Advanced 2'-O-THEX Protecting Groups for Scalable Oligoribonucleotide Production
The landscape of nucleic acid therapeutics is rapidly evolving, driven by the critical need for efficient and cost-effective synthesis of oligoribonucleotides. Patent CN100354295C introduces a groundbreaking class of 2′-O-trisubstituted silyloxymethyl-ribonucleoside derivatives that address longstanding bottlenecks in RNA manufacturing. Unlike traditional protecting groups that suffer from steric hindrance or slow deprotection kinetics, these novel derivatives utilize a unique silyl architecture featuring a tertiary carbon or heteroatom ortho to the silicon atom. This structural innovation not only stabilizes the ribonucleoside during the harsh conditions of chain elongation but also facilitates rapid removal post-synthesis. For pharmaceutical developers, this represents a pivotal shift towards more robust building blocks that can withstand the rigors of automated solid-phase synthesis while maintaining high purity profiles essential for therapeutic applications.
The Limitations of Conventional Methods vs. The Novel Approach
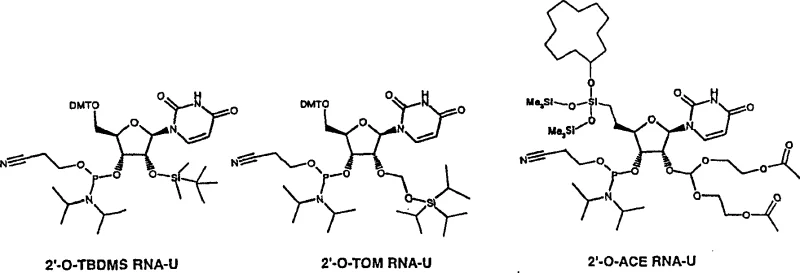
The Limitations of Conventional Methods
Historically, the chemical synthesis of oligoribonucleotides has been plagued by the inherent reactivity of the 2′-hydroxyl group, necessitating robust protection strategies that often compromise efficiency. The industry standard, 2′-O-TBDMS chemistry, while orthogonal, introduces significant steric bulk near the reactive phosphoramidite center, which drastically reduces coupling efficiency and necessitates the use of expensive activators like 5-(benzylmercapto)-1H-tetrazole. Furthermore, alternative strategies such as 2′-O-ACE chemistry, although offering lower steric hindrance, rely on acid-labile orthoesters that demand fluoride-resistant synthesizers and incompatible silica supports, creating substantial infrastructure barriers for manufacturers. Even the 2′-O-TOM approach, developed to mitigate phosphoryl migration, suffers from prolonged deprotection times, typically requiring six hours at elevated temperatures, which becomes a critical bottleneck when scaling up production for high-throughput screening or commercial drug substance manufacturing.
The Novel Approach
The technology disclosed in CN100354295C circumvents these issues by employing a 2′-O-silyloxymethyl group where at least one substituent on the silicon atom contains a tertiary carbon, such as the 1,1,2-trimethyl-propyl (THEX) group. This specific configuration creates an optimal balance of steric protection and lability. The acetal nature of the bond between the nucleoside and the protecting group prevents the notorious 2′-3′ phosphoryl migration observed in conventional silyl ethers, ensuring sequence fidelity. Moreover, the electronic and steric properties of the THEX group allow for deprotection under mild conditions—specifically, room temperature treatment with fluoride for merely 30 minutes. This dramatic reduction in processing time, coupled with the elimination of complex distillation steps during reagent preparation, offers a streamlined pathway that significantly lowers the barrier to entry for reliable oligoribonucleotide intermediate supplier operations.
Mechanistic Insights into Electrophilic Activation and Organometallic Coupling
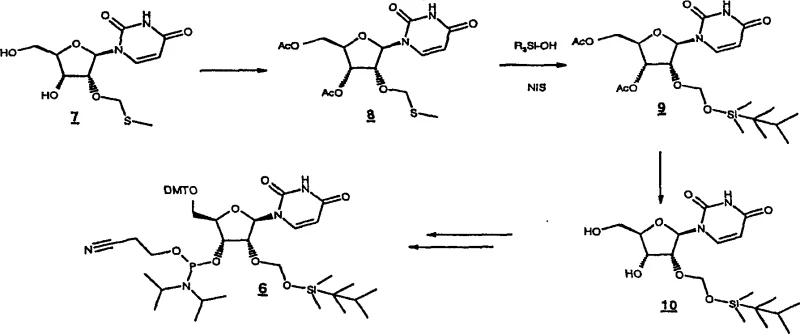
The synthesis of these advanced derivatives relies on two distinct yet complementary mechanistic pathways that showcase sophisticated control over regioselectivity. The first pathway utilizes an organometallic approach where a 5′-O-protected ribonucleoside reacts with a chloromethyl-silyl ether in the presence of a tin catalyst, such as dibutyltin dichloride. This Lewis acid coordination activates the sugar ring, facilitating the nucleophilic attack of the 2′-hydroxyl group over the 3′-hydroxyl, although chromatographic separation is still required to isolate the desired 2′-isomer. The second, more novel pathway involves the initial installation of a 2′-O-methylthiomethyl group, which serves as a temporary placeholder. This thioether is then subjected to electrophilic activation using reagents like N-iodosuccinimide (NIS) in the presence of a silanol. This activation generates a highly reactive sulfonium intermediate that is rapidly displaced by the silanol oxygen, effectively transferring the bulky silyl group to the 2′-position with high specificity. This mechanism avoids the non-selective steps associated with direct silylation and prevents the formation of difficult-to-remove impurities.
Impurity control is inherently built into the molecular design of the THEX protecting group. The presence of the tertiary carbon ortho to the silicon atom modulates the hydrolytic stability of the silyl ether, making it resistant to accidental cleavage during the acidic detritylation cycles of DNA/RNA synthesis. However, it remains sufficiently labile to be cleaved by fluoride ions without affecting the integrity of the internucleotide phosphate bonds. This orthogonality is crucial for minimizing side reactions such as depurination or backbone degradation, which are common failure modes in long-chain RNA synthesis. By preventing the migration of the protecting group to the adjacent 3′-oxygen, the process ensures that the final crude product profile is cleaner, reducing the burden on downstream purification processes like HPLC and increasing the overall yield of the full-length oligomer.
How to Synthesize 2'-O-THEX Uridine Efficiently
The preparation of these high-value building blocks follows a logical progression designed for industrial scalability. The process begins with the selective protection of the 5′-hydroxyl group, followed by the critical installation of the 2′-O-THEX moiety using either the organometallic or thiomethyl displacement method described previously. Once the 2′-position is secured, the 3′-hydroxyl is converted into a phosphoramidite, rendering the molecule ready for automated coupling. The detailed standardized synthetic steps, including specific molar ratios, solvent choices like dichloroethane, and workup procedures involving sodium bicarbonate washes, are outlined below to guide process chemists in replicating this high-efficiency route.
- Protect the 5'-hydroxyl group of the ribonucleoside with a dimethoxytrityl (DMT) group to ensure orthogonality during chain elongation.
- Introduce the 2'-O-THEX protecting group via organometallic catalysis using dibutyltin dichloride or via electrophilic activation of a 2'-O-methylthiomethyl intermediate using N-iodosuccinimide.
- Convert the 3'-hydroxyl group into a phosphoramidite moiety using standard phosphitylation reagents to finalize the building block for solid-phase synthesis.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology translates directly into tangible operational efficiencies and risk mitigation. The elimination of the final distillation step for the THEX-Cl reagent, which is mandatory for TOM-Cl preparation, removes a significant unit operation that typically consumes energy and requires specialized glassware. This simplification not only reduces the capital expenditure required for setting up production lines but also shortens the batch cycle time, allowing for greater flexibility in meeting fluctuating demand schedules. Furthermore, the robustness of the THEX protecting group against acidic conditions means that standard silica-based supports and common activators can be used without modification, preserving existing inventory investments and preventing supply chain disruptions caused by the need for exotic, fluoride-resistant equipment.
- Cost Reduction in Manufacturing: The streamlined synthesis of the protecting group reagent itself offers immediate cost savings by removing energy-intensive distillation processes. Additionally, the faster deprotection kinetics (30 minutes versus 6 hours) significantly increase the throughput of the synthesis instruments, allowing more batches to be processed per week without additional capital investment. This efficiency gain effectively lowers the cost per gram of the final oligonucleotide, making large-scale production of RNA therapeutics more economically viable.
- Enhanced Supply Chain Reliability: By utilizing reagents that are easier to prepare and store, manufacturers can reduce the risk of production delays caused by reagent instability or supply shortages. The compatibility of the THEX chemistry with standard solid-phase synthesis protocols ensures that production can be easily transferred between different contract manufacturing organizations (CMOs) without extensive re-validation. This interoperability strengthens the supply chain resilience, ensuring continuous availability of critical nucleic acid intermediates for clinical and commercial programs.
- Scalability and Environmental Compliance: The process operates under mild conditions and utilizes common organic solvents like dichloromethane and acetonitrile, which are well-understood in terms of waste management and recycling. The high selectivity of the thiomethyl activation route minimizes the generation of complex byproduct mixtures, simplifying wastewater treatment and reducing the environmental footprint of the manufacturing process. This aligns with modern green chemistry principles and facilitates regulatory approval by demonstrating a controlled and clean manufacturing environment.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of 2′-O-THEX chemistry in oligonucleotide production. These answers are derived directly from the experimental data and claims within the patent documentation, providing a reliable foundation for decision-making. Understanding these nuances is essential for R&D teams evaluating the transition from legacy protecting groups to this next-generation technology.
Q: What is the primary advantage of the 2'-O-THEX group over 2'-O-TOM?
A: The 2'-O-THEX group significantly accelerates the deprotection step, reducing the time from 6 hours at 35°C for TOM chemistry to just 30 minutes at room temperature, thereby enhancing throughput.
Q: Does the new synthesis route require distillation of the chlorinating reagent?
A: No, unlike the preparation of TOM-Cl, the synthesis of THEX-Cl does not require a final distillation step prior to reaction with the ribonucleoside, simplifying the process and reducing operational costs.
Q: Is this technology suitable for automated solid-phase synthesis?
A: Yes, the derivatives are specifically designed for automated oligoribonucleotide synthesis, offering coupling yields comparable to oligodeoxynucleotides and compatibility with standard activators.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2'-O-THEX Uridine Supplier
As the demand for RNA-based therapeutics continues to surge, the ability to produce high-quality building blocks at scale becomes a decisive competitive advantage. NINGBO INNO PHARMCHEM leverages deep expertise in nucleic acid chemistry to offer these advanced 2′-O-THEX protected ribonucleosides, ensuring that your projects benefit from the latest innovations in protecting group technology. Our facilities are equipped with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, guaranteeing that we can meet your volume requirements regardless of the project phase. We maintain stringent purity specifications and operate rigorous QC labs to ensure every batch meets the exacting standards required for GMP manufacturing of drug substances.
We invite you to optimize your supply chain by partnering with a team that understands both the chemistry and the commerce of nucleic acid production. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific oligonucleotide sequence needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how switching to our 2′-O-THEX intermediates can accelerate your development timelines and reduce overall manufacturing costs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
