup procedures, minimizing energy consumption and labor hours required for purification. Furthermore, the high regioselectivity reduces the loss of valuable starting materials to isomeric byproducts, maximizing the effective yield per batch and optimizing the overall cost of goods sold.
- Enhanced Supply Chain Reliability: Utilizing stable, non-hazardous reagents like CDI mitigates the risk of shipping delays and regulatory hold-ups that frequently plague the transport of dangerous goods. The robustness of the reaction conditions ensures that production can proceed with minimal interruption, providing a steady flow of intermediates to downstream API manufacturing sites. This reliability is crucial for maintaining continuous supply lines for critical medications, reducing the risk of stockouts and ensuring market availability.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory synthesis to multi-ton commercial production without significant re-engineering. The absence of heavy metals and pyrophoric substances simplifies environmental compliance and wastewater treatment, aligning with increasingly stringent global sustainability standards. This green chemistry approach not only future-proofs the manufacturing process against tightening regulations but also enhances the corporate sustainability profile of the producing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on process capabilities and limitations. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this technology into their existing production portfolios.
Q: Why is N,N'-carbonyldiimidazole (CDI) preferred over diethylaluminum chloride for this acylation?
A: CDI is significantly safer and cheaper than diethylaluminum chloride, which is pyrophoric and difficult to handle. CDI eliminates severe safety hazards while maintaining high regioselectivity for the 4-position nitrogen.
Q: What drives the regioselectivity towards the 4-position nitrogen in 2-methylpiperazine?
A: The selectivity is driven by steric hindrance. The methyl group at the 2-position creates significant steric bulk around the 1-position nitrogen, making the 4-position nitrogen more accessible to bulky acylating agents like benzoic acid derivatives.
Q: Can this method be scaled for industrial production of HIV inhibitors?
A: Yes, the process uses common solvents like THF and stable reagents, making it highly suitable for commercial scale-up. It avoids the complex safety infrastructure required for organo-aluminum reagents, facilitating easier industrial implementation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Acyl-2-Methylpiperazine Supplier
The technical potential of the CDI-mediated acylation method described in patent CN100448849C represents a significant opportunity for optimizing the supply chain of critical antiviral intermediates. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent, high-quality materials regardless of volume. Our facility is equipped with rigorous QC labs and advanced analytical instrumentation to verify stringent purity specifications, guaranteeing that every batch of 4-acyl-2-methylpiperazine meets the exacting standards required for pharmaceutical applications. We are committed to translating innovative patent technologies into reliable commercial realities.
We invite potential partners to engage with our technical team to explore how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this safer, more efficient protocol. We encourage you to contact our technical procurement team today to obtain specific COA data and comprehensive route feasibility assessments tailored to your production needs, ensuring a seamless integration of these high-value intermediates into your supply chain.
