Advanced Manufacturing of Chiral 3-Hydroxypiperidine Intermediates via Resolution and Inversion
Advanced Manufacturing of Chiral 3-Hydroxypiperidine Intermediates via Resolution and Inversion
The pharmaceutical industry continuously demands high-purity chiral building blocks to ensure the efficacy and safety of active pharmaceutical ingredients (APIs). A significant breakthrough in this domain is detailed in patent CN103864673A, which outlines a novel preparation method for chirality-1-tertbutyloxycarbonyl-3-hydroxy piperidine alongside a versatile method for chirality turning. This technology addresses critical bottlenecks in the synthesis of piperidine derivatives, offering a pathway that is not only chemically robust but also highly amenable to industrial scale-up. By leveraging a combination of classical resolution techniques and modern protective group strategies, this process ensures the delivery of both (S) and (R) enantiomers with exceptional optical purity, meeting the stringent requirements of global regulatory bodies.
For R&D directors and process chemists, the significance of this patent lies in its ability to bypass the limitations of asymmetric synthesis and enzymatic reduction, which often suffer from high costs or narrow substrate scopes. The described methodology utilizes readily available starting materials and standard reagents, such as camphorsulfonic acid and palladium on carbon, to achieve high enantiomeric excess (ee) values. Furthermore, the inclusion of a chirality inversion protocol provides a strategic safety net; if one enantiomer is preferentially obtained during resolution, the other can be accessed through a reliable chemical transformation, thereby maximizing atom economy and reducing waste in the overall manufacturing campaign.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral 3-hydroxypiperidines has relied on three primary strategies, each presenting distinct challenges for large-scale production. Asymmetric synthesis, while elegant, often requires expensive chiral ligands and transition metal catalysts that are difficult to remove to ppm levels, posing risks for downstream pharmaceutical applications. Enzymatic asymmetric reduction offers high selectivity but is frequently constrained by the stability of the biocatalyst and the need for specialized fermentation or isolation infrastructure. Perhaps most critically, traditional chemical resolution methods have struggled with the physical properties of the free base.
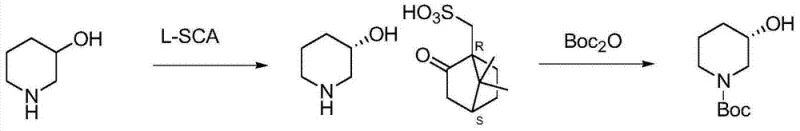
As illustrated in the background art, conventional chemical resolution often involves attempting to resolve the free 3-hydroxy piperidine directly. However, the patent explicitly notes that the free base is difficult to purify due to its high water solubility, making crystallization and isolation inefficient and prone to losing chiral integrity. This leads to lower yields and higher operational costs, as multiple recrystallization steps are required to boost optical purity. Additionally, the presence of chiral impurities in the split process can complicate the purification of the final API, necessitating more rigorous and costly analytical controls.
The Novel Approach
The innovative strategy presented in CN103864673A circumvents these issues by shifting the resolution point to the N-benzyl protected intermediate. By resolving N-benzyl-3-hydroxy piperidine as a camphorsulfonic acid salt, the process leverages the superior crystallinity and lower water solubility of the salt form compared to the free base. This modification allows for efficient separation of enantiomers through simple filtration and washing, significantly improving the practical yield and optical purity. The subsequent steps involve a streamlined debenzylation and protection sequence that consolidates multiple transformations into a single operational unit.
Moreover, the patent introduces a sophisticated chirality turning mechanism that adds immense flexibility to the supply chain. If market demand shifts or if a specific enantiomer is required that was not the primary product of the resolution, the process allows for the inversion of stereochemistry via a sulfonate intermediate. This ensures that no batch is wasted and that the manufacturer can respond dynamically to client needs without developing an entirely new synthetic route. The reaction conditions throughout this novel approach are described as gentle, avoiding extreme temperatures or pressures that could compromise equipment integrity or safety protocols in a multi-ton reactor.
Mechanistic Insights into Chiral Resolution and Inversion
The core of this technology rests on a well-defined mechanistic pathway that ensures high fidelity in stereochemical control. The initial resolution step relies on the formation of diastereomeric salts between the racemic N-benzyl-3-hydroxy piperidine and chiral camphorsulfonic acid (CSA). The difference in solubility between the (S)-camsilate and (R)-camsilate salts in solvents like isopropanol or ethyl acetate drives the enrichment of one enantiomer in the solid phase. Following isolation, the salt is treated with a base, such as sodium hydroxide or potassium carbonate, to liberate the free amine, which retains the high enantiomeric excess achieved during crystallization.
The subsequent transformation involves a tandem debenzylation and Boc-protection. Under hydrogen atmosphere with a palladium on carbon catalyst, the benzyl group is cleaved via hydrogenolysis. Simultaneously, the presence of di-tert-butyl dicarbonate (Boc2O) traps the newly formed secondary amine, preventing side reactions and stabilizing the molecule for downstream processing. This one-pot strategy minimizes handling of the reactive free amine and reduces solvent consumption. For chirality inversion, the hydroxyl group is activated by conversion to a sulfonate ester (mesylate or tosylate), creating an excellent leaving group.
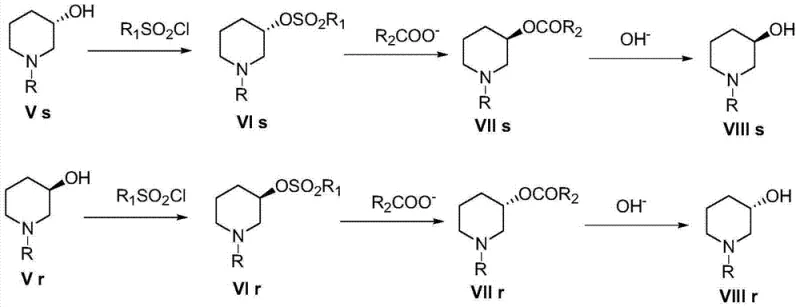
As depicted in the reaction scheme, the inversion process proceeds through a classic SN2 mechanism. The sulfonate intermediate is subjected to nucleophilic attack by a carboxylate anion, such as acetate or benzoate, which displaces the sulfonate group with inversion of configuration at the chiral center. This step is crucial for accessing the opposite enantiomer without needing to restart the resolution from the racemate. Finally, basic hydrolysis cleaves the ester bond to regenerate the hydroxyl group, completing the stereochemical flip. This mechanistic understanding allows process chemists to fine-tune reaction parameters, such as temperature and solvent polarity, to maximize the rate of inversion while minimizing elimination byproducts.
How to Synthesize 1-tertbutyloxycarbonyl-3-hydroxy piperidine Efficiently
Implementing this synthesis requires careful attention to stoichiometry and solvent selection to ensure optimal crystal formation during the resolution phase. The patent details specific molar ratios for the resolving agent, suggesting a range of 0 to 0.6 equivalents relative to the substrate, with a preferred range of 0.3 to 0.5 equivalents to balance yield and purity. The choice of solvent for the resolution step is also critical, with options including alcohols, esters, ketones, and ethers, allowing manufacturers to select based on availability and recycling capabilities. Detailed standardized synthesis steps see the guide below.
- Resolve N-benzyl-3-hydroxy piperidine using chiral camphorsulfonic acid (CSA) in solvents like isopropanol or ethyl acetate to obtain high-purity camsilate salts.
- Perform alkaline liberation to free the base, followed by palladium-carbon catalytic hydrogenation for debenzylation and simultaneous Boc protection.
- Optionally invert chirality via sulfonylation, nucleophilic substitution with carboxylate, and final hydrolysis to access the opposite enantiomer.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented process offers substantial advantages over legacy methods by simplifying the raw material portfolio and enhancing process robustness. The reliance on commodity chemicals like camphorsulfonic acid and palladium on carbon, rather than bespoke chiral ligands or fragile enzymes, significantly de-risks the supply chain. This shift ensures that production schedules are not held hostage by the lead times of specialized reagents, facilitating more predictable delivery timelines for downstream API manufacturers who depend on consistent intermediate supply.
- Cost Reduction in Manufacturing: The elimination of expensive enzymatic catalysts and complex asymmetric ligands directly translates to lower raw material costs. Furthermore, the ability to resolve the intermediate as a salt rather than a free base reduces the number of purification cycles required, leading to significant savings in solvent usage and energy consumption. The consolidation of debenzylation and protection into a single step also reduces labor hours and reactor occupancy time, driving down the overall cost of goods sold (COGS) for this critical pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: By utilizing a chemical resolution method that yields stable crystalline salts, the process mitigates the risk of batch failure associated with sensitive biological systems. The robustness of the chemistry allows for storage of intermediates at various stages, providing buffer stock flexibility. Additionally, the chirality inversion capability acts as a strategic hedge; if demand for one enantiomer spikes, inventory of the other can be converted, ensuring continuous supply continuity without the need for emergency sourcing of racemic starting materials.
- Scalability and Environmental Compliance: The reaction conditions described are mild, operating at atmospheric to moderate pressures and temperatures compatible with standard glass-lined steel reactors. This ease of scale-up reduces the capital expenditure required for specialized high-pressure equipment. Moreover, the process avoids the generation of heavy metal waste streams associated with some asymmetric catalysis methods, simplifying wastewater treatment and ensuring compliance with increasingly stringent environmental regulations regarding effluent discharge in chemical manufacturing zones.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this chiral synthesis route. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on critical process parameters. Understanding these details is essential for technology transfer teams evaluating the feasibility of adopting this method for commercial production.
Q: What resolving agents are effective for N-benzyl-3-hydroxy piperidine?
A: The patent specifies the use of chiral camphorsulfonic acid (CSA), specifically D-CSA or L-CSA, with a molar ratio ranging from 0 to 0.6 relative to the substrate.
Q: How is the chirality inverted in this process?
A: Chirality inversion is achieved by converting the hydroxyl group into a sulfonate ester (using sulfonyl chloride), followed by nucleophilic substitution with a carboxylate salt, and finally hydrolyzing the ester back to the alcohol.
Q: What are the typical reaction conditions for the debenzylation step?
A: The debenzylation and Boc protection occur simultaneously using 10% palladium on carbon under hydrogen pressure (1-30 atm) at temperatures between 0°C and 80°C.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-tertbutyloxycarbonyl-3-hydroxy piperidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the pivotal role that high-quality chiral intermediates play in the development of life-saving medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to pilot plant is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch of 1-tertbutyloxycarbonyl-3-hydroxy piperidine meets the exacting standards required for GMP manufacturing environments.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this resolution-based methodology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of chemical excellence and reliability.
