Advanced Synthesis of Trifluoromethyl-Substituted Perylene Bis-Diimides for High-Performance Organic Electronics
The landscape of organic semiconductor manufacturing is undergoing a significant transformation driven by the demand for materials with superior stability and processability. Patent CN101289447B introduces a groundbreaking methodology for the preparation of trifluoromethyl-substituted perylene bis-diimines, a class of compounds critical for next-generation optoelectronic devices. This technology addresses the longstanding limitations of traditional perylene derivatives, which often suffer from poor solubility and harsh processing requirements. By shifting the synthetic strategy from direct acid-mediated functionalization to a modular ester-based approach, this invention enables the precise introduction of fluorinated groups that dramatically enhance thermal and oxidative stability. For R&D directors and procurement specialists seeking a reliable electronic chemical supplier, understanding this pathway is essential for securing high-purity OLED material precursors that meet rigorous performance specifications.
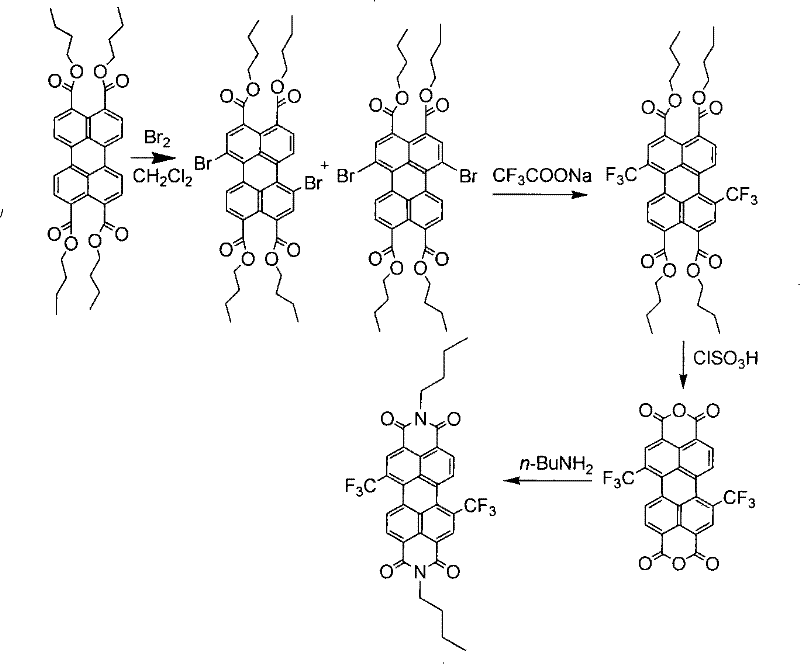
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the derivatization of the perylene core has been fraught with significant chemical engineering challenges, primarily due to the recalcitrant nature of 3,4,9,10-perylenetetracarboxylic anhydride. Conventional protocols typically necessitate direct bromination in concentrated sulfuric acid, a process that imposes severe constraints on reactor metallurgy and operational safety. The extreme acidity and corrosiveness of the medium require specialized equipment, driving up capital expenditure and maintenance costs for any facility attempting cost reduction in electronic chemical manufacturing. Furthermore, the resulting intermediates often exhibit extremely poor solubility in organic solvents, complicating purification steps and leading to broad impurity profiles that can detrimentally affect charge carrier mobility in final devices. These factors collectively create a bottleneck in the commercial scale-up of complex polymer additives and semiconductor precursors, limiting the ability of supply chains to respond flexibly to market demands.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes a soluble perylene tetracarboxylate ester as the foundational substrate, fundamentally altering the reaction kinetics and handling properties. By performing the initial bromination on the ester rather than the anhydride, the process operates in mild organic solvents such as dichloromethane or chlorobenzene at moderate temperatures ranging from 0°C to 80°C. This shift eliminates the need for hazardous concentrated sulfuric acid, thereby simplifying waste treatment protocols and enhancing operator safety. Subsequent functionalization involves a copper-catalyzed substitution where bromine atoms are replaced by trifluoromethyl or perfluoroalkyl groups. This strategy not only improves the solubility of the final perylene bis-diimide products but also allows for tunable electronic properties through the variation of fluorinated side chains, offering a versatile platform for designing high-performance organic field-effect transistors and solar cells.
Mechanistic Insights into Copper-Catalyzed Trifluoromethylation
The core innovation of this synthesis lies in the efficient copper-mediated coupling reaction that installs the critical fluorinated motifs onto the aromatic core. Specifically, the process employs cuprous iodide as a catalyst to facilitate the nucleophilic substitution of bromine atoms on the bromoperylene tetracarboxylate intermediate using sodium trifluoroacetate as the trifluoromethyl source. This reaction proceeds under inert atmosphere protection, typically argon or nitrogen, at elevated temperatures between 40°C and 160°C in polar aprotic solvents like DMF or DMSO. The mechanism likely involves the oxidative addition of the aryl bromide to the copper center, followed by transmetallation with the trifluoroacetate species and subsequent reductive elimination to forge the carbon-trifluoromethyl bond. This catalytic cycle is highly robust, tolerating the steric bulk of the perylene system while maintaining high regioselectivity, which is paramount for ensuring consistent batch-to-batch quality in high-purity OLED material production.
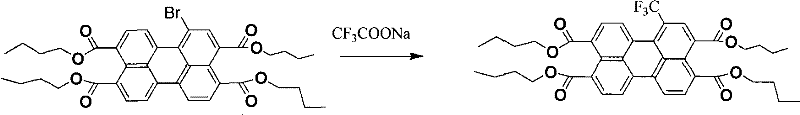
Impurity control is meticulously managed through the selection of reaction conditions and purification strategies inherent to this ester-based route. Unlike acid-slurry methods that can lead to over-bromination or sulfonation byproducts, the solution-phase bromination allows for precise stoichiometric control, yielding defined mono-, di-, tri-, or tetra-brominated intermediates. The subsequent copper-catalyzed step further refines the product profile, as unreacted bromo-species can be separated via standard chromatographic techniques before the final imidization step. The use of inorganic bases like potassium hydroxide or chlorosulfonic acid for the hydrolysis of the ester to the anhydride is also optimized to prevent degradation of the sensitive fluorinated groups. This multi-stage purification capability ensures that the final trifluoromethyl-substituted perylene bis-diimines possess the stringent purity specifications required for advanced electronic applications, minimizing trap states that could otherwise degrade device performance.
How to Synthesize Trifluoromethyl Perylene Derivatives Efficiently
The synthesis of these advanced materials follows a logical four-step sequence that balances yield with operational simplicity, making it an attractive candidate for technology transfer. The process begins with the bromination of perylene tetracarboxylate esters, followed by the crucial copper-catalyzed fluorination, hydrolysis to the anhydride, and final condensation with amines. Each step has been optimized in the patent examples to demonstrate scalability and reproducibility, providing a clear roadmap for manufacturers looking to reduce lead time for high-purity organic semiconductors. The detailed standardized synthesis steps are outlined below to guide process engineers in implementing this technology.
- Bromination of perylene tetracarboxylate esters using liquid bromine and a base like potassium carbonate in an organic solvent at 0-80°C.
- Copper-catalyzed substitution of bromine atoms with trifluoromethyl groups using sodium trifluoroacetate or perfluoroalkyl iodides at 40-160°C.
- Hydrolysis of the substituted esters to form the corresponding anhydrides using inorganic bases or chlorosulfonic acid, followed by condensation with amines.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented methodology offers substantial advantages that directly impact the bottom line and supply chain resilience. By transitioning away from hazardous concentrated acid processes to solution-phase organic chemistry, manufacturers can significantly lower their environmental compliance costs and insurance premiums associated with handling dangerous reagents. The use of commercially available starting materials, such as perylene esters and sodium trifluoroacetate, ensures a stable supply base that is not subject to the geopolitical volatility often seen with exotic reagents. Furthermore, the improved solubility of the intermediates facilitates easier filtration and crystallization, reducing solvent consumption and energy usage during downstream processing, which aligns with global sustainability goals.
- Cost Reduction in Manufacturing: The elimination of concentrated sulfuric acid removes the need for expensive acid-resistant reactors and complex neutralization waste streams, leading to drastic simplification of the production infrastructure. Additionally, the high yields reported in the patent examples for the copper-catalyzed steps indicate efficient atom economy, minimizing raw material waste. The ability to perform reactions in common solvents like DMF and ethanol further reduces procurement costs compared to specialized fluorinated solvents often required in other fluorination chemistries.
- Enhanced Supply Chain Reliability: The modular nature of this synthesis allows for the decoupling of intermediate production, meaning that brominated esters can be stockpiled as stable inventory buffers against demand fluctuations. The robustness of the copper catalysis system ensures consistent conversion rates, reducing the risk of batch failures that could disrupt delivery schedules. Sourcing partners can leverage this reliability to offer more predictable lead times, a critical factor for clients managing just-in-time inventory for high-value electronic component assembly.
- Scalability and Environmental Compliance: Solution-phase reactions are inherently easier to scale from laboratory glassware to industrial steel reactors than heterogeneous acid slurries, allowing for seamless capacity expansion without extensive re-engineering. The process generates less hazardous waste, simplifying disposal and reducing the environmental footprint of the manufacturing site. This compliance advantage is increasingly valuable as regulatory frameworks tighten around chemical manufacturing emissions and effluent quality.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on process capabilities and material properties. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating these materials into their product pipelines.
Q: How does this new synthesis method improve solubility compared to traditional perylene derivatives?
A: By introducing trifluoromethyl or long-chain perfluoroalkyl groups onto the perylene core prior to imidization, the resulting molecules exhibit significantly enhanced solubility in common organic solvents, overcoming the poor solubility issues of unsubstituted perylene tetracarboxylic anhydride.
Q: What catalysts are used in the key trifluoromethylation step?
A: The process utilizes copper-based catalysts, specifically cuprous iodide (CuI) for trifluoromethylation with sodium trifluoroacetate, or copper powder/copper-tin alloy for coupling with perfluoroalkyl iodides.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the method avoids the use of concentrated sulfuric acid for direct ring bromination, utilizing solution-phase chemistry in standard organic solvents like DMF and DMSO, which is far more amenable to safe and efficient scale-up in industrial reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Perylene Derivatives Supplier
As the demand for high-performance organic semiconductors continues to surge, partnering with an experienced CDMO is vital for translating laboratory innovations into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped to handle the specific solvent systems and catalytic requirements of this patent, adhering to stringent purity specifications and operating within rigorous QC labs to guarantee product integrity.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific application requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this fluorinated perylene platform. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, ensuring a seamless transition from development to full-scale manufacturing.
