Scalable Synthesis of Restricted 9-cis-Retinoic Acid Analogs for Cardiovascular Therapeutics
Scalable Synthesis of Restricted 9-cis-Retinoic Acid Analogs for Cardiovascular Therapeutics
The pharmaceutical landscape for cardiovascular disease management is continually evolving, with a significant focus on modulating lipid profiles to mitigate the risks associated with coronary artery disease (CAD). Patent CN1176248A introduces a novel class of restricted 9-cis-retinoic acid analogs that demonstrate potent efficacy in increasing high-density lipoprotein (HDL) levels, thereby offering protection against early-stage atherosclerosis. These compounds function by enhancing the expression of apolipoprotein A-I (Apo A-I), the major protein component of plasma HDL, through specific interactions with retinoic acid receptors (RXRα). The structural versatility of these analogs allows for fine-tuning of biological activity while maintaining metabolic stability. As a leading entity in fine chemical manufacturing, understanding the synthetic accessibility of these complex molecules is crucial for ensuring a reliable supply chain for downstream drug development.
The core innovation lies in the specific substitution patterns on the naphthalene or tetrahydronaphthalene rings, which restrict the conformational freedom of the molecule compared to natural retinoids. This restriction enhances receptor selectivity and reduces potential off-target effects often seen with broader-spectrum retinoids. The general structure, designated as Formula I, encompasses a variety of substituents including alkyl, alkoxy, and trifluoromethyl groups, providing a broad chemical space for optimization. For procurement specialists and R&D teams, the ability to access these specific scaffolds reliably is paramount. 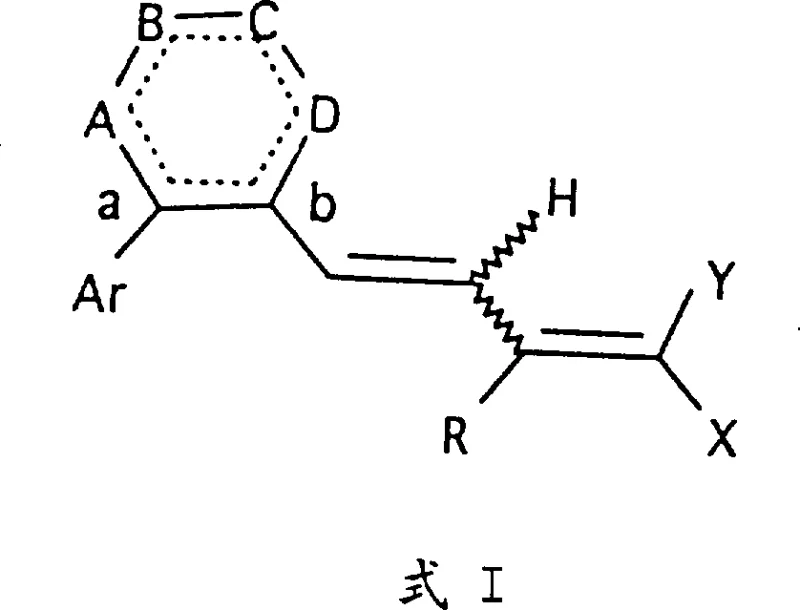
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of retinoid analogs often relies on classical condensation reactions that can suffer from poor stereochemical control and harsh reaction conditions. Conventional routes frequently involve high-temperature isomerization steps that lead to mixtures of cis and trans isomers, necessitating difficult and costly separation processes to isolate the biologically active 9-cis form. Furthermore, older methodologies may utilize unstable intermediates that degrade rapidly upon exposure to light or oxygen, resulting in lower overall yields and inconsistent batch-to-batch quality. The reliance on non-selective catalysts in earlier generations of retinoid synthesis often produced significant impurity profiles, complicating the purification process and increasing the cost of goods sold (COGS). These factors collectively pose significant challenges for scaling up production to meet commercial demand for cardiovascular therapeutics.
The Novel Approach
The methodology outlined in CN1176248A represents a significant advancement by employing palladium-catalyzed cross-coupling reactions to construct the critical carbon-carbon bonds under milder conditions. This approach allows for the precise assembly of the aryl-vinyl framework with high fidelity, minimizing the formation of unwanted geometric isomers. By utilizing organozinc intermediates generated in situ from aryl bromides, the process achieves excellent chemoselectivity, tolerating various functional groups present on the naphthalene ring system. The subsequent functionalization steps, including controlled reductions and oxidations, are designed to preserve the integrity of the polyene chain. This strategic design not only improves the purity of the final product but also streamlines the workflow, making it highly attractive for industrial application where efficiency and reproducibility are key drivers of success.
Mechanistic Insights into Palladium-Catalyzed Cross-Coupling
The heart of this synthetic strategy is the Negishi-type coupling reaction, which facilitates the union of the aromatic core with the polyene side chain precursor. The mechanism initiates with the lithiation of an aryl bromide species using tert-butyllithium at low temperatures, typically around -78°C, to generate a reactive aryl lithium intermediate. This species is subsequently transmetallated with zinc chloride to form an organozinc reagent, which is more tolerant of functional groups and less prone to side reactions than its lithium counterpart. The organozinc species then reacts with a vinyl triflate in the presence of a tetrakis(triphenylphosphine)palladium(0) catalyst. This catalytic cycle involves oxidative addition of the vinyl triflate to the palladium center, followed by transmetallation with the organozinc compound and reductive elimination to forge the new C-C bond. This sequence is highly efficient and proceeds with retention of configuration at the double bond, ensuring the desired stereochemistry is maintained throughout the synthesis.
Following the construction of the core skeleton, the side chain is elaborated through a series of functional group interconversions that allow for the introduction of various terminal groups such as carboxylic acids, esters, or amides. A key transformation involves the reduction of ester intermediates to primary alcohols using hydride reducing agents like lithium aluminum hydride, followed by selective oxidation to aldehydes using activated manganese dioxide. 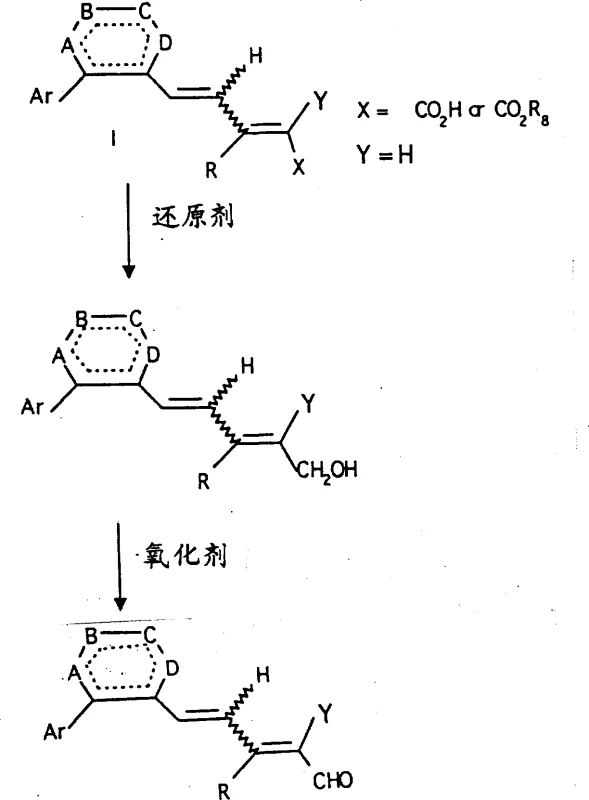
This aldehyde serves as a versatile pivot point for the final olefination step, where a Horner-Wadsworth-Emmons or Wittig reaction installs the remaining unsaturated units of the side chain. The choice of base and reaction conditions in this final step dictates the E/Z ratio of the newly formed double bond, allowing chemists to tune the biological profile of the analog. The robustness of these mechanistic steps ensures that the process can be adapted for the synthesis of a wide library of analogs by simply varying the starting aryl halide or the phosphonate reagent. This modularity is a significant advantage for R&D teams looking to explore structure-activity relationships (SAR) without redesigning the entire synthetic route from scratch.
How to Synthesize Restricted 9-cis-Retinoic Acid Analogs Efficiently
The synthesis of these high-value pharmaceutical intermediates requires precise control over reaction parameters to ensure optimal yield and purity. The process begins with the preparation of the organometallic reagent under inert atmosphere conditions to prevent quenching by moisture or oxygen. Subsequent coupling reactions are typically conducted in ethereal solvents such as tetrahydrofuran, which solubilize both the organic substrates and the metal complexes effectively. Work-up procedures involve standard aqueous extractions and drying steps, followed by purification via column chromatography or recrystallization depending on the physical properties of the specific analog. Detailed operational protocols are essential for maintaining consistency, particularly when handling sensitive reagents like organolithiums and palladium catalysts. For a comprehensive guide on the specific stoichiometry and temperature profiles required for each step, please refer to the standardized synthesis instructions below.
- Preparation of organozinc intermediate from aryl bromide using tert-butyllithium and zinc chloride.
- Palladium-catalyzed coupling of the organozinc species with a vinyl triflate to form the core skeleton.
- Functional group manipulation including reduction to alcohol, oxidation to aldehyde, and final Wittig-type olefination.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the synthetic route described in this patent offers several distinct advantages that translate into enhanced reliability and potential cost efficiencies. The starting materials, such as brominated tetrahydronaphthalenes and vinyl triflates, are commercially available or can be synthesized from commodity chemicals, reducing the risk of raw material shortages. The use of established catalytic systems like palladium tetrakis means that the technology is well-understood and can be implemented in standard multipurpose reactors without the need for specialized high-pressure equipment. This compatibility with existing infrastructure significantly lowers the barrier to entry for contract manufacturing organizations (CMOs) and facilitates faster technology transfer. Furthermore, the ability to purify intermediates via crystallization rather than relying solely on chromatography reduces solvent consumption and waste generation, aligning with modern green chemistry principles and environmental compliance standards.
- Cost Reduction in Manufacturing: The streamlined nature of the coupling reactions minimizes the number of unit operations required to reach the final target molecule. By avoiding complex protection-deprotection sequences often seen in older retinoid syntheses, the overall process time is reduced, leading to lower labor and utility costs. Additionally, the high selectivity of the palladium-catalyzed step reduces the burden on downstream purification, allowing for higher throughput in finishing operations. The elimination of expensive transition metals other than palladium, which can be recovered and recycled, further contributes to a favorable economic profile for large-scale production runs.
- Enhanced Supply Chain Reliability: The robustness of the chemical transformations ensures consistent output quality, which is critical for maintaining uninterrupted supply to downstream API manufacturers. The tolerance of the reaction conditions to minor variations in temperature or reagent grade means that the process is less susceptible to batch failures caused by raw material fluctuations. This stability allows for better inventory planning and reduces the need for safety stock, optimizing working capital for procurement managers. Moreover, the modular nature of the synthesis allows for the rapid qualification of alternative suppliers for key building blocks, mitigating single-source risks.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing solvents and reagents that are manageable on a multi-kilogram to ton scale. The waste streams generated are primarily aqueous and organic solvents that can be treated using standard effluent treatment protocols, simplifying regulatory compliance. The avoidance of highly toxic reagents or extreme conditions enhances workplace safety and reduces the environmental footprint of the manufacturing facility. This alignment with sustainability goals is increasingly important for pharmaceutical companies seeking to partner with suppliers who demonstrate a commitment to responsible manufacturing practices.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders involved in the development and sourcing of cardiovascular therapeutics. The following questions address common inquiries regarding the chemical properties, regulatory status, and logistical aspects of these intermediates. The answers provided are based on the technical disclosures within the patent literature and general industry knowledge regarding retinoid chemistry. For specific technical data sheets or custom synthesis queries, direct engagement with the technical team is recommended to ensure all project requirements are met accurately.
Q: What is the primary therapeutic application of these 9-cis-retinoic acid analogs?
A: These analogs are designed to increase HDL levels and treat coronary artery disease by modulating apolipoprotein A-I expression through RXRα receptors.
Q: How is stereochemistry controlled during the synthesis?
A: Stereochemistry is controlled through specific olefination conditions using phosphonates or ylides, yielding predominantly E,E or Z,E isomers as required.
Q: Are these intermediates suitable for large-scale manufacturing?
A: Yes, the process utilizes standard reagents and purification techniques like crystallization, making it amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 9-cis-Retinoic Acid Analog Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of life-saving medications. Our facility is equipped with state-of-the-art reactor trains capable of handling sensitive organometallic chemistry under strict inert conditions, ensuring that every batch meets the rigorous standards required for pharmaceutical applications. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, allowing us to support your project from early-stage clinical trials through to full commercial launch. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize advanced analytical techniques to verify identity and potency. We understand that time-to-market is crucial, and our agile manufacturing processes are designed to accelerate your supply chain without compromising on safety or compliance.
We invite you to collaborate with us to optimize your sourcing strategy for these complex retinoid analogs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. By leveraging our expertise in process optimization, we can help identify opportunities to reduce lead times and improve overall project economics. Please contact our technical procurement team today to request specific COA data and route feasibility assessments for your next project. Let us be your partner in bringing innovative cardiovascular therapies to patients worldwide through reliable and efficient chemical manufacturing.
