Advanced Green Synthesis of Tea Geometrid Sex Pheromone for Scalable Agrochemical Production
Advanced Green Synthesis of Tea Geometrid Sex Pheromone for Scalable Agrochemical Production
The global shift towards sustainable agriculture has intensified the demand for biopesticides, specifically insect sex pheromones that offer species-specific control without environmental toxicity. Patent CN103880784A introduces a breakthrough methodology for synthesizing cis-6,7-epoxy-(z,z)-3,9-octadecadiene, the primary sex pheromone of the tea geometrid moth (Ectropis obliqua). This document outlines a robust, four-step synthetic pathway that leverages inexpensive linolenic acid esters as feedstock, bypassing the economic and safety hurdles of legacy processes. By employing a novel Keggin-type di-deficient silicotungstic heteropolyacid catalyst coupled with hydrogen peroxide, the invention achieves high selectivity in the critical epoxidation step while minimizing hazardous waste generation. For R&D directors and procurement strategists, this technology represents a viable pathway to secure a reliable agrochemical intermediate supplier capable of delivering high-purity pheromones at a fraction of the traditional cost.
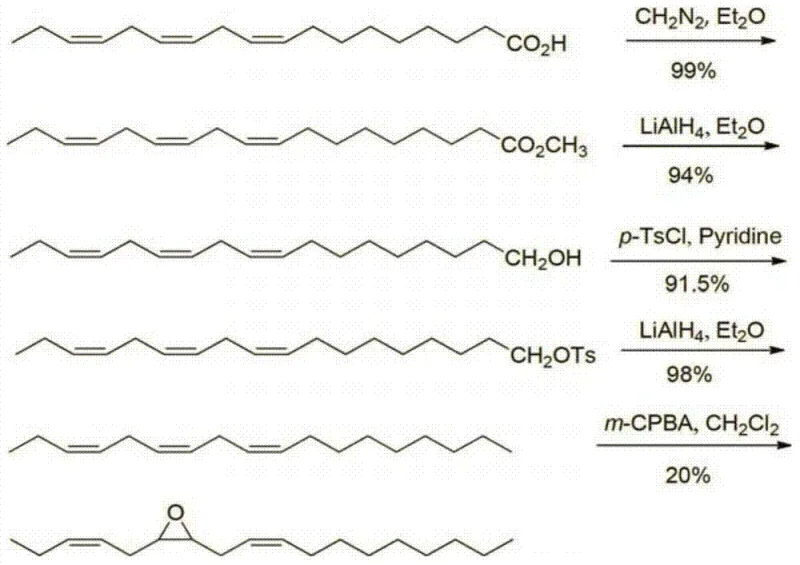
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of lepidopteran sex pheromones has been plagued by complex multi-step sequences involving expensive chiral auxiliaries or hazardous reagents. Early reports, such as those by Liu Tianlin et al., utilized linolenic acid but required dangerous reagents like diazomethane for esterification and m-chloroperoxybenzoic acid (m-CPBA) for the final epoxidation. These reagents are not only costly and thermally unstable but also generate significant amounts of chlorinated organic waste, complicating downstream purification and disposal. Furthermore, alternative routes involving Sharpless asymmetric epoxidation, while effective for obtaining optical isomers, demand strictly anhydrous conditions and expensive titanium catalysts, rendering them economically unfeasible for large-volume agricultural applications. The cumulative effect of these limitations is a high cost of goods sold (COGS) and extended lead times, which restricts the widespread adoption of pheromone-based pest management strategies in developing tea-growing regions.
The Novel Approach
The patented methodology fundamentally reengineers the synthesis by prioritizing atom economy and operational safety through the use of green oxidants and robust heterogeneous-style catalysis. Instead of relying on stoichiometric oxidants like m-CPBA, the process utilizes aqueous hydrogen peroxide activated by a Keggin-type di-deficient silicotungstic heteropolyacid tetrabutyl quaternary ammonium salt. This catalyst system operates effectively at mild temperatures between 40°C and 60°C, significantly reducing energy consumption compared to cryogenic conditions required by other methods. Crucially, the process omits intermediate purification steps between the reduction and tosylation stages, allowing for a telescoped workflow that drastically cuts solvent usage and processing time. This streamlined approach not only enhances the overall yield but also aligns perfectly with modern green chemistry principles, offering a compelling solution for cost reduction in agrochemical intermediate manufacturing.
Mechanistic Insights into Heteropoly Acid-Catalyzed Epoxidation
The core innovation of this synthesis lies in the unique electronic and structural properties of the Keggin-type di-deficient silicotungstic heteropolyacid catalyst. Unlike simple metal salts, these lacunary polyoxometalates possess vacant coordination sites that can activate hydrogen peroxide to form peroxo-species capable of transferring oxygen to the olefinic double bond. The tetrabutyl ammonium cation serves as a phase-transfer agent, enhancing the solubility of the polar catalyst in the organic reaction medium containing the lipophilic triene substrate. This biphasic compatibility ensures that the oxidation occurs selectively at the 6,7-position of the (z,z,z)-3,6,9-octadecatriene chain, minimizing over-oxidation to diols or cleavage products which are common side reactions in non-catalytic peroxide oxidations. The reaction mechanism is believed to proceed via a surface-type interaction where the alkene coordinates to the activated peroxo-tungsten center, facilitating a concerted oxygen transfer that preserves the stereochemistry of the adjacent double bonds.
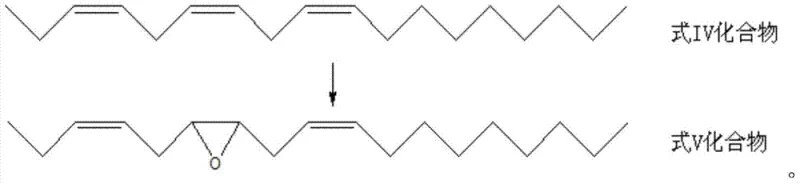
From an impurity control perspective, the specificity of this catalytic system is paramount for ensuring the biological efficacy of the final pheromone product. Insect olfactory receptors are exquisitely sensitive to stereoisomers; even minor deviations in double bond geometry or epoxide positioning can render the pheromone inactive or even act as an antagonist. The mild reaction conditions (40-60°C) prevent thermal isomerization of the sensitive Z-configured double bonds, a frequent issue in high-temperature processes. Additionally, the use of hydrogen peroxide generates water as the sole byproduct, eliminating the formation of chlorinated or sulfur-containing impurities that are difficult to separate from the non-polar pheromone matrix. This inherent cleanliness of the reaction profile simplifies the final workup, requiring only basic extraction and silica gel chromatography to achieve the stringent purity specifications demanded by regulatory bodies for biopesticide registration.
How to Synthesize cis-6,7-epoxy-(z,z)-3,9-octadecadiene Efficiently
The practical implementation of this synthesis involves a logical sequence of functional group transformations starting from readily available methyl or ethyl linolenate. The initial reduction of the ester to the corresponding alcohol sets the stage for subsequent activation, followed by conversion to a leaving group and final reductive elimination to establish the triene backbone. The final epoxidation step is the critical value-add operation where the catalyst loading and oxidant ratio must be precisely controlled to maximize conversion while maintaining selectivity. Detailed standard operating procedures regarding stoichiometry, temperature ramping, and quenching protocols are essential for reproducibility. For a comprehensive guide on executing these steps with optimal safety and yield, please refer to the standardized protocol below.
- Reduce linolenic acid ester to linoleyl alcohol using lithium aluminum hydride in tetrahydrofuran at 0-20°C.
- Convert linoleyl alcohol to linoleyl p-toluenesulfonate using p-toluenesulfonyl chloride and triethylamine in dichloromethane.
- Reduce the tosylate intermediate to (z,z,z)-3,6,9-octadecatriene using lithium aluminum hydride in diethyl ether.
- Perform selective epoxidation of the triene using hydrogen peroxide and a Keggin-type di-deficient silicotungstic heteropolyacid catalyst at 40-60°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this patented synthesis route offers tangible strategic benefits beyond mere technical feasibility. The substitution of exotic starting materials with commodity chemicals like linolenic acid esters decouples production costs from the volatility of specialized fine chemical markets. This stability in raw material sourcing ensures consistent pricing and availability, which is critical for long-term contracts with agricultural formulation companies. Furthermore, the elimination of hazardous reagents such as diazomethane removes the need for specialized explosion-proof infrastructure and extensive safety training, thereby lowering the barrier to entry for contract manufacturing organizations (CMOs) and reducing overall facility overheads.
- Cost Reduction in Manufacturing: The economic model of this process is driven by the drastic simplification of the synthetic sequence and the use of low-cost oxidants. By replacing expensive chiral catalysts and stoichiometric oxidants with a reusable heteropoly acid system and hydrogen peroxide, the direct material costs are significantly lowered. Additionally, the ability to telescope steps without intermediate isolation reduces solvent consumption and labor hours, contributing to substantial cost savings in agrochemical intermediate manufacturing. The avoidance of complex waste treatment for chlorinated byproducts further enhances the financial viability of the process on an industrial scale.
- Enhanced Supply Chain Reliability: Reliance on globally available feedstocks such as vegetable oil derivatives ensures a resilient supply chain that is less susceptible to geopolitical disruptions affecting specialty chemical imports. The robustness of the catalyst system allows for flexible production scheduling, as the reaction tolerances are wider than those of moisture-sensitive organometallic alternatives. This flexibility enables manufacturers to respond rapidly to seasonal spikes in demand for pest control agents, ensuring reducing lead time for high-purity agrochemical intermediates during critical planting seasons.
- Scalability and Environmental Compliance: The green nature of this synthesis aligns seamlessly with increasingly stringent environmental regulations governing chemical manufacturing. The use of hydrogen peroxide and the generation of water as a byproduct minimize the environmental footprint, facilitating easier permitting and compliance with local discharge standards. The process is inherently scalable from gram to ton quantities without significant re-engineering, supporting the commercial scale-up of complex pheromones required for area-wide pest management programs.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its potential for integration into existing production portfolios. The following questions address common inquiries regarding catalyst recovery, stereochemical integrity, and regulatory status. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a factual basis for decision-making.
Q: What are the advantages of using Keggin-type heteropoly acids for pheromone synthesis?
A: Keggin-type di-deficient silicotungstic heteropolyacids offer high selectivity and activity for epoxidizing polyunsaturated fatty acid derivatives. Unlike traditional oxidants like m-CPBA, they work efficiently with hydrogen peroxide, generating water as the only byproduct, which significantly simplifies waste treatment and reduces environmental impact.
Q: How does this method compare to previous synthesis routes in terms of safety?
A: Conventional methods often rely on hazardous reagents such as diazomethane for methylation or m-chloroperoxybenzoic acid for epoxidation, which pose explosion risks and generate chlorinated waste. This patented process eliminates these dangerous chemicals, utilizing stable hydrogen peroxide and recyclable catalysts, thereby enhancing operational safety for large-scale manufacturing.
Q: Can this synthesis route be scaled for commercial agrochemical production?
A: Yes, the process is designed for scalability by using inexpensive, commercially available starting materials like linolenic acid esters. The reaction conditions are mild (40-60°C) and do not require stringent anhydrous environments for the final oxidation step, making it highly suitable for cost reduction in agrochemical intermediate manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable cis-6,7-epoxy-(z,z)-3,9-octadecadiene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of green catalytic technologies in the agrochemical sector. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes like this heteropoly acid-catalyzed epoxidation are successfully translated into robust manufacturing operations. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced chromatographic and spectroscopic tools to guarantee the stereochemical fidelity of every batch of pheromone produced. Our commitment to quality assurance ensures that the biological activity of the final product meets the highest standards required for effective field application.
We invite industry partners to collaborate with us to leverage this cost-effective synthesis for their pest management portfolios. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your supply chain efficiency and product competitiveness in the global biopesticide market.
