Advanced Iron-Carborane Pincer Catalysts for Efficient Pharmaceutical Intermediate Manufacturing
Advanced Iron-Carborane Pincer Catalysts for Efficient Pharmaceutical Intermediate Manufacturing
The landscape of organometallic catalysis is undergoing a significant transformation, driven by the urgent need for sustainable, cost-effective, and robust synthetic methodologies in the fine chemical and pharmaceutical sectors. A pivotal advancement in this domain is detailed in Chinese Patent CN111732612A, which discloses a novel class of iron complexes featuring bisphosphine meta-carborane ligands. These unique Pincer-type complexes leverage the exceptional electronic and steric properties of the carborane cage to stabilize the iron center, resulting in catalysts that are not only highly active but also remarkably stable under ambient conditions. For R&D directors and process chemists, this represents a paradigm shift away from expensive, air-sensitive noble metals toward earth-abundant iron systems that do not compromise on performance. The structural integrity provided by the rigid carborane backbone ensures that the catalytic active site remains protected, facilitating efficient turnover in challenging C-H activation and condensation reactions.
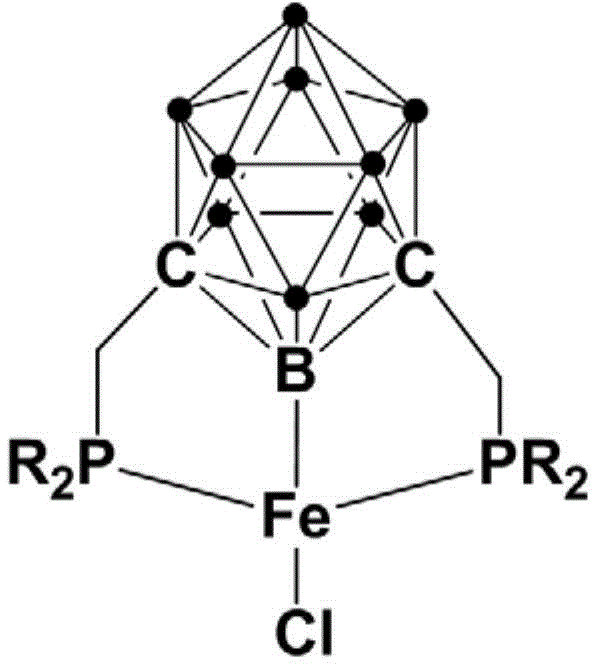
Furthermore, the versatility of this catalytic platform extends to the synthesis of trans-2-alkenylazaarenes, which are critical scaffolds in numerous bioactive molecules and pharmaceutical intermediates. The patent highlights that these iron complexes can be synthesized via a straightforward, one-pot procedure involving the lithiation of meta-carborane followed by phosphorylation and metallation. This streamlined preparation method contrasts sharply with the multi-step, protection-deprotection sequences often required for traditional Pincer ligands. By integrating the unique three-dimensional aromaticity of the carborane cluster with the coordinating power of phosphine arms, the resulting complexes offer a tunable electronic environment that can be optimized for specific substrate classes. This technological breakthrough positions the bisphosphine meta-carborane iron complex as a cornerstone for next-generation green chemistry applications, particularly where supply chain resilience and cost containment are paramount.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
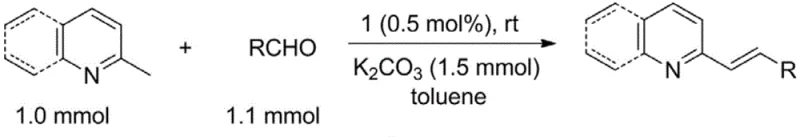
Historically, the synthesis of 2-alkenylazaarenes, which serve as key precursors for drugs like asthma medications and other biologically active agents, has been plagued by significant inefficiencies. The classical approach typically involves the condensation of 2-methylazaarenes with aromatic aldehydes in the presence of large excesses of acetic anhydride and acetic acid. This legacy method necessitates harsh reaction conditions, often requiring temperatures between 120°C and 160°C to drive the reaction to completion. Such high thermal energy input not only escalates operational costs but also poses safety risks and limits the compatibility of sensitive functional groups. Moreover, the reliance on stoichiometric amounts of corrosive acids generates substantial chemical waste, complicating downstream processing and environmental compliance. While some transition metal-catalyzed alternatives have been explored, they frequently depend on precious metals like palladium or ruthenium, which are subject to volatile market pricing and supply constraints. Additionally, many of these sophisticated catalysts are extremely sensitive to air and moisture, demanding rigorous inert atmosphere techniques that are difficult to maintain on a large industrial scale.
The Novel Approach
In stark contrast, the technology encapsulated in CN111732612A introduces a mild, room-temperature protocol that fundamentally alters the economic and operational dynamics of this transformation. By utilizing the novel bisphosphine meta-carborane iron complexes, the direct condensation of 2-methylazaarenes and aromatic aldehydes proceeds efficiently at ambient temperature in common solvents like toluene. This elimination of high-temperature requirements drastically reduces energy consumption and allows for the use of simpler, less specialized reaction vessels. The catalyst loading is exceptionally low, often in the range of 0.5 mol% or less, yet it delivers high yields and excellent stereoselectivity for the trans-isomer. The robustness of the iron-carborane framework means the catalyst tolerates the presence of air and moisture far better than its noble metal counterparts, simplifying the operational workflow for manufacturing teams. This novel approach effectively decouples high performance from high cost and complexity, offering a scalable solution that aligns perfectly with the principles of green chemistry and sustainable manufacturing.
Mechanistic Insights into Iron-Carborane Pincer Catalysis
The exceptional performance of these catalysts can be attributed to the unique mechanistic features imparted by the meta-carborane Pincer ligand architecture. The carborane cage acts as a rigid, electron-withdrawing scaffold that enforces a specific geometry around the iron center, preventing the formation of inactive oligomeric species that often deactivate homogeneous catalysts. The tridentate coordination mode, involving the central carbon of the carborane and the two phosphine arms, creates a highly stable chelate effect. This stability is crucial for maintaining the active oxidation state of the iron throughout the catalytic cycle, which likely involves the deprotonation of the 2-methyl group of the azaarene by the base (K2CO3) to form a reactive nucleophilic species. The iron center then facilitates the coupling with the aldehyde electrophile, promoting the elimination of water to form the alkene bond. The steric bulk of the substituents on the phosphine atoms (such as isopropyl or phenyl groups) can be tuned to optimize the approach of the substrates, thereby enhancing both reaction rates and selectivity.
From an impurity control perspective, the well-defined nature of the Pincer complex ensures a consistent active site, which minimizes the formation of side products often seen with ill-defined heterogeneous catalysts or simple iron salts. The reaction mechanism favors the formation of the thermodynamically more stable trans-alkene isomer, reducing the burden on purification teams who would otherwise need to separate cis/trans mixtures. Furthermore, the stability of the ligand prevents leaching of the metal into the product stream, a critical consideration for pharmaceutical applications where residual metal limits are strictly regulated. The ability of the carborane cage to delocalize electron density also helps in stabilizing transition states that might otherwise be too high in energy, allowing the reaction to proceed rapidly even at room temperature. This deep understanding of the structure-activity relationship allows process chemists to confidently predict the behavior of the catalyst across a wide range of substrate derivatives, ensuring robust process performance.
How to Synthesize Bisphosphine Meta-Carborane Iron Complex Efficiently
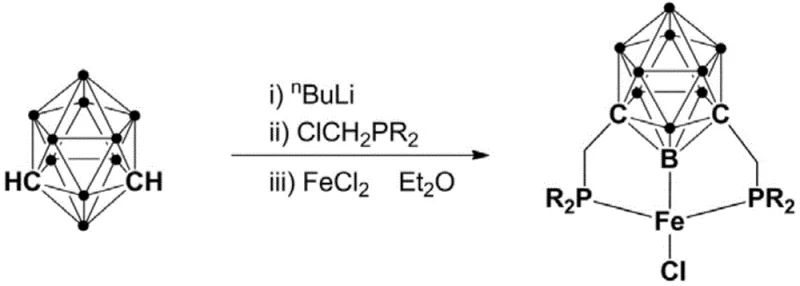
The preparation of these high-value catalysts is designed for operational simplicity, utilizing a one-pot strategy that minimizes solvent exchanges and isolation steps. The process begins with the controlled lithiation of meta-carborane using n-butyllithium at low temperatures to ensure regioselectivity, followed by the introduction of chloromethylphosphines to install the coordinating arms. The final metallation step with ferrous chloride completes the assembly of the Pincer complex. This streamlined synthetic route is amenable to scale-up, as it avoids the use of exotic reagents or extreme conditions. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below.
- Lithiation of meta-carborane using n-BuLi at low temperature (-5 to 5°C) followed by warming to room temperature.
- Addition of halogenated phosphine (e.g., ClCH2PR2) to form the bisphosphine ligand precursor in situ.
- Metallation with FeCl2 in diethyl ether to yield the final trivalent iron pincer complex after purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this iron-carborane technology offers compelling strategic advantages that extend beyond mere technical performance. The shift from precious metals to iron represents a fundamental decoupling from the volatile pricing dynamics of the platinum group metals market. Since iron is one of the most abundant and inexpensive metals on earth, the raw material cost for the catalyst core is negligible compared to palladium or rhodium systems. Furthermore, the ligand synthesis relies on commercially available phosphines and carboranes, ensuring a stable and diversified supply base. The robustness of the catalyst also translates into reduced logistical costs; because the complexes are air-stable, they do not require expensive cold-chain shipping or specialized inert-gas packaging, simplifying inventory management and reducing the risk of spoilage during transit.
- Cost Reduction in Manufacturing: The implementation of this catalytic system drives significant cost savings through multiple vectors. Primarily, the replacement of expensive noble metals with earth-abundant iron drastically lowers the bill of materials for the catalyst itself. Additionally, the ability to run reactions at room temperature eliminates the energy costs associated with heating reactors to over 100°C, which is standard for conventional methods. The high activity of the catalyst allows for very low loading levels, meaning less catalyst mass is needed per kilogram of product, further reducing waste disposal costs. The simplified workup, owing to the stability of the catalyst and the high selectivity of the reaction, reduces the consumption of solvents and chromatography media during purification. Collectively, these factors contribute to a substantially lower cost of goods sold (COGS) for the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commodity chemicals and stable catalyst forms. Unlike air-sensitive catalysts that degrade upon exposure, these iron complexes can be stored on the shelf for extended periods without loss of activity, allowing manufacturers to maintain strategic stockpiles without fear of expiration. The synthesis of the catalyst itself is robust and high-yielding, ensuring that production bottlenecks are minimized. By reducing dependence on geographically concentrated sources of precious metals, companies can mitigate geopolitical risks and ensure continuous production flows. The compatibility of the catalyst with standard industrial solvents like toluene also means that existing infrastructure can be utilized without the need for costly retrofits or specialized equipment.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the mild reaction conditions and the absence of hazardous reagents like acetic anhydride in stoichiometric amounts. The reduction in corrosive waste streams simplifies effluent treatment and lowers the environmental footprint of the manufacturing site. The high atom economy of the condensation reaction, combined with the recyclability potential of the robust iron complex, aligns with increasingly stringent global environmental regulations. This ease of scale-up ensures that demand surges can be met rapidly, reducing lead times for high-purity pharmaceutical intermediates and enabling faster time-to-market for new drug candidates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic technology. These insights are derived directly from the experimental data and claims within the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the fit of this technology within your specific process development pipeline.
Q: What are the stability characteristics of this iron-carborane catalyst compared to traditional noble metal catalysts?
A: Unlike many air- and moisture-sensitive noble metal catalysts, the bisphosphine meta-carborane iron complexes described in CN111732612A exhibit exceptional stability in air and water, simplifying storage and handling protocols significantly.
Q: Can this catalytic system be applied to a broad range of substrates for drug synthesis?
A: Yes, the patent demonstrates high efficiency across various 2-methylazaarenes (such as quinolines and pyridines) and aromatic aldehydes with different electronic properties, including electron-donating and electron-withdrawing groups.
Q: Does this process require harsh reaction conditions like high temperatures?
A: No, a key advantage of this technology is the ability to perform the condensation reaction at room temperature in toluene, eliminating the need for the high temperatures (120-160°C) required by conventional acetic anhydride methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Iron-Carborane Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced organometallic catalysis in modern drug synthesis. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into industrial reality. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications for complex organometallic species, guaranteeing that every batch of catalyst meets the exacting standards required for GMP manufacturing. We understand that consistency is key in catalytic processes, and our quality assurance protocols are designed to minimize batch-to-batch variability, providing our partners with the confidence needed to validate their processes.
We invite you to collaborate with us to leverage this cutting-edge iron-carborane technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific target molecule, demonstrating exactly how this switch can improve your margins. Please contact our technical procurement team today to request specific COA data for our catalyst library and to discuss route feasibility assessments for your upcoming campaigns. Let us help you build a more resilient and cost-effective supply chain for your critical pharmaceutical intermediates.
