Advanced Stereoselective Synthesis of Vitamin D Analogues for Commercial Scale Production
Introduction to Advanced Vitamin D Intermediate Synthesis
The pharmaceutical industry constantly seeks more efficient pathways for synthesizing complex active pharmaceutical ingredients (APIs), particularly for high-value compounds like vitamin D analogues such as calcitriol. Patent CN1179938C introduces a groundbreaking stereoselective method that addresses the longstanding challenges associated with constructing the A-ring portion of these molecules. This technology enables the conversion of specific epoxide precursors directly into allyl alcohol intermediates with exceptional control over stereochemistry at carbons 1 and 3. By leveraging a specialized palladium-catalyzed system, this process eliminates the need for the cumbersome multi-step sequences and extensive chromatographic purifications that have historically plagued vitamin D manufacturing. For R&D directors and process chemists, this represents a significant leap forward in synthetic efficiency, offering a route that maintains high fidelity to the desired stereochemical configuration while streamlining the overall production workflow.
The core innovation lies in the ability to transform compounds of Formula 1 into compounds of Formula 2 without scrambling the critical stereocenters. If the starting material possesses an R-configuration at carbon 3, the resulting product retains this configuration, ensuring that the biological activity inherent to the specific enantiomer is preserved throughout the synthesis. This level of precision is paramount in the production of hormonal therapies where even minor impurities can lead to significant regulatory hurdles. The patent details a robust chemical framework that supports the reliable production of high-purity pharmaceutical intermediates, positioning this technology as a cornerstone for modern vitamin D analogue manufacturing strategies aimed at both cost optimization and quality assurance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of vitamin D analogues has been fraught with inefficiencies that drive up costs and extend lead times for procurement teams. Traditional routes often rely on lengthy linear sequences involving numerous protection and deprotection steps, each introducing potential yield losses and impurity profiles that are difficult to manage. As noted in prior art references within the patent, such as those by Norman and Uskokovic, conventional methods frequently necessitate rigorous chromatographic purification to isolate the desired isomers from complex mixtures. This reliance on chromatography is a major bottleneck in commercial scale-up, as it limits batch sizes, increases solvent consumption, and complicates waste management protocols. Furthermore, the lack of stereoselectivity in older methodologies often results in the formation of diastereomers that are chemically similar but biologically inactive or even harmful, requiring additional downstream processing to remove.
From a supply chain perspective, these conventional processes are fragile. The sensitivity of intermediates to reaction conditions often leads to batch-to-batch variability, making it difficult for procurement managers to forecast accurate delivery schedules. The use of harsh reagents or extreme conditions in traditional routes can also degrade sensitive functional groups, leading to lower overall yields and higher raw material costs. Additionally, the environmental footprint of these older methods is substantial due to the high volume of organic solvents required for purification. For manufacturers aiming to meet increasingly stringent environmental regulations, continuing to rely on these outdated synthetic pathways presents a significant compliance risk and operational burden that undermines long-term sustainability goals.
The Novel Approach
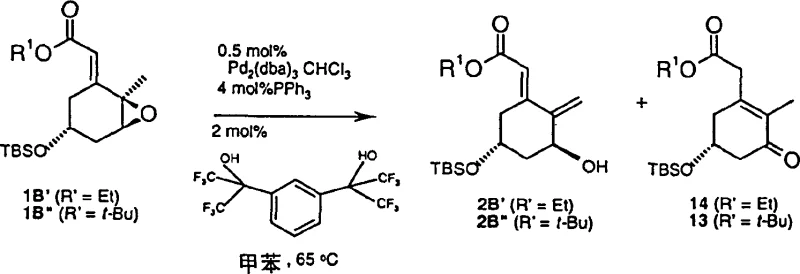
In stark contrast, the novel approach described in Patent CN1179938C utilizes a sophisticated palladium-catalyzed isomerization that dramatically simplifies the synthetic landscape. This method employs a palladium-phosphine catalyst system in conjunction with specific fluorinated alcohols to drive the reaction towards the desired allyl alcohol with remarkable selectivity. By operating under milder conditions, typically around 35°C in solvents like toluene, the process minimizes thermal degradation of sensitive intermediates. The key differentiator is the use of fluorinated alcohols with a pKa of less than 9, which act as crucial additives to protonate basic intermediates effectively, thereby suppressing the formation of unwanted enone byproducts. This chemical elegance translates directly into operational simplicity, allowing for the production of high-purity intermediates with minimal downstream processing.
This new methodology offers a transformative solution for cost reduction in pharmaceutical intermediates manufacturing. By achieving high selectivity ratios, often exceeding 19:1 in favor of the desired product, the need for extensive chromatographic separation is virtually eliminated. This not only reduces the consumption of expensive silica gel and solvents but also significantly shortens the cycle time for each batch. For supply chain heads, this means a more predictable and reliable production schedule, as the process is less susceptible to the variabilities associated with purification bottlenecks. The ability to produce the A-ring fragment with high stereochemical integrity from readily available starting materials like carvone further enhances the economic viability of this route, making it an attractive option for large-scale commercial production of vitamin D analogues.
Mechanistic Insights into Palladium-Catalyzed Isomerization
The mechanistic foundation of this process rests on the intricate interplay between the palladium catalyst, the phosphine ligand, and the fluorinated alcohol additive. The reaction initiates with the oxidative addition of the palladium(0) species to the allylic epoxide substrate, forming a pi-allyl palladium intermediate. In the absence of specific additives, this intermediate can undergo various pathways, often leading to a mixture of allyl alcohols and conjugated enones. However, the introduction of fluorinated alcohols with a pKa below 9 fundamentally alters the reaction trajectory. These acidic alcohols facilitate the protonation of the basic oxygen species generated during the ring-opening event, steering the equilibrium decisively towards the formation of the allyl alcohol. The patent data highlights a clear correlation between the acidity of the alcohol additive and the selectivity of the reaction, with stronger acids like hexafluoroisopropanol derivatives yielding superior results compared to weaker acids or non-fluorinated alcohols.
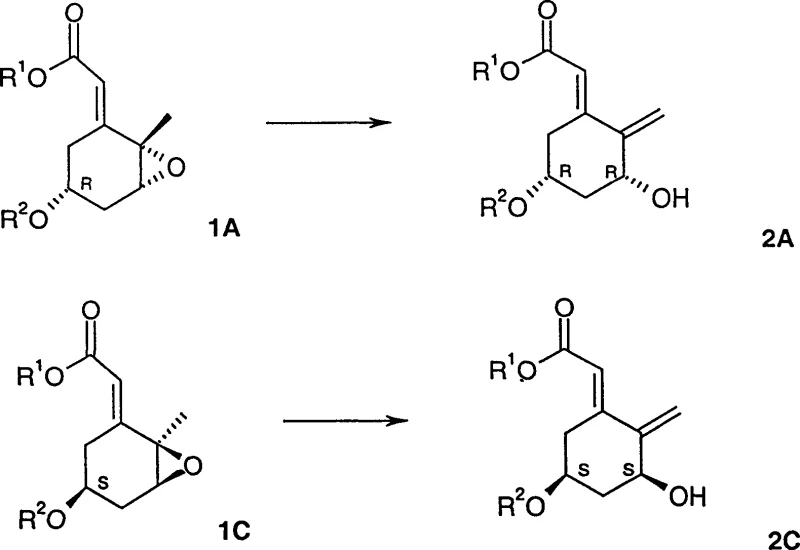
Furthermore, the choice of phosphine ligand plays a critical role in stabilizing the palladium center and modulating its electronic properties. Ligands such as triphenylphosphine or tris(2-methoxyphenyl)phosphine are effective in promoting the catalytic cycle while preventing the decomposition of the active species. The synergy between the ligand and the fluorinated alcohol ensures that the catalyst remains active over extended periods, allowing for the use of low catalyst loadings which is economically advantageous. This mechanistic understanding allows process chemists to fine-tune reaction conditions to maximize yield and purity. For instance, the patent demonstrates that increasing the concentration of the fluorinated alcohol can further improve the ratio of allyl alcohol to enone, providing a handle for optimizing the process for specific substrates. This level of control is essential for ensuring consistent product quality in a GMP manufacturing environment.
How to Synthesize Vitamin D Analogues Efficiently
Implementing this stereoselective synthesis requires careful attention to catalyst preparation and reaction parameters to ensure optimal performance. The process begins with the in situ generation of the active palladium catalyst by combining a palladium source, such as Pd2dba3(CHCl3), with an excess of a phosphine ligand in an inert solvent like toluene. Once the catalyst is formed, the fluorinated alcohol additive is introduced to the mixture, followed by the addition of the epoxide substrate. The reaction is then heated to moderate temperatures, typically around 35°C, and monitored until completion. The detailed standardized synthesis steps for this high-efficiency pathway are outlined in the guide below, providing a clear roadmap for technical teams looking to adopt this technology.
- Prepare the palladium catalyst in situ by mixing Pd2dba3(CHCl3) and a phosphine ligand such as triphenylphosphine in toluene under nitrogen.
- Add a fluorinated alcohol with a pKa less than 9, such as 1,3-bis(1,1,1,3,3,3-hexafluoro-2-hydroxypropyl)benzene, to the catalyst mixture to enhance selectivity.
- Introduce the epoxide substrate (Formula 1) to the reaction mixture and heat to 35°C to facilitate the stereoselective isomerization to the allyl alcohol (Formula 2).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented synthesis route offers compelling strategic advantages that extend beyond simple chemical efficiency. The primary benefit lies in the substantial cost savings achieved through process intensification. By eliminating the need for multiple chromatographic purification steps, manufacturers can significantly reduce the consumption of solvents and stationary phases, which are major cost drivers in fine chemical production. This reduction in material usage directly translates to lower variable costs per kilogram of product. Moreover, the simplified workflow reduces the labor hours required for processing and quality control, further enhancing the overall economic profile of the manufacturing operation. These efficiencies make the final vitamin D analogues more price-competitive in the global market.
- Cost Reduction in Manufacturing: The streamlined nature of this palladium-catalyzed process removes several unit operations that are traditionally expensive and time-consuming. By avoiding complex purification sequences, facilities can operate with smaller equipment footprints and lower utility consumption. The high selectivity of the reaction means that raw materials are converted into the desired product with minimal waste, maximizing the return on investment for every kilogram of starting material purchased. This efficiency is crucial for maintaining healthy margins in the competitive pharmaceutical intermediates sector.
- Enhanced Supply Chain Reliability: A simpler process is inherently more robust and easier to control, leading to greater consistency in production output. The use of stable reagents and moderate reaction conditions reduces the risk of batch failures due to thermal runaways or reagent degradation. This reliability allows supply chain planners to commit to tighter delivery schedules with confidence, knowing that the production process is less prone to unexpected disruptions. Additionally, the scalability of the chemistry ensures that supply can be ramped up quickly to meet surges in demand without compromising on quality.
- Scalability and Environmental Compliance: The reduction in solvent waste and hazardous byproducts aligns perfectly with modern green chemistry principles and environmental regulations. Facilities implementing this process will find it easier to comply with strict discharge limits and waste disposal mandates. The ability to scale this reaction from laboratory benchtop to multi-ton commercial production without significant re-engineering provides a clear path for long-term growth. This scalability ensures that the supply of critical vitamin D intermediates remains secure and sustainable, supporting the continuous availability of life-saving medications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this stereoselective synthesis technology. These answers are derived directly from the experimental data and specifications provided in Patent CN1179938C, ensuring accuracy and relevance for decision-makers evaluating this process for their own manufacturing pipelines. Understanding these nuances is key to successfully integrating this advanced chemistry into existing production frameworks.
Q: What is the key advantage of using fluorinated alcohols in this synthesis?
A: Fluorinated alcohols with a pKa below 9 significantly increase the selectivity for the desired allyl alcohol product over unwanted enone byproducts, often achieving ratios greater than 19:1.
Q: Can this process be scaled for commercial manufacturing?
A: Yes, the process utilizes robust conditions including moderate temperatures (35°C) and common solvents like toluene, making it highly suitable for scaling from laboratory to multi-ton production.
Q: How does this method improve upon conventional vitamin D synthesis?
A: Unlike conventional methods that require multiple steps and extensive chromatographic purification, this stereoselective approach minimizes purification needs and preserves stereochemistry at carbons 1 and 3.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vitamin D Analogues Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a manufacturing partner who understands the complexities of synthesizing high-value pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. We are equipped with rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch of vitamin D analogues meets the highest industry standards. Our commitment to quality and consistency makes us the preferred choice for global pharmaceutical companies seeking a dependable source for their key intermediates.
We invite you to collaborate with us to explore how this advanced synthesis technology can optimize your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production volumes. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your project goals. Let us help you secure a stable and cost-effective supply of high-purity vitamin D analogues for your next commercial launch.
