Advanced Nickel-Catalyzed Synthesis of Cis-Configuration Unsaturated Esters for Commercial Fragrance Applications
Advanced Nickel-Catalyzed Synthesis of Cis-Configuration Unsaturated Esters for Commercial Fragrance Applications
The global fragrance and flavor industry constantly seeks novel compounds that offer unique olfactory profiles while maintaining economic viability in production. Patent CN1135220C introduces a groundbreaking methodology for the synthesis of cis-configuration unsaturated esters, a class of compounds renowned for their distinct green, fruity, and floral notes. This technology addresses the longstanding challenge of achieving high stereoselectivity in the production of unsaturated esters, which is crucial because the olfactory properties of cis and trans isomers can differ drastically. The patent details a robust catalytic system that enables the efficient conversion of cyclic vinyl ethers into valuable fragrance intermediates with exceptional purity. By leveraging a specialized nickel-catalyzed Grignard reaction, manufacturers can access compounds such as 3-methyl-cis-4-hexenyl acetate, which possesses a desirable pear or apple-like aroma, and 2-methyl-cis-3-pentenyl acetate, known for its apple and floral scent. 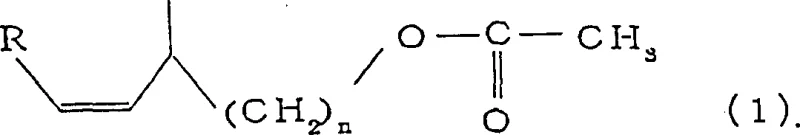 This innovation not only expands the palette of available fragrance ingredients but also provides a scalable route for reliable fragrance intermediate supplier operations, ensuring consistent quality for high-end perfume and cosmetic formulations.
This innovation not only expands the palette of available fragrance ingredients but also provides a scalable route for reliable fragrance intermediate supplier operations, ensuring consistent quality for high-end perfume and cosmetic formulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for producing unsaturated alcohols and their corresponding esters often suffer from significant drawbacks regarding stereoselectivity and catalyst efficiency. Conventional methods frequently rely on higher loadings of transition metal catalysts, which not only increases the raw material costs but also complicates the downstream purification processes due to heavy metal residues. In many historical protocols, the ratio of cis to trans isomers is difficult to control, often resulting in mixtures that require expensive and time-consuming separation techniques like fractional distillation or preparative chromatography to isolate the desired cis-isomer. Furthermore, older methodologies may utilize harsh reaction conditions or less stable intermediates that pose safety risks during commercial scale-up of complex fragrance intermediates. The inability to consistently achieve high cis-selectivity means that a portion of the production capacity is wasted on trans-isomers that may possess undesirable odor characteristics or no olfactory value at all, thereby reducing the overall atom economy and profitability of the manufacturing process.
The Novel Approach
The methodology disclosed in CN1135220C represents a paradigm shift by employing a highly efficient nickel-based catalytic system that operates with remarkably low catalyst loading. This novel approach utilizes a combination of a nickel compound and a triarylphosphine ligand to facilitate the ring-opening of cyclic vinyl ethers by Grignard reagents. The key breakthrough lies in the ability to maintain the cis-configuration of the double bond throughout the reaction, achieving cis/trans ratios of up to 99.1/0.9 or even 100/0 in optimized examples. This high selectivity eliminates the need for complex isomer separation, directly translating to cost reduction in synthetic flavors manufacturing. Additionally, the process allows for the use of widely available starting materials such as 3-methyl-2,3-dihydrofuran and 4-methyl-3,4-dihydro-2H-pyran, which react smoothly under moderate temperatures ranging from 30°C to 100°C. The versatility of this system allows for the introduction of various R groups, including methyl and phenyl, enabling the synthesis of a diverse library of esters tailored to specific fragrance applications without compromising on yield or purity.
Mechanistic Insights into Nickel-Catalyzed Grignard Ring-Opening
The core of this technological advancement is the precise interaction between the nickel catalyst, the phosphine ligand, and the cyclic vinyl ether substrate. The reaction mechanism involves the oxidative addition of the nickel species to the cyclic vinyl ether, followed by the nucleophilic attack of the Grignard reagent. 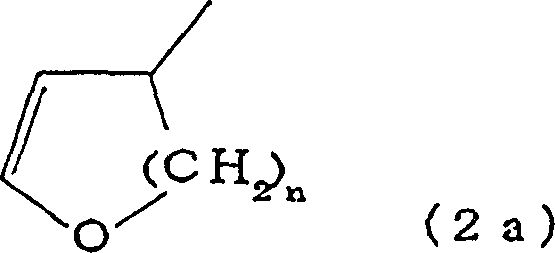 The presence of the triarylphosphine ligand, such as triphenylphosphine, is critical as it stabilizes the nickel center and modulates its electronic properties to favor the formation of the cis-alkene product. The patent specifies that the molar ratio of the triarylphosphine to the nickel atom can range from 0.1 to 3000, with preferred ranges between 2 and 2000 to maximize both reaction rate and selectivity. This ligand environment prevents the isomerization of the double bond, which is a common side reaction in metal-catalyzed alkene formations. The reaction proceeds through a concerted pathway where the stereochemistry of the starting cyclic ether is translated into the linear cis-unsaturated alcohol with high fidelity. Understanding this mechanistic nuance is vital for R&D teams aiming to replicate these results, as deviations in ligand purity or nickel source can impact the delicate balance required for high-purity cis-configuration unsaturated ester production.
The presence of the triarylphosphine ligand, such as triphenylphosphine, is critical as it stabilizes the nickel center and modulates its electronic properties to favor the formation of the cis-alkene product. The patent specifies that the molar ratio of the triarylphosphine to the nickel atom can range from 0.1 to 3000, with preferred ranges between 2 and 2000 to maximize both reaction rate and selectivity. This ligand environment prevents the isomerization of the double bond, which is a common side reaction in metal-catalyzed alkene formations. The reaction proceeds through a concerted pathway where the stereochemistry of the starting cyclic ether is translated into the linear cis-unsaturated alcohol with high fidelity. Understanding this mechanistic nuance is vital for R&D teams aiming to replicate these results, as deviations in ligand purity or nickel source can impact the delicate balance required for high-purity cis-configuration unsaturated ester production.
Impurity control is inherently built into this catalytic cycle due to the high specificity of the nickel-phosphine complex. The low catalyst loading, typically between 0.00001 and 0.05 mol per mole of substrate, minimizes the generation of metal-containing byproducts that could contaminate the final fragrance ingredient. In comparative examples provided in the patent, increasing the nickel loading without the optimal phosphine ratio led to a significant drop in cis-selectivity, with cis/trans ratios falling to 85.3/14.7. This demonstrates that the specific catalytic environment is what drives the selectivity, rather than just the presence of the metal. Furthermore, the reaction conditions are optimized to suppress competing pathways such as polymerization or over-reduction. The workup procedure involves quenching with aqueous ammonium chloride or dilute acid, followed by extraction with organic solvents like toluene or hexane, which effectively removes the catalyst residues and magnesium salts. This streamlined purification process ensures that the final product meets the stringent purity specifications required for use in cosmetics and food flavorings, where trace impurities can have significant regulatory and sensory implications.
How to Synthesize 3-Methyl-Cis-4-Hexenyl Acetate Efficiently
The synthesis of high-value fragrance esters like 3-methyl-cis-4-hexenyl acetate follows a logical two-step sequence that is amenable to both laboratory and pilot plant scales. The first step involves the preparation of the cis-unsaturated alcohol intermediate via the nickel-catalyzed reaction of 4-methyl-3,4-dihydro-2H-pyran with methylmagnesium bromide. This step requires careful control of temperature, typically initiating at 20°C and ramping to 50-70°C, under an inert nitrogen atmosphere to prevent oxidation of the sensitive organometallic species. The detailed standardized synthesis steps see the guide below. The second step is the acylation of the resulting alcohol, which is a straightforward esterification reaction. By reacting the crude or purified cis-alcohol with acetic anhydride in the presence of a base like 4-dimethylaminopyridine (DMAP) or triethylamine, the hydroxyl group is converted into an acetate ester. This acylation step is highly exothermic and requires temperature control to remain below 40°C to prevent degradation of the unsaturated bond. The final product is isolated via aqueous workup and distillation, yielding a colorless liquid with a boiling point around 84°C at reduced pressure. This protocol exemplifies how modern catalytic methods can simplify the production of complex molecules, making them accessible for commercial application.
- Prepare a catalytic system using a nickel compound (e.g., dichlorobis(triphenylphosphine)nickel(II)) and triarylphosphine ligand in an aromatic solvent under inert atmosphere.
- React the cyclic vinyl ether substrate with a Grignard reagent (RMgX) at controlled temperatures between 30°C and 100°C to generate the cis-unsaturated alcohol intermediate with high stereoselectivity.
- Perform acylation of the resulting cis-unsaturated alcohol using acetic anhydride or acetyl chloride in the presence of a base to yield the final cis-configuration unsaturated ester product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the technology described in CN1135220C offers tangible strategic benefits beyond mere technical superiority. The shift towards this catalytic method addresses several pain points associated with traditional fragrance ingredient sourcing, particularly regarding cost stability and supply continuity. By optimizing the catalyst system to use minimal amounts of nickel, the process significantly reduces the dependency on expensive transition metals, which are subject to volatile market pricing. This reduction in catalyst consumption directly lowers the variable cost of goods sold, allowing for more competitive pricing structures in long-term supply contracts. Moreover, the high selectivity of the reaction means that less raw material is wasted on unwanted isomers, improving the overall yield and throughput of the manufacturing facility. This efficiency gain translates into a more reliable supply chain, as production batches are more predictable and less prone to failure due to selectivity issues.
- Cost Reduction in Manufacturing: The implementation of this low-loading nickel catalyst system eliminates the need for expensive heavy metal scavengers that are typically required to meet purity standards in pharmaceutical and fine chemical grades. By minimizing the initial metal input, the burden on downstream purification units is drastically reduced, leading to substantial cost savings in solvent usage and waste disposal. The qualitative improvement in atom economy ensures that a higher percentage of the starting material ends up in the final product, effectively lowering the cost per kilogram of the active fragrance ingredient. Additionally, the use of commercially available cyclic vinyl ethers and standard Grignard reagents avoids the need for custom-synthesized precursors, further streamlining the procurement budget and reducing lead times for raw material acquisition.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain resilience by reducing the complexity of the manufacturing process. Since the reaction tolerates a range of conditions and uses stable starting materials, the risk of batch-to-batch variability is minimized, ensuring consistent delivery schedules for customers. The ability to source key intermediates like dihydrofurans and pyrans from multiple global suppliers mitigates the risk of single-source bottlenecks. This flexibility allows supply chain heads to build more agile inventory strategies, knowing that the production process is not reliant on exotic or hard-to-find reagents. Consequently, this leads to reducing lead time for high-purity fragrance intermediates, enabling faster response to market demands for new scent profiles.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this process aligns well with modern green chemistry principles. The reduced metal loading decreases the heavy metal content in the effluent, simplifying wastewater treatment and ensuring compliance with increasingly strict environmental regulations. The reaction can be safely scaled from 100 kgs to 100 MT/annual commercial production without significant re-engineering, as the thermal profile is manageable and does not require extreme pressures or cryogenic temperatures. This scalability ensures that the technology can grow with market demand, providing a future-proof solution for fragrance manufacturers looking to expand their portfolio of sustainable and high-performance ingredients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these cis-configuration unsaturated esters. These answers are derived directly from the experimental data and claims found in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing supply chains. These insights are crucial for assessing the potential return on investment and the technical readiness of the process for immediate deployment in industrial settings.
Q: What is the primary advantage of the nickel-catalyzed method described in CN1135220C?
A: The primary advantage is the exceptional stereoselectivity for the cis-configuration, achieving cis/trans ratios as high as 99.1/0.9 or even 100/0, which is critical for specific fragrance profiles that trans-isomers cannot replicate.
Q: How does the catalyst loading in this process compare to conventional methods?
A: This process utilizes significantly lower catalyst loading, typically ranging from 0.00001 to 0.05 mol of nickel compound per mole of cyclic vinyl ether, compared to approximately 0.1 mol in conventional methods, leading to substantial cost reductions.
Q: Can this synthesis method be scaled for industrial production of fragrance ingredients?
A: Yes, the method is designed for industrial feasibility, utilizing commercially available starting materials like 3-methyl-2,3-dihydrofuran and standard Grignard reagents, with reaction conditions (30-100°C) that are manageable in large-scale reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cis-Configuration Unsaturated Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of next-generation fragrance compositions. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative nickel-catalyzed routes described in CN1135220C can be successfully transferred to full-scale manufacturing. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with advanced analytical instrumentation like GC-MS and NMR. Our capability to handle complex organometallic chemistry allows us to offer a reliable fragrance intermediate supplier partnership that guarantees consistency and quality. Whether you require 3-methyl-cis-4-hexenyl acetate for a fruity note or phenyl-substituted variants for floral accords, our infrastructure is ready to support your volume requirements with precision and reliability.
We invite you to collaborate with us to explore how this advanced synthesis technology can enhance your product portfolio. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production volumes. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can drive value for your organization. Let us help you secure a stable supply of high-purity cis-configuration unsaturated esters that will set your fragrance formulations apart in the competitive global market.
